Clear Sky Science · en
Aminoadipate-semialdehyde synthase, a potential target for substrate reduction therapy in glutaric aciduria type 1
Why this rare childhood disease matters
Glutaric aciduria type 1 is a rare inherited disorder that mostly affects infants and young children, but it illustrates a much broader idea: how tweaking a single step in the body’s chemistry can protect the brain from lifelong damage. Despite modern newborn screening and careful diets, many children with this condition still develop movement problems, brain changes, and kidney issues. This study explores a new way to cut off the production of harmful substances at their source, using a strategy called substrate reduction therapy, and tests whether it could lead to safer and more effective treatments.
A traffic jam in the body’s chemistry
In glutaric aciduria type 1, the body cannot properly break down certain building blocks of protein, especially the amino acid lysine. A missing or faulty enzyme known as glutaryl-CoA dehydrogenase causes a metabolic traffic jam, leading to the buildup of glutaric acid and related compounds. These substances are particularly toxic to specific brain regions that help control movement, especially early in life. Even when children are identified shortly after birth and placed on special low-lysine diets, about one in three still develop serious neurological symptoms, and many show progressive white matter and kidney problems as they grow older.
A new place to put up a roadblock
Current treatment focuses on limiting how much lysine enters the system and helping the body remove toxic by-products, but it does not directly change how the faulty pathway works. The researchers asked a key question: instead of trying to handle the accumulating waste, could we slow down the input into the faulty pathway itself? They focused on another enzyme called aminoadipate-semialdehyde synthase, which carries out the first two steps in one of the main lysine breakdown routes. People who naturally lack this enzyme typically have high lysine levels but few or no health problems, suggesting that blocking it might be relatively safe.
Testing the idea in engineered mice
To probe this strategy, the team used mice bred to lack glutaryl-CoA dehydrogenase, a well-established model of the human disease. These animals accumulate large amounts of toxic acids in their tissues and, when challenged with a lysine-rich diet, develop seizures, movement problems, brain damage, and often die. The researchers then created a double knockout mouse that lacked both the disease enzyme and aminoadipate-semialdehyde synthase. Under standard feeding conditions, these double knockout mice looked and behaved like healthy animals in a wide battery of tests, while still showing only mild biochemical abnormalities.
Less toxic buildup and better brain health
The real test came when both types of disease-model mice were exposed to a high-lysine diet designed to stress the vulnerable pathway. Mice missing only the disease enzyme accumulated very high levels of toxic acids in the brain, liver, kidney, blood, and urine, and many developed severe neurological symptoms, seizures, and weight loss. In contrast, the double knockout animals had dramatically lower glutaric acid levels in all these tissues, far fewer signs of illness, and near-normal behavior. Microscopic examination of their brains showed that the tiny vacuoles and structural changes seen in the single knockout mice were greatly reduced or absent in the double knockouts. Importantly, detailed checks of body composition, bone density, heart function, immune markers, and blood chemistry found no major non-neurological problems caused by blocking this early step in lysine breakdown.
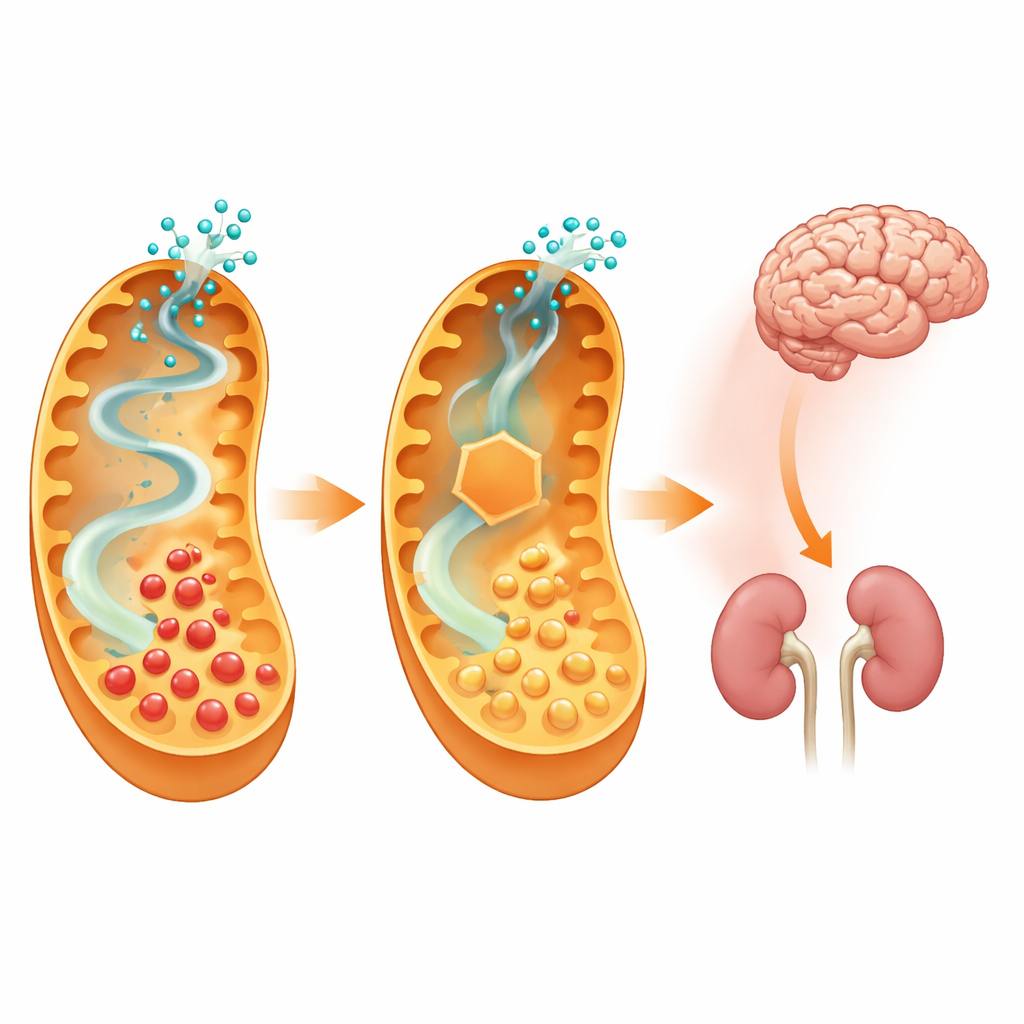
What this means for future treatments
By shutting down the first step of a specific lysine pathway, the researchers were able to lower the brain’s exposure to toxic metabolites and largely protect mice from a severe, diet-triggered disease flare, without causing obvious new health issues. This suggests that drugs designed to partially inhibit aminoadipate-semialdehyde synthase could offer a powerful new tool alongside diet and other treatments for children with glutaric aciduria type 1. Although the current work relies on genetic engineering in mice, and suitable medicines must still be developed and tested for safety in humans, the study provides strong proof of principle that carefully targeted blockage of upstream chemistry can reshape the course of a devastating metabolic brain disease.
Citation: Saad, C., Jung-Klawitter, S., Dimitrov, B. et al. Aminoadipate-semialdehyde synthase, a potential target for substrate reduction therapy in glutaric aciduria type 1. Sci Rep 16, 10995 (2026). https://doi.org/10.1038/s41598-026-44377-9
Keywords: glutaric aciduria type 1, lysine metabolism, substrate reduction therapy, AASS inhibition, inherited metabolic disorders