Clear Sky Science · en
ID1 in TAMs promoted the progression of non-small-cell carcinoma via increasing NF-κB/NPM1/SHP1/SHP2 signaling induced M2 polarization
Why the body’s defenders can turn into helpers of lung cancer
Lung cancer remains one of the deadliest cancers worldwide, in part because many tumors learn to hide from and even exploit the body’s own immune system. This study looks at a special group of immune cells that gather inside lung tumors and asks a simple but crucial question: what makes these cells switch from fighting cancer to helping it grow? Understanding that switch could open the door to new treatments that re-train the immune system rather than just attacking the tumor directly.
Immune cells that switch sides
Inside every tumor is a bustling neighborhood of non-cancer cells called the tumor microenvironment. Among its most important residents are macrophages, immune cells normally tasked with swallowing germs and damaged tissue. In lung tumors, many of these cells, known as tumor-associated macrophages, settle into a so‑called “M2” state that soothes inflammation, repairs tissue, and, unfortunately, nurtures cancer. Patients with non‑small‑cell lung cancer, the most common form of lung cancer, often have many of these M2‑type macrophages, which are linked to rapid tumor growth, spread, and poor responses to therapy.
A master switch called ID1
The researchers focused on a protein named ID1, already known to push various cancers toward more aggressive behavior. They suspected that ID1 in tumor‑associated macrophages helps steer these cells into their cancer‑supporting M2 state. Using public gene databases, they first showed that ID1 levels rise together with a key control factor called NF‑κB in lung cancer samples, hinting that these molecules work as partners. They then built laboratory models of human macrophages and used genetic tools and drugs to either boost ID1 or block NF‑κB, carefully tracking how this altered other molecules in the same chain.
How ID1 rewires macrophages
In these macrophages, higher ID1 levels turned on NF‑κB and another protein, NPM1, which together activated a pair of enzymes called SHP1 and SHP2. This chain reaction pushed NF‑κB subunits into the cell nucleus, where they switched on genes that favor the M2‑like state. As a result, the macrophages began to produce higher amounts of substances such as IL‑4, IL‑10, arginase‑1, CD206, and several cathepsins—molecules known to dampen anti‑tumor immunity, remodel tissue, and make it easier for cancer cells to move and invade. When scientists interrupted this signaling, either by blocking NF‑κB or by carefully adjusting NPM1, the levels of SHP1, SHP2, and these M2‑associated substances dropped, and the macrophages looked less tumor‑friendly.
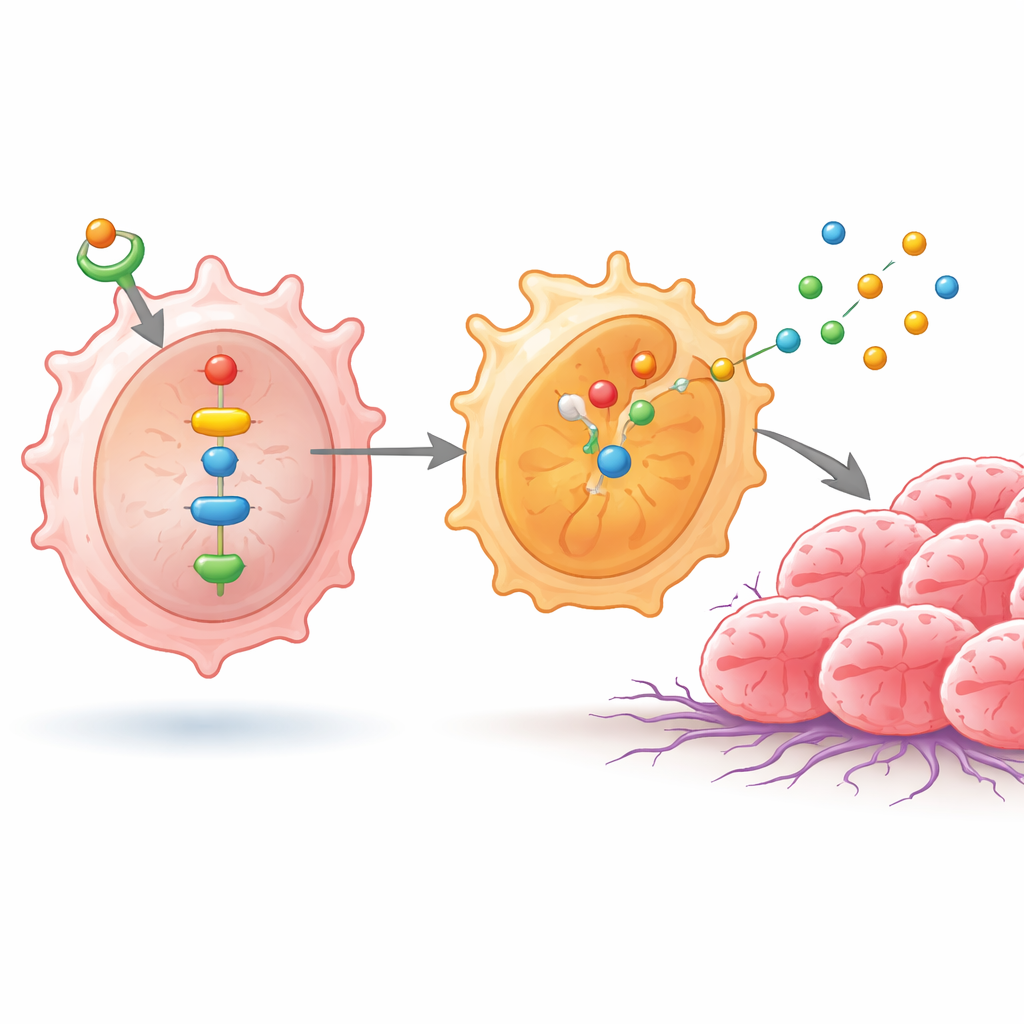
How reprogrammed macrophages boost tumor growth
To see how these immune changes affect cancer cells, the team grew lung cancer cell lines together with the differently treated macrophages. When ID1 was high and the signaling chain intact, the cancer cells multiplied faster and were better at migrating through artificial barriers—behaviors linked to spread within the body. When NF‑κB was blocked, these aggressive traits fell off, but they came back if NPM1 was forced back on, underscoring the importance of this pathway. The researchers then moved to mice, implanting mixtures of lung cancer cells and treated macrophages under the skin. Tumors formed alongside ID1‑rich, M2‑like macrophages grew larger and heavier, whereas tumors paired with pathway‑blocked macrophages remained smaller. Human lung tumor samples also showed higher levels of ID1 and activated NF‑κB compared with nearby non‑cancerous tissue, tying the laboratory findings to real patients.
What this means for future lung cancer treatments
Taken together, the work paints ID1 as an important “master switch” that helps turn helpful immune cells into silent partners of lung cancer. By driving a chain of events through NF‑κB, NPM1, and SHP1/SHP2, ID1 nudges macrophages into an M2‑like state that calms immune attacks and releases growth‑promoting factors, thereby speeding tumor progression. For patients, the message is hopeful: instead of targeting only the cancer cells, future therapies could aim to cut this signaling chain or flip macrophages back toward a cancer‑fighting mode. While these findings still need to be tested and refined in clinical settings, they highlight a promising strategy—changing the behavior of the tumor’s own neighborhood to slow, or even reverse, the course of lung cancer.
Citation: Jiang, P., Chen, Z., Cui, L. et al. ID1 in TAMs promoted the progression of non-small-cell carcinoma via increasing NF-κB/NPM1/SHP1/SHP2 signaling induced M2 polarization. Sci Rep 16, 14222 (2026). https://doi.org/10.1038/s41598-026-44075-6
Keywords: non-small-cell lung cancer, tumor-associated macrophages, immune microenvironment, macrophage polarization, cancer immunotherapy