Clear Sky Science · en
Development and validation of a gra1–bag1 RT-qPCR assay as an alternative to the mouse bioassay for assessing Toxoplasma gondii viability
Why this tiny parasite matters to your dinner table
Toxoplasma gondii is a microscopic parasite that quietly cycles through farm animals, pets, and people worldwide. Most infections go unnoticed, but in pregnant women and people with weak immune systems it can cause miscarriage, brain damage in newborns, and serious eye disease. Because humans are often infected by eating undercooked meat, food-safety scientists need reliable ways to tell whether the parasite in meat is still alive and dangerous. Until now, the gold-standard test has involved infecting live mice and waiting weeks for signs of disease—an approach that is slow, expensive, and increasingly at odds with modern animal-welfare standards. This study presents a new laboratory test that aims to predict parasite viability without relying so heavily on animal experiments.
From farm animals to food on the plate
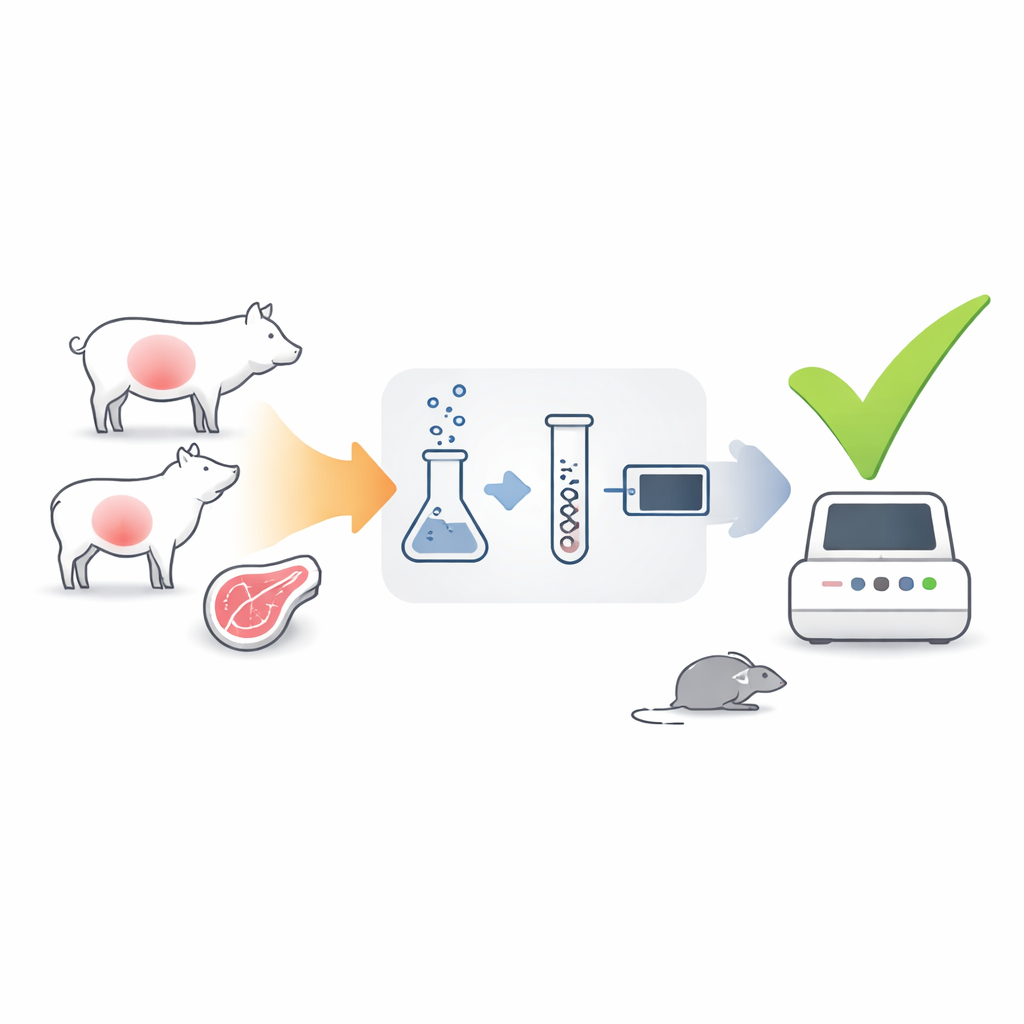
Pigs, sheep, and goats are key links in the chain that brings T. gondii from the environment to human kitchens. The parasite forms hardy cysts in their muscles and brains, which can remain infectious if meat is eaten raw or undercooked. Traditional DNA-based tests are good at spotting whether parasite genetic material is present in a sample, but they cannot tell whether that parasite is alive or dead—much like finding a wrecked car does not prove it can still drive. For that reason, regulators and researchers have continued to depend on the mouse bioassay, in which digested tissue from livestock is injected into mice and the animals are monitored for signs of infection over more than a month.
A new way to listen for signs of life
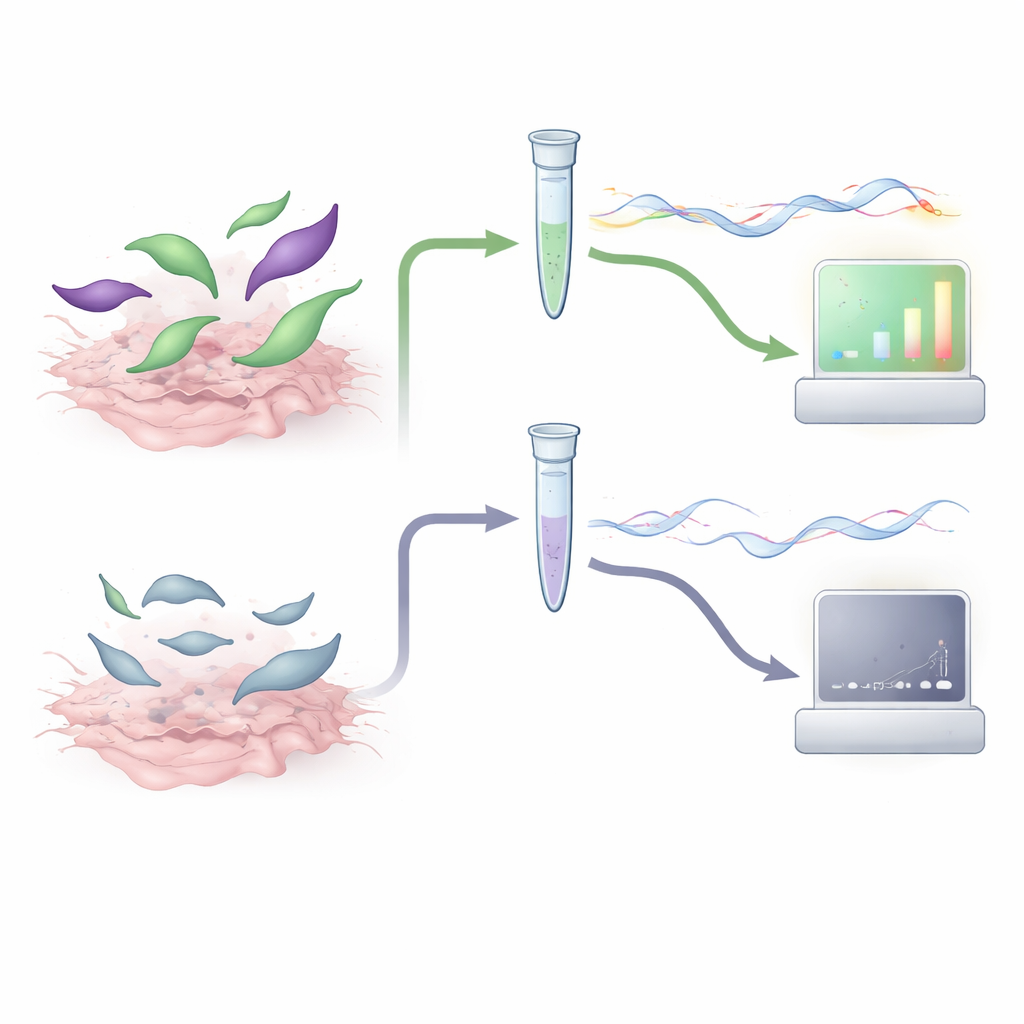
The authors developed a test that focuses not on the parasite’s DNA, but on its messenger RNA (mRNA)—short-lived molecules that are produced only by active, living cells. They chose two parasite genes as signals: gra1, which is strongly active when the parasite is rapidly multiplying, and bag1, which is switched on when it hunkers down in long-lived tissue cysts. Using a technique called RT-qPCR, they converted parasite mRNA from tissue samples into DNA and then amplified it, allowing them to count how many copies of gra1 and bag1 were present. Because mRNA decays quickly once a parasite dies, strong signals from these genes should indicate that infectious parasites are still there.
Putting the test through its paces in the lab
To see whether this idea worked, the team first grew T. gondii in cell culture and subjected the parasites to treatments that mimic what happens during meat digestion and processing. Some samples were simply exposed to digestive enzymes, while others were heated to levels that kill the parasite and then treated with enzymes that destroy leftover RNA. When they applied their gra1–bag1 RT-qPCR test, live or enzyme-treated parasites gave strong mRNA signals, whereas heat-killed parasites produced almost none and failed to grow in fresh cell cultures. The test could reliably detect the equivalent of just a handful of parasites, and the measurements were consistent from run to run, suggesting that the assay is both sensitive and technically robust.
How the new test compares to live-mouse experiments
The crucial question was whether mRNA levels in real animal tissues match the actual risk of infection. The researchers used stored muscle and brain samples from piglets and sheep that had been experimentally infected with known numbers of T. gondii oocysts. Each sample was tested in parallel by four methods: the classic mouse bioassay, a widely used DNA-based qPCR, an additional nested PCR, and the new gra1–bag1 RT-qPCR. By comparing results, they showed that tissues with high gra1 and bag1 signals almost always infected all the mice that were exposed, whereas samples with mRNA levels below a certain threshold never caused infection. When they used these thresholds as cutoffs, agreement between the mRNA test and the mouse bioassay was on par with, or better than, traditional DNA-based methods. Importantly, only samples predicted to contain enough viable parasites to matter would need to go on to mouse testing.
What this means for safer food and fewer animal tests
To a non-specialist, the take-home message is that scientists are getting closer to a fast, ethical way to decide whether meat contains live, infectious T. gondii. By “listening” for short-lived mRNA signals from two key parasite genes, the gra1–bag1 RT-qPCR assay can act as a prescreen: it flags tissues that clearly harbor dangerous levels of viable parasites, and it reassures that others are effectively safe from the parasite’s point of view. While more work is needed in naturally infected animals and processed meat products, this approach could eventually reduce the number of mice used in food-safety testing and help regulators and industry better assess which meats pose a real risk of toxoplasmosis to consumers.
Citation: Largo-de la Torre, A., Velasco-Jiménez, N., Ortega-Mora, L.M. et al. Development and validation of a gra1–bag1 RT-qPCR assay as an alternative to the mouse bioassay for assessing Toxoplasma gondii viability. Sci Rep 16, 14370 (2026). https://doi.org/10.1038/s41598-026-43786-0
Keywords: toxoplasmosis, foodborne parasites, PCR testing, meat safety, animal-free assays