Clear Sky Science · en
Lung mTOR activation leads to lung fibrosis or emphysema via senescence of specific lung cells
Why aging lungs matter to all of us
As we grow older, our lungs slowly change, making us more vulnerable to breathing problems such as emphysema and fibrosis. These conditions damage the air sacs and supporting tissue of the lung, leaving people short of breath and easily fatigued. The study behind this article asks a simple but important question: what goes wrong inside specific lung cells that causes such different patterns of damage, and can removing those damaged cells help the lung recover?
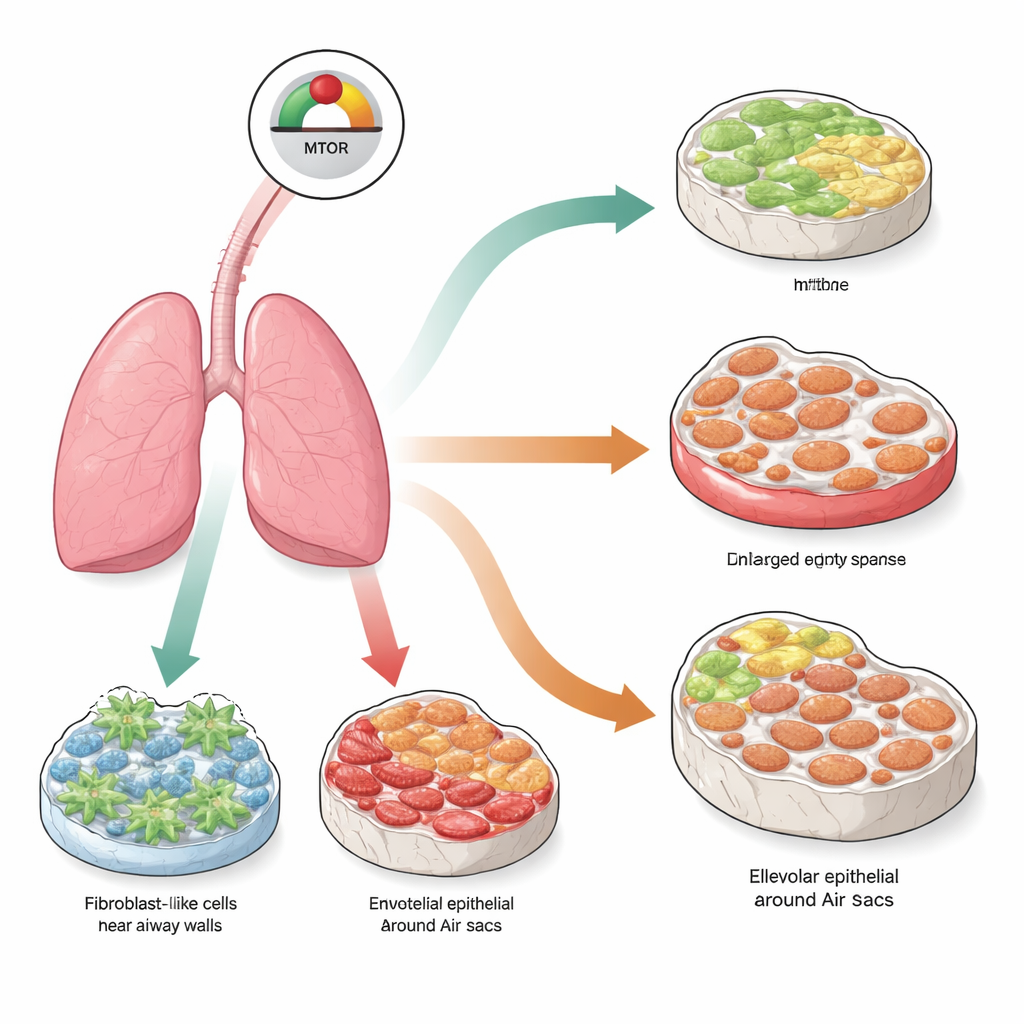
A central growth switch in lung cells
At the heart of this work is a molecular control hub called mTOR, which helps cells sense nutrients and decide whether to grow, divide, or stay quiet. In normal tissue, mTOR activity is carefully balanced. When it is pushed too hard for too long, cells can enter a state called senescence, where they permanently stop dividing and start releasing a mix of inflammatory and tissue-remodeling molecules. The researchers asked whether switching mTOR into permanent overdrive in different lung cell types would push those cells into senescence and, in turn, create different forms of lung disease that resemble what is seen in older people.
Three cell types, three patterns of lung damage
Using genetically engineered mice, the team selectively removed a natural brake on mTOR, called TSC1, in three major lung cell populations: fibroblasts that produce the supporting matrix, endothelial cells that line blood vessels, and alveolar epithelial cells that form the surface of the air sacs. Three months after this targeted change, all three models showed strong signs of senescence and activation of the mTOR pathway. Yet the visible lung damage was strikingly different depending on which cells were affected. When mTOR was overactive in fibroblasts, the lungs developed heavy scarring, or fibrosis, with thick collagen-rich walls. When the same signal was turned on in endothelial cells, the lungs instead developed emphysema, with enlarged, broken air spaces but little scarring. Turning mTOR on in the alveolar surface cells produced a mixed picture: both emphysema-like destruction and fibrotic thickening, mimicking a condition in humans known as combined pulmonary fibrosis and emphysema.
Clearing damaged cells to rescue the lung
The researchers then tested whether getting rid of these senescent cells could reverse the damage. They treated the mice with ABT-263, a drug designed to selectively trigger the death of senescent cells. In all three engineered mouse strains, ABT-263 lowered senescence markers and reduced signals from the mTOR pathway. This cellular cleanup went hand in hand with measurable improvements in lung structure. Scarring decreased in the fibroblast-targeted mice, emphysema lessened in the endothelial-targeted mice, and both scarring and destruction improved, though not completely, in the mice with altered alveolar surface cells. These results suggest that, at least in relatively young adult lungs, senescent cells driven by mTOR overactivity are major players in driving structural damage and that removing them can allow healthier cells to repair the tissue.
What happens in naturally aging lungs
The team also studied normal mice as they aged from one month to 20 months, roughly spanning early life to old age. With advancing age, the lungs gradually accumulated more collagen, developed larger air spaces, and showed higher levels of senescence markers along with stronger mTOR signaling. When older mice were treated with the same senolytic drug, markers of cell aging and mTOR activity declined, and there was a modest reduction in scarring. However, the structural improvements were far less dramatic than in the engineered mice, and emphysema did not clearly improve. This suggests that, in fully aged lungs, damage has become more deeply ingrained and is harder to reverse, perhaps because many different cell types are already affected or because repair systems themselves have worn down.
What this means for future lung health
Taken together, the study shows that turning a single growth-control switch, mTOR, up too high in different lung cells can lead to very different disease patterns: scarring, air-space destruction, or a blend of both. It also shows that these harmful effects are closely linked to the buildup of senescent cells and that clearing such cells can substantially, though not completely, restore lung structure in younger adult animals. In aging lungs, the same strategy improves cellular signals but only slightly reshapes the tissue, hinting that early intervention may be crucial. For people at risk of chronic lung diseases, this work highlights mTOR and cellular senescence as central, interconnected targets and raises the possibility that combination therapies aimed at slowing mTOR and removing senescent cells could one day help preserve lung function into old age.
Citation: Houssaini, A., Marcos, E., Gros, V. et al. Lung mTOR activation leads to lung fibrosis or emphysema via senescence of specific lung cells. Sci Rep 16, 13822 (2026). https://doi.org/10.1038/s41598-026-43628-z
Keywords: lung aging, cellular senescence, mTOR pathway, pulmonary fibrosis, emphysema