Clear Sky Science · en
Peritoneal MSCs-derived exosomes suppress CCL24 synthesis through miR-320d delivery contributing to the improvement of peritoneal dialysis-associated fibrosis
Why protecting the dialysis lining matters
For people whose kidneys have failed, peritoneal dialysis offers the freedom to clean their blood at home using the body’s own abdominal lining as a natural filter. But over years, this delicate membrane can scar and thicken, eventually forcing patients to abandon the treatment. This study explores why that scarring happens and tests a cutting‑edge cell‑based strategy that uses tiny biological parcels to calm damaging immune signals and help keep the membrane healthy.
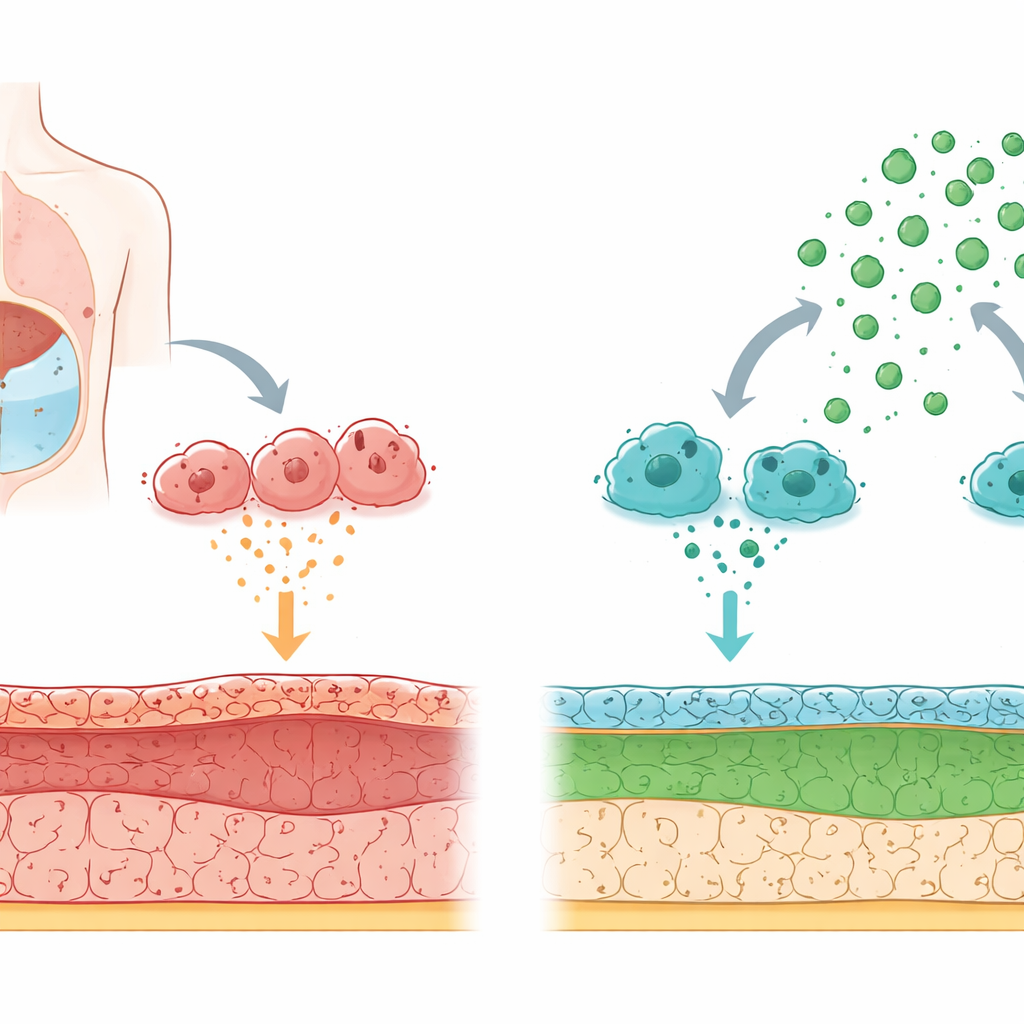
How the body’s filter gets damaged over time
Peritoneal dialysis works by filling the abdomen with a special fluid so wastes can pass from the blood across the peritoneal membrane. Long‑term exposure to high‑glucose dialysis fluid and repeated inflammation slowly injure this lining. The cells that face the fluid can change character: instead of behaving like smooth, protective tiles, they begin to act more like scar‑forming cells that lay down stiff fibers. This shift, called a transition from lining cells to scar‑like cells, drives thickening and loss of function of the membrane.
Immune messengers that push cells toward scarring
The researchers focused on macrophages, immune cells that patrol the peritoneal cavity. In a rat model of dialysis‑induced injury, they found high levels of a signaling protein called CCL24 mainly in these macrophages. When macrophages made more CCL24, nearby lining cells lost their orderly structure and started producing fibrous proteins, a hallmark of scarring. Blocking CCL24 in macrophages, or blocking its surface receiver (known as CCR3) or its downstream chemical switch (a pathway involving the protein p38), prevented the lining cells from turning into scar‑forming cells. This placed macrophage‑derived CCL24 at the center of the scarring process.
Stem cell parcels that quiet harmful signals
The team then turned to mesenchymal stem cells collected from the waste fluid of dialysis patients. These cells naturally release exosomes—nanometer‑sized vesicles that carry molecular messages such as small RNAs. When macrophages were exposed to fluid from these stem cells, or to purified exosomes, their production and release of CCL24 dropped sharply. As a result, the damaging influence of macrophage secretions on the peritoneal lining was reduced, and the lining cells retained more of their protective features. Removing exosomes from the stem cell fluid largely erased this benefit, showing that these tiny parcels are the main therapeutic messengers.
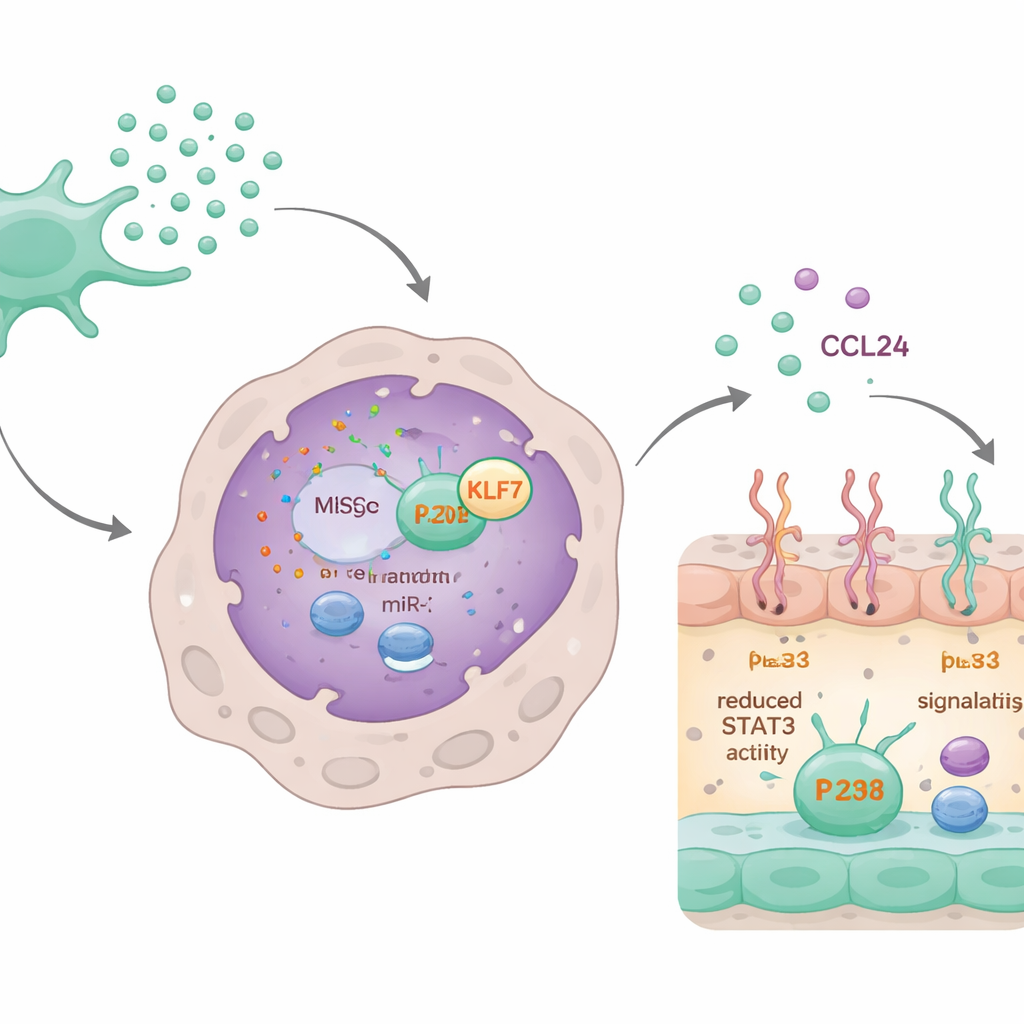
The microRNA circuit that switches off scarring
Digging deeper, the scientists discovered that a specific microRNA, miR‑320d, carried inside the exosomes was key. Macrophages readily took up exosomes loaded with this microRNA. Once inside, miR‑320d bound to and lowered the levels of a protein called KLF7, which normally helps activate another protein, STAT3. Activated STAT3 binds to the CCL24 gene and boosts its production. By cutting down KLF7, miR‑320d reduced STAT3 activation, which in turn lowered CCL24 synthesis and secretion. When miR‑320d was blocked or KLF7 was artificially increased, the calming effect of the exosomes on CCL24 and on macrophage behavior largely disappeared.
From cell culture to living animals
The researchers engineered stem cells to release exosomes enriched with extra miR‑320d and compared them with unmodified exosomes. The boosted exosomes were taken up more efficiently by macrophages and produced stronger drops in KLF7 levels, STAT3 activity, and CCL24 release. In rats undergoing dialysis‑like treatment, weekly injections of either standard or miR‑320d‑rich exosomes lessened thickening and collagen buildup in the peritoneal membrane. The enhanced exosomes provided the greatest protection, preserving more normal tissue appearance and dampening the key signaling steps that lead from macrophage activation to lining‑cell transformation and scarring.
What this could mean for people on dialysis
This work outlines a chain of events in which macrophages release CCL24, driving the dialysis lining toward a scarred, less functional state. Exosomes from peritoneal stem cells, especially when packed with miR‑320d, break this chain by silencing the KLF7–STAT3 control switch that turns on CCL24. In everyday terms, the study suggests that a patient’s own stem‑cell‑derived exosomes could one day be used as a personalized, cell‑free therapy to slow or prevent damage to the peritoneal membrane, helping people stay on peritoneal dialysis longer with fewer complications.
Citation: Zhao, X., Xi, M., Qi, H. et al. Peritoneal MSCs-derived exosomes suppress CCL24 synthesis through miR-320d delivery contributing to the improvement of peritoneal dialysis-associated fibrosis. Sci Rep 16, 11998 (2026). https://doi.org/10.1038/s41598-026-42489-w
Keywords: peritoneal dialysis, peritoneal fibrosis, macrophages, exosomes, microRNA therapy