Clear Sky Science · en
Comprehensive characterization of VSIR reveals dual epigenetic regulation and immune landscape across hematological malignancies
Why this matters for patients with blood cancer
Doctors have powerful new immune therapies for many solid tumors, but similar breakthroughs have lagged behind in blood cancers like leukemia and lymphoma. This study focuses on a lesser-known brake on the immune system called VISTA, controlled by a gene named VSIR. By mapping where and how VSIR is switched on in different blood cancers, and how this relates to patient outcomes and treatment response, the researchers point to new ways to match patients to immunotherapy and design smarter drug combinations.
Where this immune brake shows up in the body
The team first asked a basic question: in healthy people, which cells normally carry this immune brake? By analyzing large public datasets of gene activity from thousands of samples, they found that VSIR is most active in white blood cells of the myeloid family, such as neutrophils and monocytes, and much less active in many other tissues. When they looked across dozens of cancer types, a striking pattern emerged: among all cancers, acute myeloid leukemia (AML) showed the strongest increase in VSIR, with other myeloid blood cancers also high, while most solid tumors had lower or even reduced levels. This set AML apart as a disease where the VISTA brake might be especially important.
How cancer cells rewire the VSIR gene
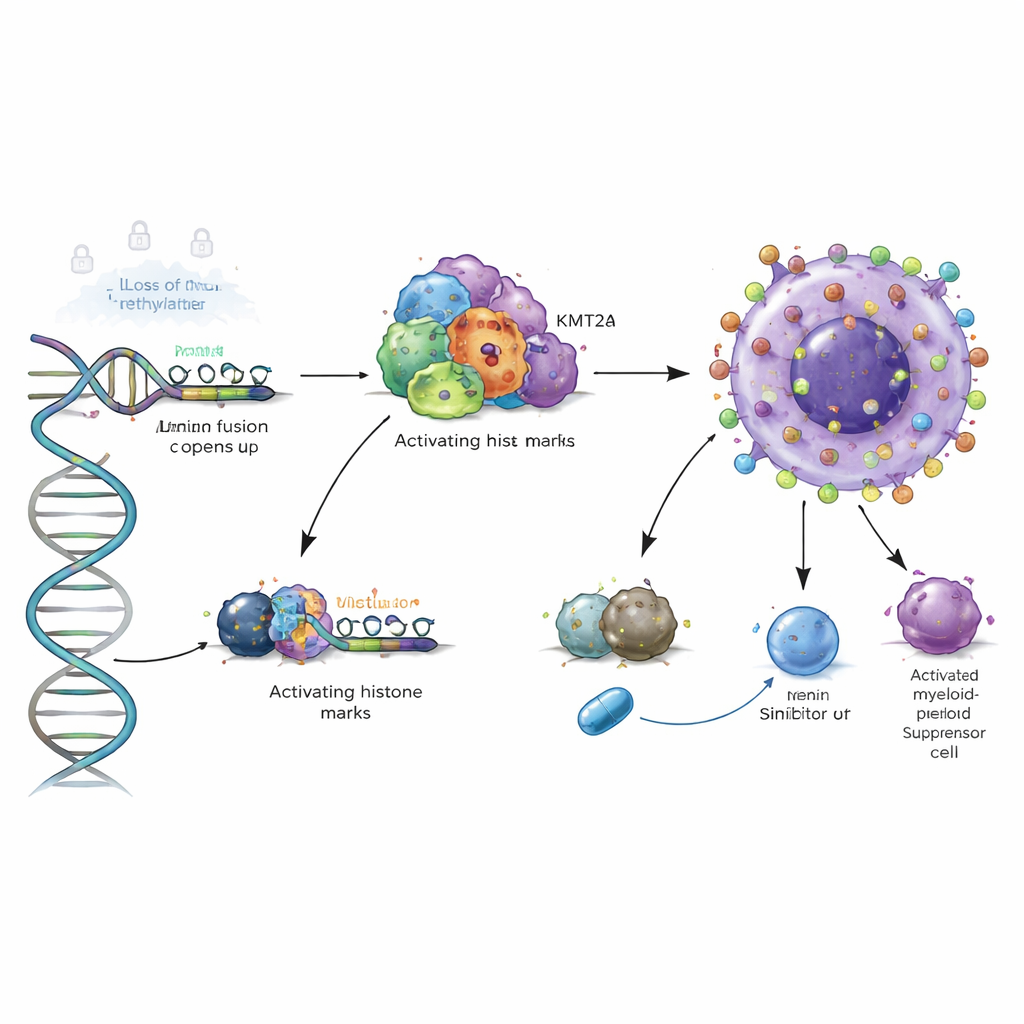
Next, the researchers wanted to know what actually drives this abnormal increase. They found that classic DNA mutations in VSIR are rare and cannot explain the change. Instead, the main culprits are epigenetic changes—chemical tags and protein complexes that control how tightly DNA is packed and whether genes are accessible. In AML and related blood disorders, key control regions near VSIR lose methyl groups, a type of chemical mark that usually keeps genes silent. This loss of methylation was confirmed in several patient cohorts and in a dedicated set of 168 clinical samples, showing a stepwise drop from healthy bone marrow through myelodysplastic syndromes to full-blown AML. As methylation fell, VSIR activity rose, tying this epigenetic shift directly to the gene’s overactivation.
A powerful chromatin switch in high‑risk leukemia
The study went a layer deeper by examining the 3D protein scaffolds that sit on DNA. In high‑risk AML types that carry KMT2A gene fusions or NPM1 mutations, the authors showed that fusion proteins and their co‑factor Menin bind directly at the VSIR control region, bringing in “activating” histone marks that open the local chromatin. Blocking Menin with experimental drugs in cell models reduced these marks and lowered VSIR levels, and degrading mutant NPM1 had a similar effect. This means VSIR is not just incidentally active in these dangerous leukemias; it is directly wired into an oncogenic chromatin program that can potentially be switched off with targeted inhibitors.
What VSIR means for the tumor’s immune neighborhood
Because VISTA acts on immune cells, the authors studied how VSIR levels track with the tumor microenvironment. Using both bulk and single‑cell data from many blood cancers, they found that VSIR is concentrated on monocytes and macrophages, and also present on some killer T cells and natural killer cells. VSIR‑high tumors tended to show strong signatures of immune activity and inflammation, but the balance between helpful and harmful responses differed by disease. In AML, VSIR was linked to cells and pathways that suppress T‑cell attack, consistent with an immune‑evasive leukemia niche. In contrast, in some lymphoid cancers, high VSIR appeared alongside features of more effective immune surveillance.
Predicting who benefits from immunotherapy
The team then asked whether VSIR could signal how well a patient might respond to immune checkpoint drugs. Using a computational tool that predicts response to immunotherapy and real‑world data from patients with melanoma and pancreatic cancer, they found that tumors with higher VSIR expression were more likely to respond and that these patients lived longer. In blood cancers, models predicted that VSIR‑high patients with AML, myelodysplastic syndromes, myeloma, and several leukemias may derive particular benefit from checkpoint blockade, though this still needs direct clinical testing. Interestingly, VSIR’s impact on survival was disease‑specific: in AML, higher VSIR marked poorer outcomes even after accounting for other risk factors, whereas in diffuse large B‑cell lymphoma and myeloma it was associated with better survival.
How these findings could change care
Taken together, the work paints VSIR as a finely tuned immune brake that is hijacked in certain blood cancers through two main routes: loss of DNA methylation and direct activation by Menin‑containing chromatin complexes. In aggressive AML, this helps leukemia cells shield themselves from immune attack and marks patients at higher risk, but at the same time it may flag those whose tumors are primed to respond to immunotherapy. The study suggests that combining Menin inhibitors with drugs that block VISTA could be a promising strategy, especially in KMT2A‑rearranged and NPM1‑mutant AML. More broadly, measuring VSIR could help doctors tailor immune‑based treatments to the specific biology of each blood cancer.
Citation: Xu, Zj., Wu, Xm., Chang, R. et al. Comprehensive characterization of VSIR reveals dual epigenetic regulation and immune landscape across hematological malignancies. Sci Rep 16, 11556 (2026). https://doi.org/10.1038/s41598-026-41978-2
Keywords: VISTA immune checkpoint, acute myeloid leukemia, epigenetic regulation, Menin KMT2A NPM1, cancer immunotherapy