Clear Sky Science · en
Integrative ensemble learning framework for forecasting controlled drug release based on Raman spectral signatures
Why timing matters for pills
Many medicines work best when they dissolve slowly and arrive at just the right spot in the gut, such as the colon, instead of releasing all at once in the stomach. Designing these “long‑acting” and “targeted” pills is tricky and usually requires many trial‑and‑error lab experiments. This study shows how combining light‑based measurements of pill coatings with advanced computer models can forecast how a drug will be released over time, potentially cutting development time, cost, and waste.
From coated pills to smart predictions
The researchers focused on oral tablets coated with plant‑based sugars called polysaccharides, used to protect a drug as it travels through the digestive tract and to delay its release until it reaches the colon. Their test case was a common anti‑inflammatory medicine, 5‑aminosalicylic acid, often used to treat bowel diseases. Instead of relying only on conventional dissolution tests, they used Raman spectroscopy, a technique that shines light on a sample and reads back a molecular “fingerprint” from the scattered light. For each of 155 coated formulations, they captured more than 1,500 spectral features, recorded what type of polysaccharide and liquid medium was used, and measured how much drug had been released after 2, 8, and 24 hours under gut‑like conditions.
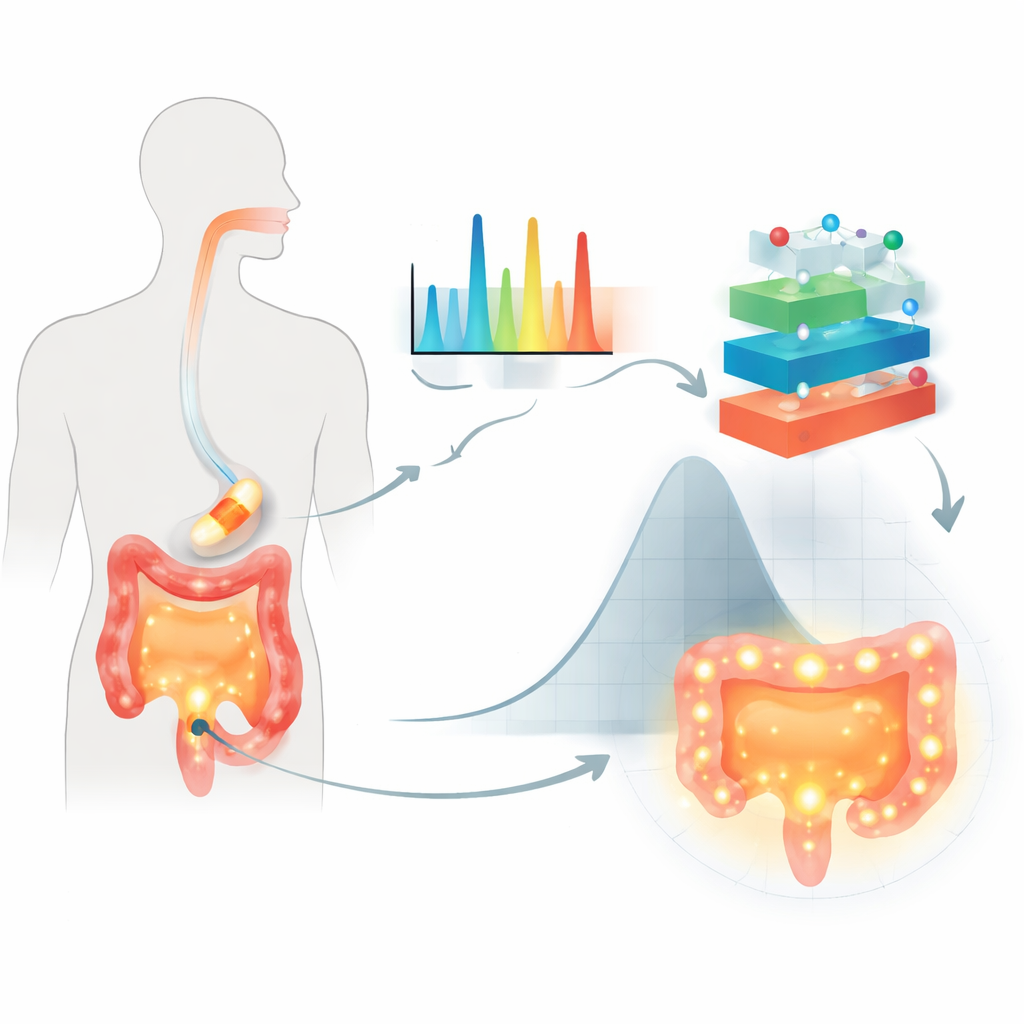
Why old models fall short
Traditional equations for drug release assume simple structures and smooth diffusion, which often do not match the tangled reality of modern coatings and changing gut environments. They also struggle with large, complex datasets, such as thousands of Raman features that encode subtle differences in polymer structure and drug–polymer interaction. Older statistical design methods can handle multiple variables, but become unwieldy when many ingredients and test conditions change at once. The result is that important combinations may be missed, and models may not reliably predict behavior across different fluids that mimic the stomach and intestines.
Blending two minds and two search strategies
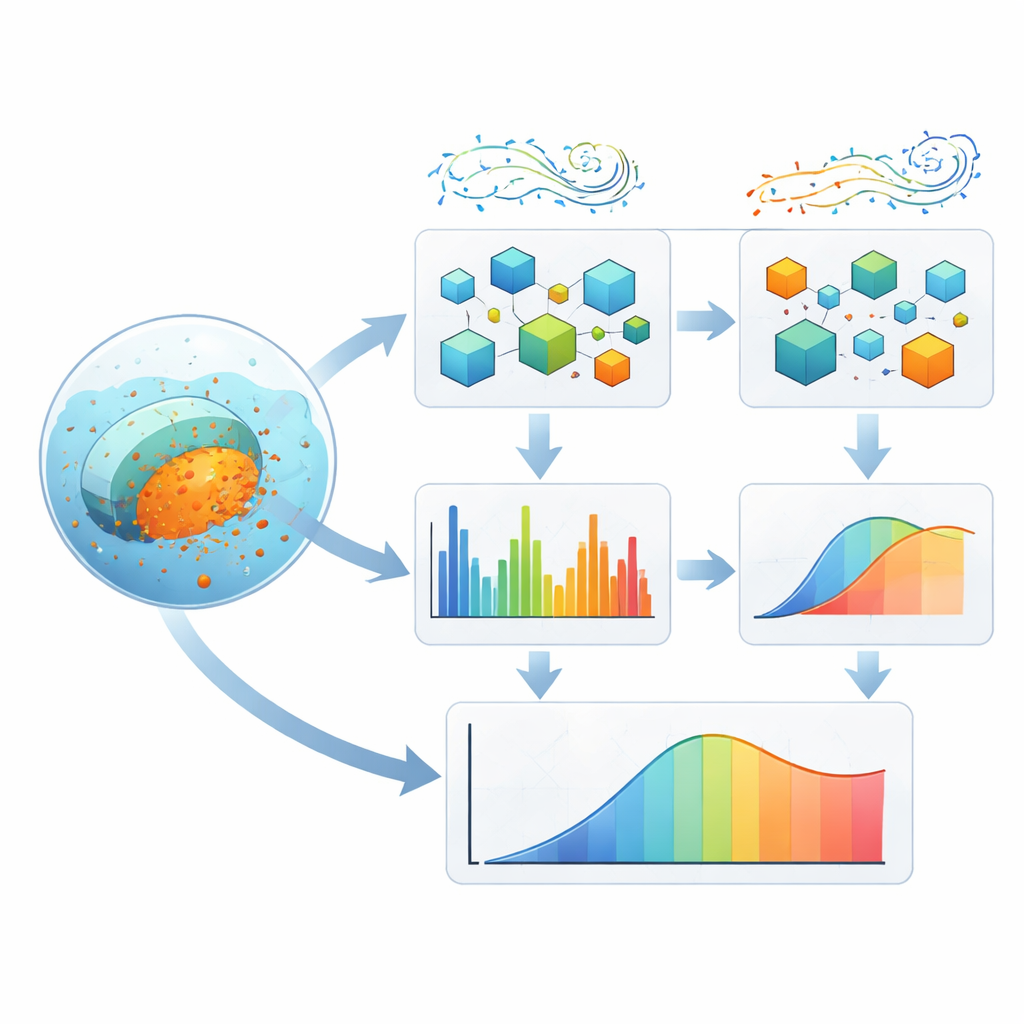
To tackle this complexity, the team built a layered learning system that combines two machine‑learning methods—XGBoost and AdaBoost—inside a specialized ensemble called a Damsphere Weighted Ensemble. Rather than letting one model dominate, the ensemble adjusts how much it trusts each method based on how well it performs, keeping their influences balanced on a kind of mathematical “sphere.” On top of this, two nature‑inspired search procedures, the Puma Optimizer and the Black‑Winged Kite Algorithm, take turns exploring and refining the many knobs and dials (hyperparameters) that control how the models learn. One emphasizes broad exploration of possibilities; the other focuses on fine‑tuning promising solutions. Together, they search for settings that produce accurate, stable predictions without overfitting the data.
Reading the pill’s molecular fingerprint
After careful training and checking using five‑fold cross‑validation and multiple error measures, the framework predicted drug release with very high agreement to experiments (explaining over 99% of the variation in the data). Just as important, the authors probed which inputs mattered most. Simple factors such as time and the type of medium—representing where the pill might be in the body—emerged as the strongest drivers of release, in line with known diffusion and erosion behavior. Certain Raman bands, tied to specific features of the polysaccharide coatings, also stood out, linking molecular structure to how quickly the drug seeped out. Statistical tests confirmed that these patterns were not just random quirks of the dataset but reflected consistent relationships.
What this means for future medicines
To a non‑specialist, the key message is that this approach acts like a smart simulator for coated pills: feed in the pill’s molecular fingerprint and test conditions, and it forecasts how much drug will be released over time. While it does not replace detailed physical models or clinical trials, it can quickly screen candidate coatings, narrow down promising recipes, and reduce the number of lab experiments needed. By cutting material use and speeding development, the framework supports greener, more efficient drug design and offers a clearer window into why some coatings release drugs slowly and reliably while others do not.
Citation: Albariqi, A.H., Safhi, A.Y., Alqahtani, S.S. et al. Integrative ensemble learning framework for forecasting controlled drug release based on Raman spectral signatures. Sci Rep 16, 11343 (2026). https://doi.org/10.1038/s41598-026-41837-0
Keywords: controlled drug release, Raman spectroscopy, machine learning, oral drug delivery, polymer coatings