Clear Sky Science · en
Cancer-Associated fibroblasts regulate the development of cholangiocarcinoma through IL-6/STAT3/AKR1C3 signaling axis
Why the Tumor’s Neighborhood Matters
Cholangiocarcinoma, a cancer of the bile ducts in and around the liver, is notoriously hard to detect early and difficult to treat once found. This study asks a deceptively simple question with big implications: how does the tissue surrounding these tumors help them grow, resist chemotherapy, and rewire their energy use? By uncovering a key communication route between support cells and cancer cells, the researchers point to a new weak spot that future therapies could target.
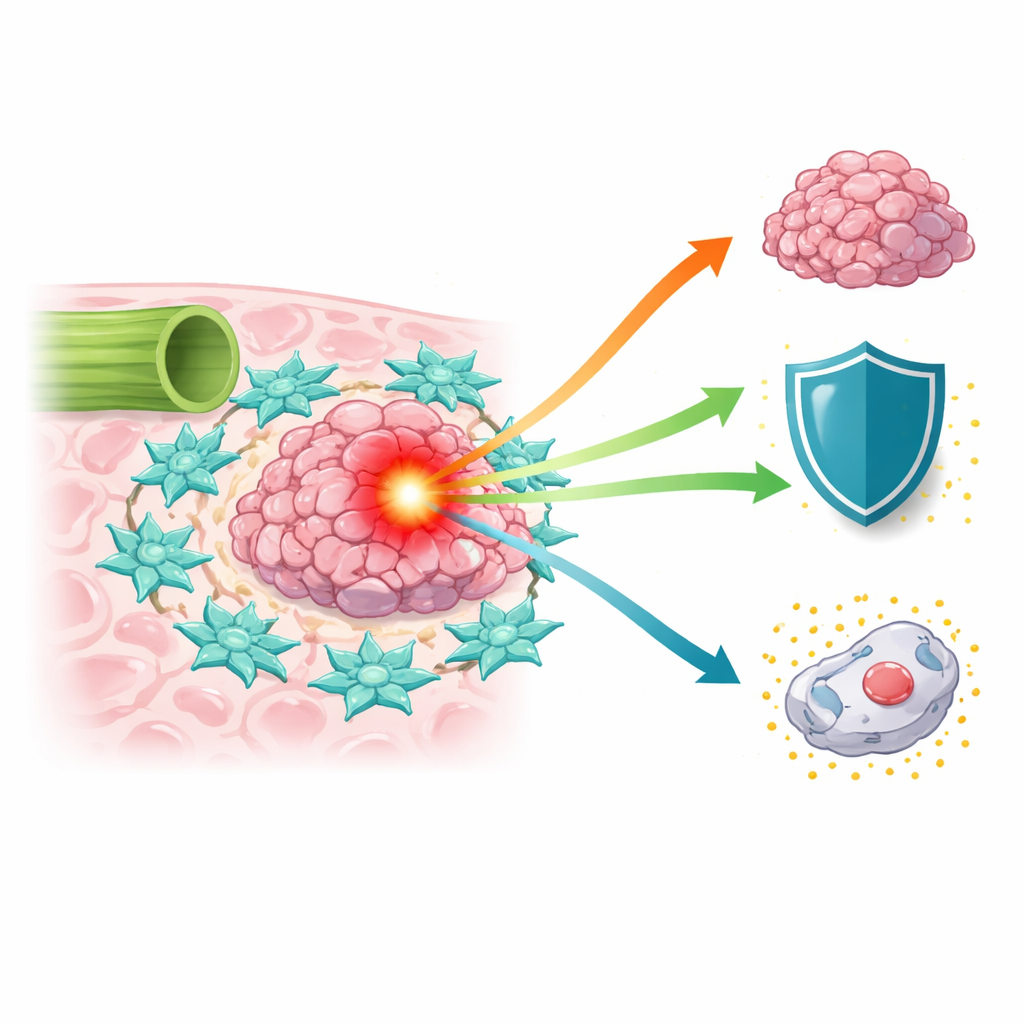
A Hard-to-Treat Cancer on the Rise
Bile duct cancer has been increasing worldwide over the past two decades, yet patients often face poor survival because tumors are discovered late and complete surgical removal is challenging. Standard chemotherapy drugs, such as 5-fluorouracil and platinum-based agents, offer limited benefit. Scientists have realized that the tumor is not just a clump of malignant cells; it sits in a complex “microenvironment” of blood vessels, immune cells, and connective tissue cells that can either restrain or support cancer. Among these helpers, cancer-associated fibroblasts—cells that normally build and remodel tissue—emerge as powerful accomplices that can feed tumors and blunt the effects of treatment.
A Troublemaking Enzyme Inside Tumor Cells
The team focused on a metabolic enzyme called AKR1C3, previously linked to several other cancers but poorly studied in bile duct tumors. Analyzing patient samples, they found that AKR1C3 levels were much higher in cholangiocarcinoma tissue than in nearby normal bile ducts. Patients whose tumors strongly expressed this protein had larger cancers, more lymph node spread, more advanced stage disease, and shorter overall survival. In laboratory-grown cancer cells, boosting AKR1C3 made cells divide faster, form more colonies, and withstand chemotherapy better, while dialing it down slowed growth and made the drugs more effective. These results suggest that AKR1C3 is not just a passenger but an active driver of aggressive behavior.
Fueling Cancer’s Appetite for Sugar
Cancer cells often favor a sugar-hungry strategy called aerobic glycolysis, burning glucose quickly even when oxygen is plentiful. This metabolic shortcut supports rapid growth and reshapes the surrounding tissue. When the researchers reduced AKR1C3 in bile duct cancer cells, the cells took up less glucose, released less lactate (a byproduct of glycolysis), and showed lower measures of acid production and energy generation. In other words, AKR1C3 helps re-tune tumor metabolism toward this high-speed, high-demand mode. Without it, the cancer cells became metabolically less aggressive, hinting that blocking AKR1C3 could starve tumors as well as slow their growth.
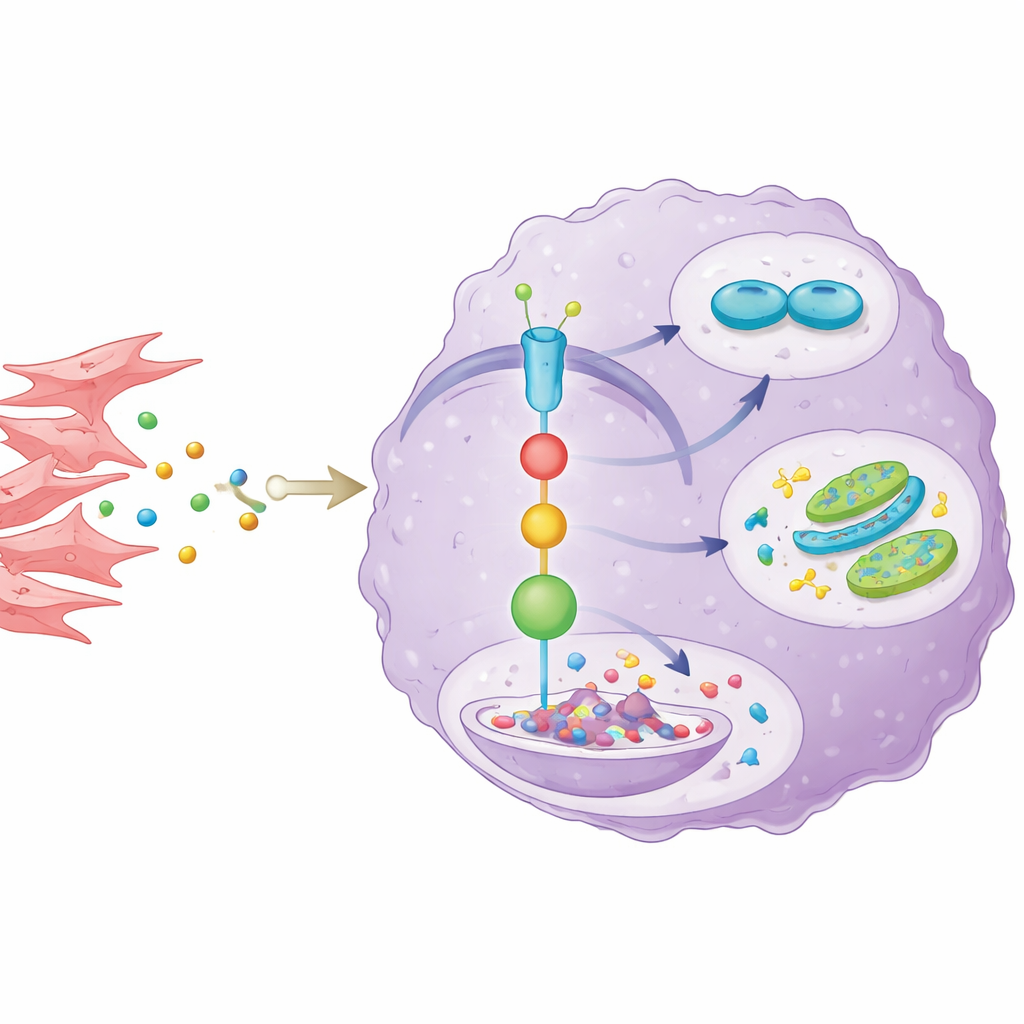
A Signal Relay from Support Cells to Cancer Cells
To understand how the tumor’s neighbors influence AKR1C3, the scientists isolated cancer-associated fibroblasts from patient tumors and grew them with cholangiocarcinoma cells. Fibroblasts secreted the inflammatory messenger IL-6, which activated a well-known communication route inside cancer cells called the STAT3 pathway. This, in turn, raised AKR1C3 levels. When the team blocked IL-6 or a key step in STAT3 signaling, AKR1C3 dropped and the cancer cells lost some of their growth, drug resistance, and sugar-burning advantages. They also uncovered a further twist: AKR1C3 interfered with SOCS1, a natural brake on STAT3, by promoting its breakdown. This formed a self-reinforcing loop, in which fibroblast-derived IL-6 switches on STAT3, STAT3 boosts AKR1C3, and AKR1C3 keeps STAT3 active by weakening its inhibitor.
From Mouse Models to Possible Therapies
The story held up in animal models. When human bile duct cancer cells with reduced AKR1C3 were implanted into mice, tumors grew more slowly. Mixing these cancer cells with fibroblasts normally sped up tumor growth and made them harder to treat with chemotherapy. Yet if AKR1C3 was knocked down in the cancer cells, even the presence of fibroblasts could not fully restore their vigor, and the tumors remained more sensitive to 5-fluorouracil. Tumor samples from these mice confirmed lower AKR1C3, weaker STAT3 activity, fewer signs of cell division, and dampened metabolic markers, reinforcing the central role of this signaling axis in living tissue.
What This Means for Patients
In plain terms, this research shows that certain support cells in bile duct tumors send a chemical message that flips on a protein inside cancer cells, making them grow faster, resist chemotherapy, and burn sugar more intensely. That protein, AKR1C3, also helps keep the message flowing by disabling a built-in safety switch. Because of its strong link to poor outcomes and its central position in this loop, AKR1C3 could become both a marker to identify high-risk patients and a target for new drugs. While clinical applications will require further testing, disrupting this IL-6/STAT3/AKR1C3 pathway offers a promising strategy to weaken bile duct cancers by attacking the crosstalk between tumor cells and their nurturing neighborhood.
Citation: Huang, Tc., Feng, Wd., Fang, Gx. et al. Cancer-Associated fibroblasts regulate the development of cholangiocarcinoma through IL-6/STAT3/AKR1C3 signaling axis. Sci Rep 16, 10467 (2026). https://doi.org/10.1038/s41598-026-37583-y
Keywords: cholangiocarcinoma, cancer-associated fibroblasts, AKR1C3, IL-6 STAT3 signaling, tumor metabolism