Clear Sky Science · en
Single-nucleus RNA sequencing dataset of diverse tissues from wild-type monkey and Tau-P301L transgenic monkey
Why This Monkey Study Matters
Alzheimer’s disease is best known for robbing people of memory, but it also alters movement, balance, and the health of many kinds of brain cells. To design better treatments, scientists need animal models that mimic the human condition as closely as possible. This study introduces a detailed cellular map from a specially engineered monkey that develops Alzheimer-like changes, offering a powerful new window into how different brain and spinal cord cells respond as disease takes hold.
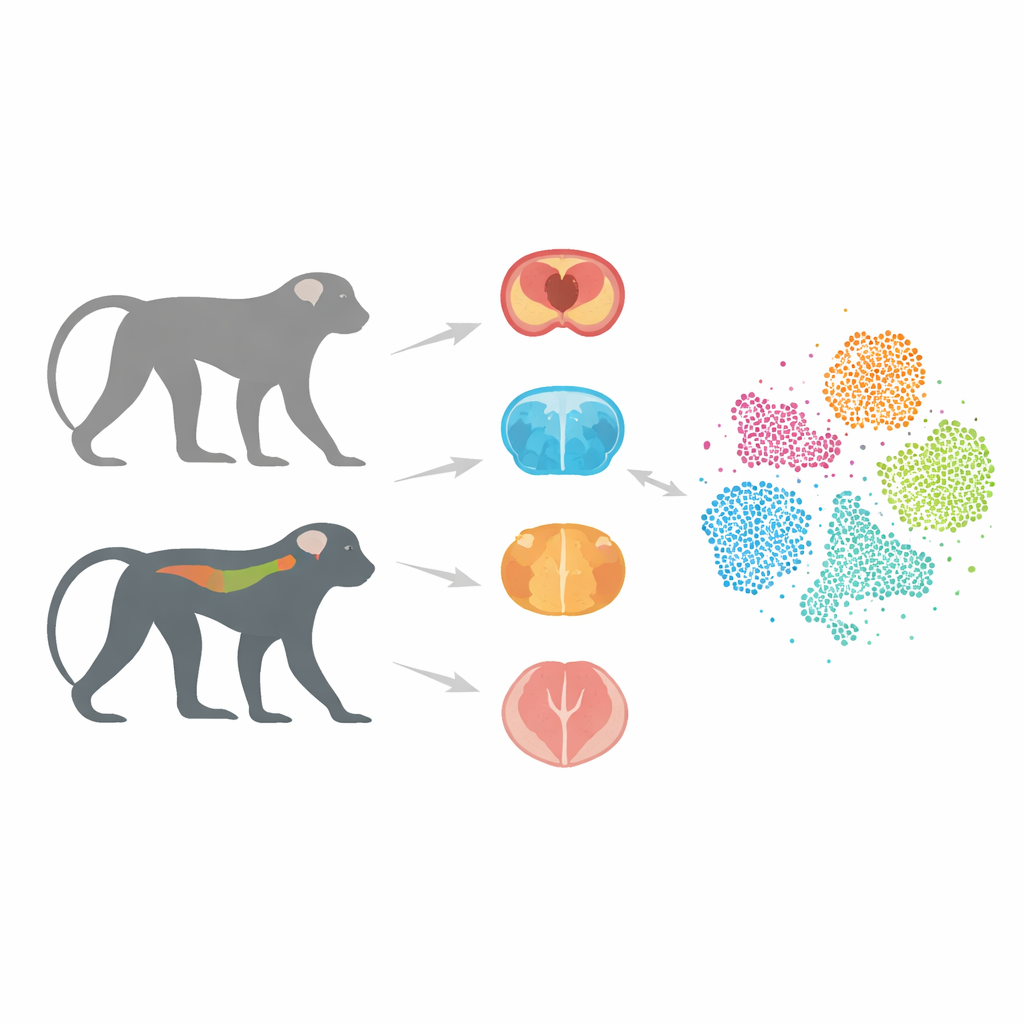
A Closer Animal Stand-In for People
Most Alzheimer’s research has relied on mice that carry human genes linked to the illness. These models have taught researchers a lot, but they often fail to predict whether a potential drug will work in people. One big reason is that mouse brains and immune systems differ markedly from ours. Monkeys, by contrast, share a much more similar brain structure and gene makeup with humans. In earlier work, the authors created cynomolgus monkeys that carry a human version of the tau protein, called Tau-P301L, which clumps abnormally in several dementias. These monkeys gradually develop nerve-cell damage and movement problems, resembling key features of human tau-driven disease.
Reading the Messages Inside Thousands of Cells
In this project, the team focused on three key regions: the hippocampus, important for memory; the striatum, involved in movement and motivation; and the spinal cord, which helps control posture and gait and is increasingly recognized as part of the Alzheimer’s picture. From both transgenic and healthy monkeys, the researchers isolated individual nuclei from cells in these tissues and used single-nucleus RNA sequencing. This technique reads out which genes are active in each cell, allowing scientists to distinguish cell types and states with great precision. After careful quality checks to remove damaged cells and technical errors, they compiled an atlas of 36,623 high-quality cells.
A Cellular Map of the Monkey Nervous System
By grouping cells with similar gene activity patterns, the team identified 13 major cell types, including neurons, several kinds of support cells such as astrocytes and oligodendrocytes, immune-like microglia, and rare populations like T cells and ventricular zone cells. They verified each group using known marker genes and showed that the clusters were clean and consistent. Comparing diseased and healthy monkeys, they found that certain microglia—a type of brain immune cell—expanded in the Tau-P301L animals. These microglia expressed genes previously linked to damage-sensing and inflammatory responses seen around protein deposits in human Alzheimer’s brains, hinting that the monkey model captures an important immune reaction missing or muted in many mouse studies.
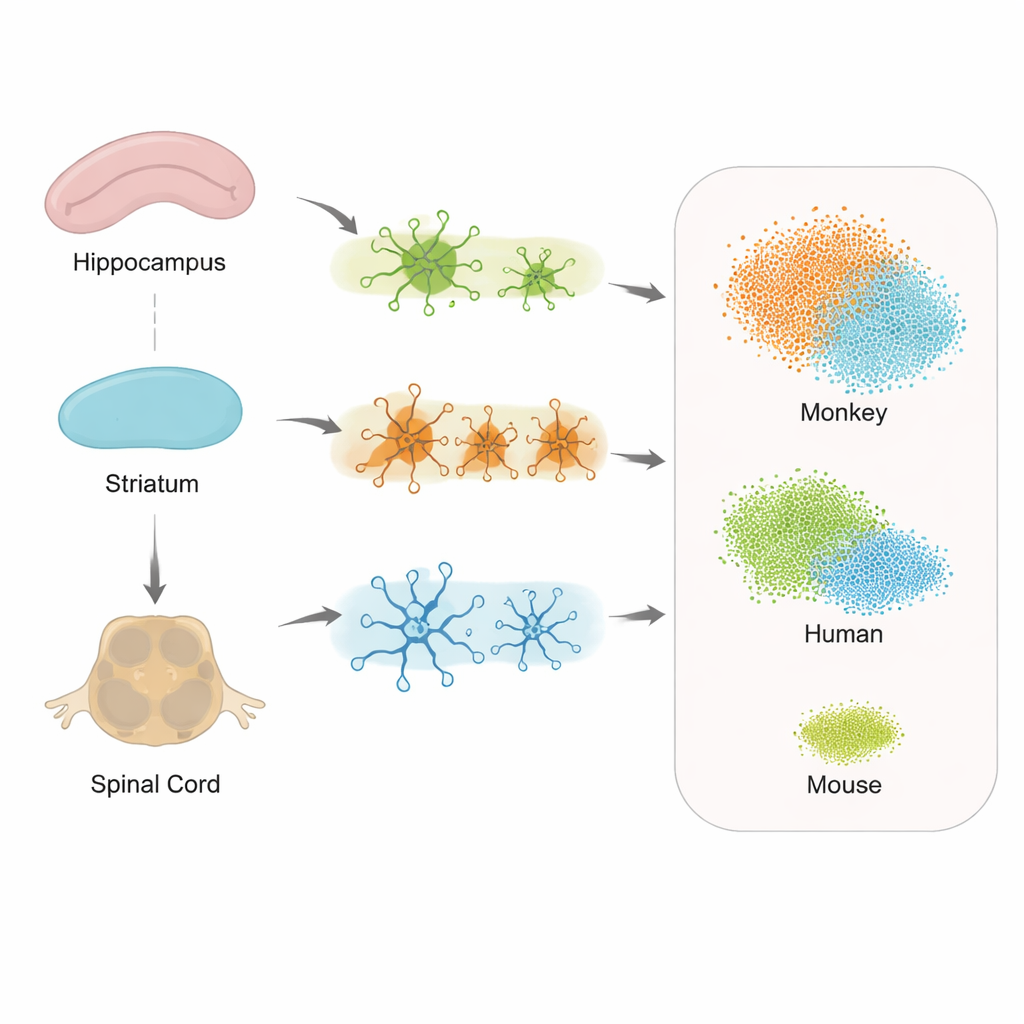
Different Regions, Different Cell Responses
When the researchers examined how genes changed in support cells across the hippocampus, striatum, and spinal cord, they saw both shared and region-specific responses to tau pathology. Astrocytes, microglia, and oligodendrocytes all showed altered activity, but the exact sets of genes varied by location. This suggests that each part of the nervous system mounts its own version of a stress response, which could help explain why some symptoms, like walking difficulties, may appear before obvious memory loss. Importantly, the spinal cord showed strong involvement, supporting the idea that Alzheimer’s is not only a disease of the brain, but of the wider central nervous system.
Monkey Data That Look Strikingly Human
To test how well the monkey model mirrors human disease, the team compared their hippocampal data to existing datasets from people with early-onset Alzheimer’s and from another tau-mutant mouse model. They focused on microglia and on a special “disease-associated” state of these cells that has been described in human patients. The gene activity patterns in monkey microglia overlapped far more with those in human brains than did the mouse patterns, including many key immune and stress-related genes that barely changed in mice. This indicates that the Tau-P301L monkey captures human-like disease signatures at the single-cell level better than current rodent models.
What This Means for Future Alzheimer’s Research
For non-specialists, the main message is that this study delivers a rich, publicly available cell-by-cell map of how a humanized tau protein disrupts brain and spinal cord cells in a species much closer to us than mice. The atlas highlights which cell types and regions are most affected and shows that the monkey model reproduces human immune-cell changes more faithfully than standard mouse models. As a result, these data offer a valuable testing ground for ideas about how Alzheimer’s begins and spreads, and they may help researchers design therapies that stand a better chance of working when they finally reach human patients.
Citation: Han, B., Chen, Y., Ouyang, W. et al. Single-nucleus RNA sequencing dataset of diverse tissues from wild-type monkey and Tau-P301L transgenic monkey. Sci Data 13, 497 (2026). https://doi.org/10.1038/s41597-026-06882-4
Keywords: Alzheimer’s disease, tau protein, non-human primate model, single-cell RNA sequencing, microglia