Clear Sky Science · en
Ligand binding modes of the bitter taste receptor T2R14 and T2R46
Why bitterness matters beyond the tongue
Most of us think of bitter flavors as something to avoid in food or drink, but the same sensors that make coffee or tonic water taste sharp are also scattered throughout the body, where they influence metabolism, inflammation, and even cancer. This study focuses on two of the broadest "bitter detectors," called T2R14 and T2R46, and shows in atomic detail how they recognize very different bitter molecules. By revealing where these compounds land on the receptors and how that triggers a signal inside cells, the work opens the door to designing new medicines that tap into the body’s bitterness circuitry without relying on chance discoveries.
Two versatile sentinels for bitter chemicals
Humans have 25 known bitter taste receptors, but T2R14 and T2R46 stand out because they respond to an especially wide range of plant compounds and drugs. These receptors are not only on the tongue; they are also found in the gut, blood vessels, fat tissue and airways, where they help regulate cholesterol, inflammation, obesity, and tumor growth. Yet drug developers have struggled to target them because their natural ligands tend to bind weakly and with poor selectivity. To tackle this problem, the authors used high-resolution cryo–electron microscopy to determine seven three-dimensional structures of T2R14 and T2R46, either empty or bound to different bitter molecules, all caught in the act of signaling through a G protein inside the cell.
One receptor, two doors for bitter molecules
The first surprise came from T2R14, which turned out to use two separate docking areas for its ligands. Earlier work had suggested an internal site closer to the cell interior, but the new structures show that many of its best-known plant compounds, such as the citrus flavone tangeretin and the antiseptic chlorhexidine, actually settle into a larger pocket open to the outside of the cell. This outer pocket can cradle bulky, often uncharged or positively charged molecules, while a smaller inner pocket favors compact, negatively charged acids. Computer docking of 385 known T2R14 activators showed that about 90% prefer the outer pocket, while only certain small acidic or phenylpropanoid molecules favor the inner one. This dual-entry system helps explain why T2R14 can recognize hundreds of very different bitter substances.
Two receptors, one ligand, very different fits
The study then compared how the same compound, tangeretin, binds to T2R14 and T2R46. Although both receptors sense this citrus molecule, the structures reveal that tangeretin sits upright in T2R14 but lies sideways in T2R46, rotated by about 90 degrees. This dramatic reorientation is caused by subtle differences in how the outermost helices are positioned, especially a segment called helix VII, and by which amino acids line the binding pocket. T2R14 uses a cluster of bulky, greasy residues at the pocket entrance to grip one end of tangeretin, whereas T2R46 relies on a more classic, centrally placed pocket where a single highly conserved tryptophan acts as a hub, stacking against the core of several unrelated ligands, including tangeretin, strychnine, artemisinin, and denatonium. Mutating these key residues sharply reduced receptor activation, confirming that the different arrangements are essential for how each receptor chooses and responds to its ligands.
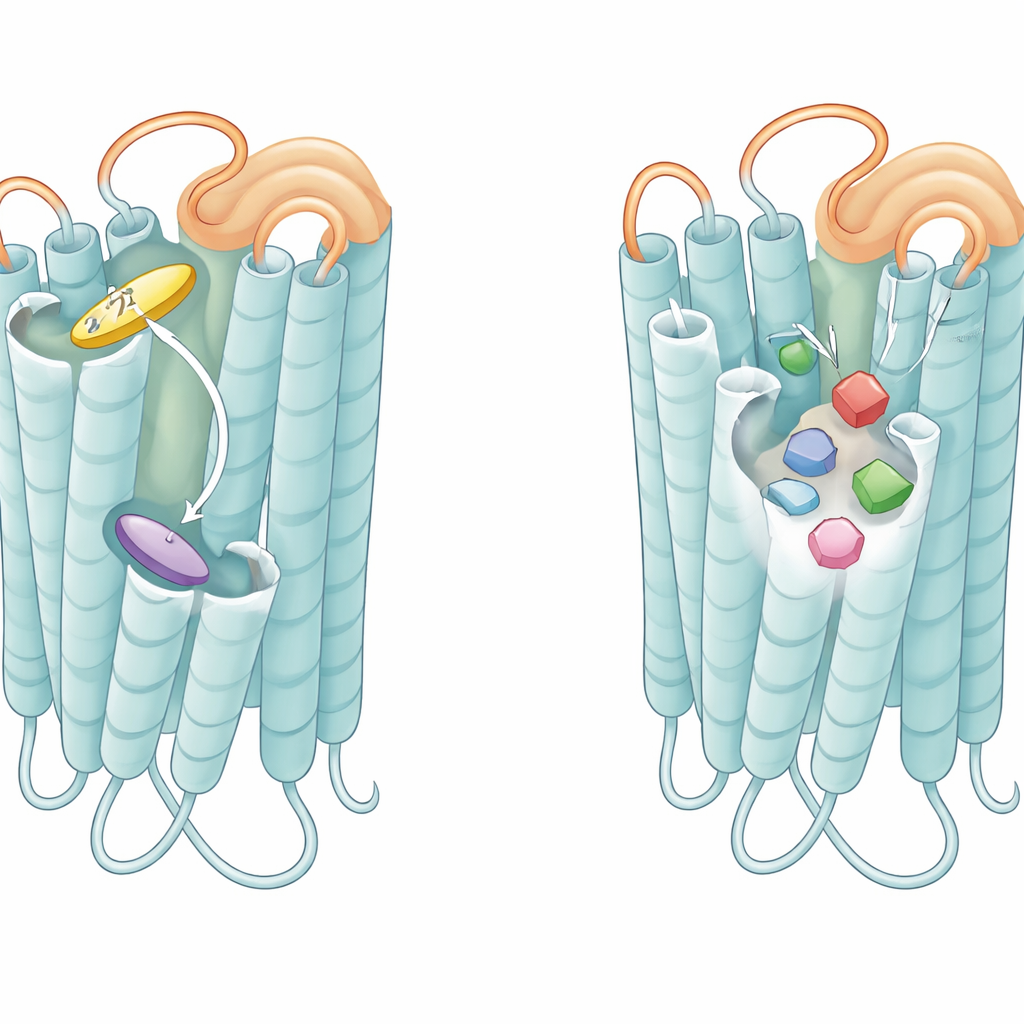
How loops and pockets tune bitter sensitivity
Beyond the pockets themselves, the flexible loop that hangs over the outer face of each receptor, known as the second extracellular loop, emerged as a crucial player. In T2R46, when no tastant is bound, this loop folds down into the pocket and mimics a ligand, stacking against the same tryptophan that real bitter molecules use. Peptides copied from this loop could activate the receptor on their own, and altering loop residues reduced the receptor’s background activity and its response to weak agonists. A similar peptide derived from T2R14’s loop strongly activated T2R14 and, to a lesser extent, T2R46. These findings suggest that the loop behaves like a built-in, tethered mini-agonist that keeps the receptor hovering in a pre-alert state, ready to respond quickly even to low-affinity bitter compounds.
What this means for future medicines
By mapping exactly how different bitter compounds nestle into T2R14 and T2R46, and how a built-in loop helps them switch on despite weak binding, this work provides a blueprint for rational drug design. For T2R14, chemists can aim either at the spacious outer pocket or the tighter inner one to tune selectivity and charge preferences. For T2R46, the conserved central pocket and its interaction with the flexible loop offer a stable framework for designing ligands that act more like precise tools than blunt, broadly bitter drugs. Because these receptors influence metabolism, inflammation and cancer far from the tongue, such targeted molecules could one day harness our bitter-sensing machinery to treat disease—without necessarily changing how dinner tastes.
Citation: Tan, Q., Yu, Y., Han, X. et al. Ligand binding modes of the bitter taste receptor T2R14 and T2R46. Nat Struct Mol Biol 33, 691–700 (2026). https://doi.org/10.1038/s41594-026-01786-8
Keywords: bitter taste receptors, T2R14, T2R46, ligand binding, drug discovery