Clear Sky Science · en
Transcriptomic and phenotypic convergence of neurodevelopmental disorder risk genes in vitro and in vivo
Why different genes can cause similar brain challenges
Many children diagnosed with autism or developmental delay have very different genetic mutations, yet often share similar learning, behavior, or sensory problems. This study asks a deceptively simple question with big implications: when dozens of different risk genes are altered, do they end up disturbing the same core processes in brain cells, and could those shared weak points be used to guide future treatments?
Looking at many genes in brain cells at once
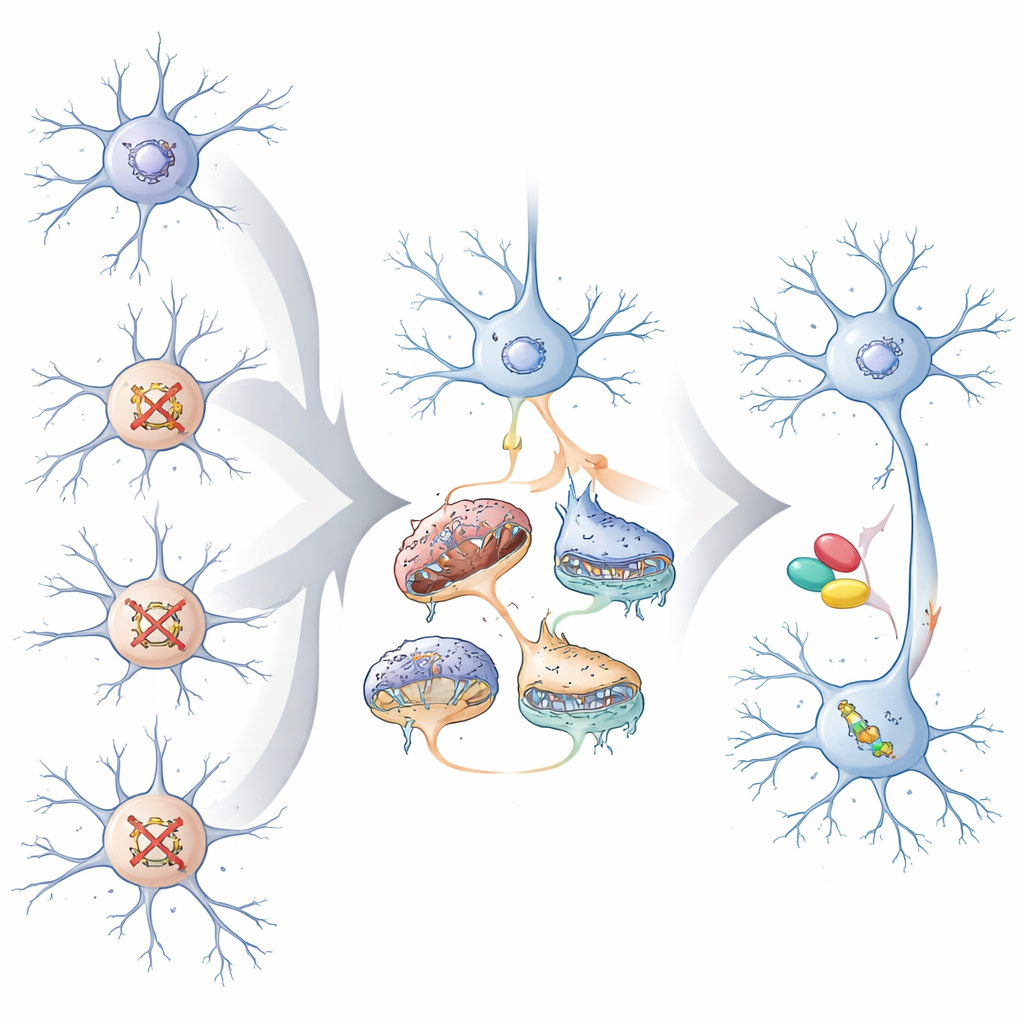
The researchers focused on 23 high-impact genes linked to neurodevelopmental disorders that mostly help control how DNA is packaged and read, plus a few that are vital for communication between nerve cells. Using human stem cells, they grew three key cell types found in the developing cortex: dividing neural progenitor cells, excitatory (glutamatergic) neurons that stimulate brain activity, and inhibitory (GABAergic) neurons that dampen it. With a pooled CRISPR approach, they switched off each risk gene in thousands of single cells, then measured which other genes in those cells turned up or down. This allowed them to see, across many different mutations, where their downstream effects “converged” on the same sets of genes or pathways.
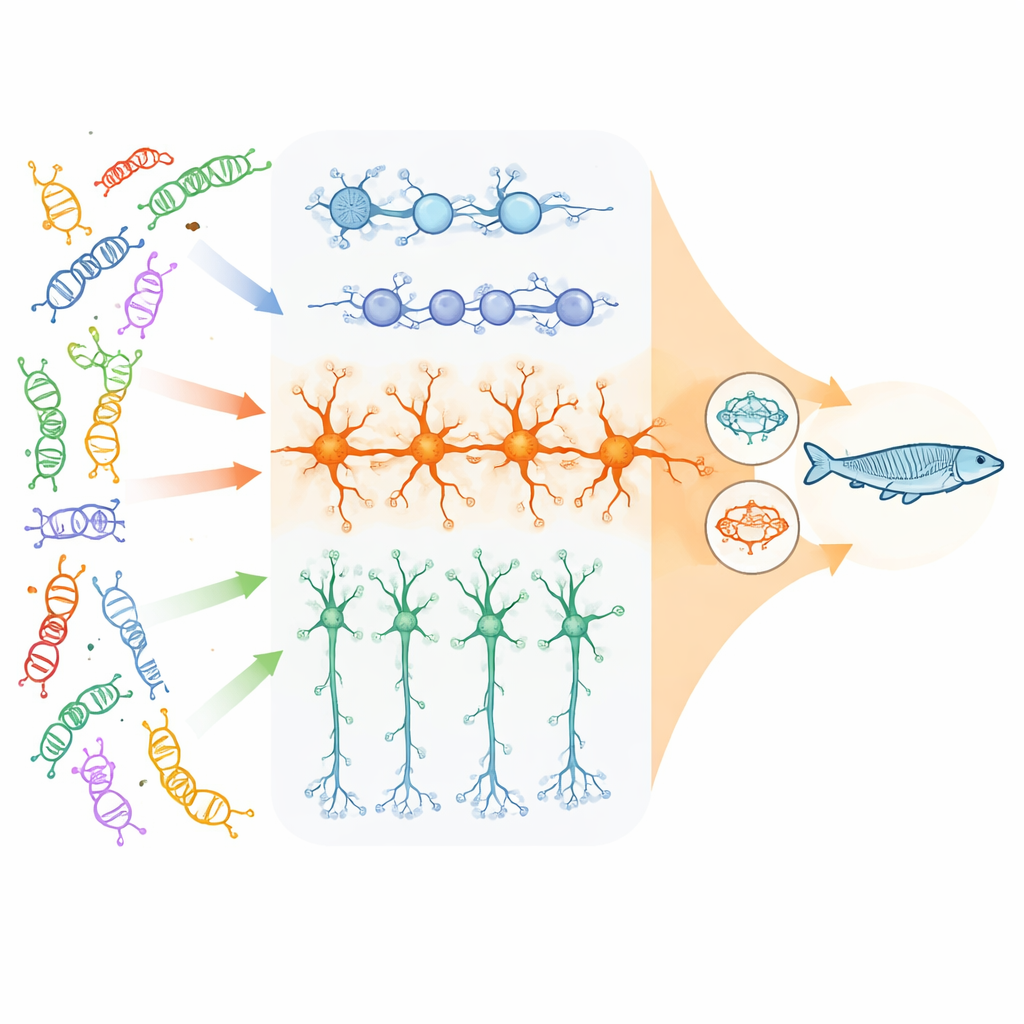
Convergence is strongest in excitatory neurons
The team found that convergence depends strongly on cell type and developmental stage. While each gene knockout left its own fingerprint, the most extensive and tightly connected shared changes appeared in mature excitatory neurons. Here, thousands of genes were altered in similar ways across many different risk genes. The affected networks centered on three themes: synapses (the junctions where neurons communicate), the cellular machinery that controls which genes are turned on or off, and, unexpectedly, mitochondria—the cell’s energy factories. In contrast, neural progenitors mainly showed shared changes in cell division and early brain growth pathways, and inhibitory neurons displayed more modest and distinct convergence patterns.
From gene networks to brain function and behavior
To test whether these converging changes matter for disease, the authors compared the shared gene signatures to large genetic studies of psychiatric conditions. The convergent genes and networks in excitatory neurons were enriched for risk variants linked to autism, schizophrenia, and intellectual disability, as well as known targets of Fragile X protein. Machine-learning models trained on these data predicted that, across more than 100 known neurodevelopmental risk genes, those tied mainly to autism tend to converge in excitatory neurons, whereas those tied more to global developmental delay converge in inhibitory neurons. The team also showed experimentally that several representative risk genes, when disrupted individually, produced distinct but related problems in how neurons generate new cells and in how their mitochondria are structured and consume oxygen—matching the convergent patterns seen in the pooled screens.
Testing convergence in living animals and drugs
Because cell cultures cannot capture whole-brain circuits or behavior, the researchers turned to zebrafish carrying mutations in a subset of the same genes. Automated tracking of sleep, arousal, and startle responses grouped these fish into four behavioral “sets” with similar patterns, such as altered sleep or heightened sensitivity to light changes. When the authors overlaid these behavioral groupings with their human cell data, they found that each set was associated with its own pattern of convergent genes, again strongest in excitatory neurons. They then used drug-response databases and prior zebrafish drug screens to predict medicines that might reverse the convergent signatures and oppose the mutant behaviors. In follow-up tests, 10 of 11 selected drugs improved at least one abnormal behavior in the zebrafish mutants, and some produced striking rescue of sleep or sensory responses.
Why this matters for future treatments
To a non-specialist, the key message is that very different autism- and developmental delay–related genes can push brain cells toward a smaller set of shared problems, especially in the excitatory neurons that drive cortical circuits. These common problems include how synapses function, how gene activity is controlled, and how mitochondria supply energy. By charting these convergent pathways, the study suggests that treatments might one day be matched not just to a person’s specific mutation, but to the shared cellular “fault lines” that mutation activates. The zebrafish experiments hint that targeting these shared pathways, even after the brain has developed, can partly normalize behavior, raising hope that convergent biology could guide more precise and broadly useful therapies.
Citation: Fernandez Garcia, M., Retallick-Townsley, K., Pruitt, A. et al. Transcriptomic and phenotypic convergence of neurodevelopmental disorder risk genes in vitro and in vivo. Nat Neurosci 29, 1079–1094 (2026). https://doi.org/10.1038/s41593-026-02247-7
Keywords: autism genetics, neurodevelopmental disorders, excitatory neurons, mitochondrial function, zebrafish behavior