Clear Sky Science · en
Autoimmune neuroinflammation leads to neuronal death via MIF nuclease-mediated parthanatos
Why inflammation in the brain matters
Multiple sclerosis and related diseases do not only attack the insulation around nerve fibers—they also slowly kill the nerve cells themselves. This hidden loss of neurons drives problems with walking, vision, and thinking that can continue even when relapses are under control. The study summarized here asks a basic but crucial question: during autoimmune inflammation in the brain and spinal cord, exactly how do neurons die, and can that process be blocked without shutting down the immune system entirely?
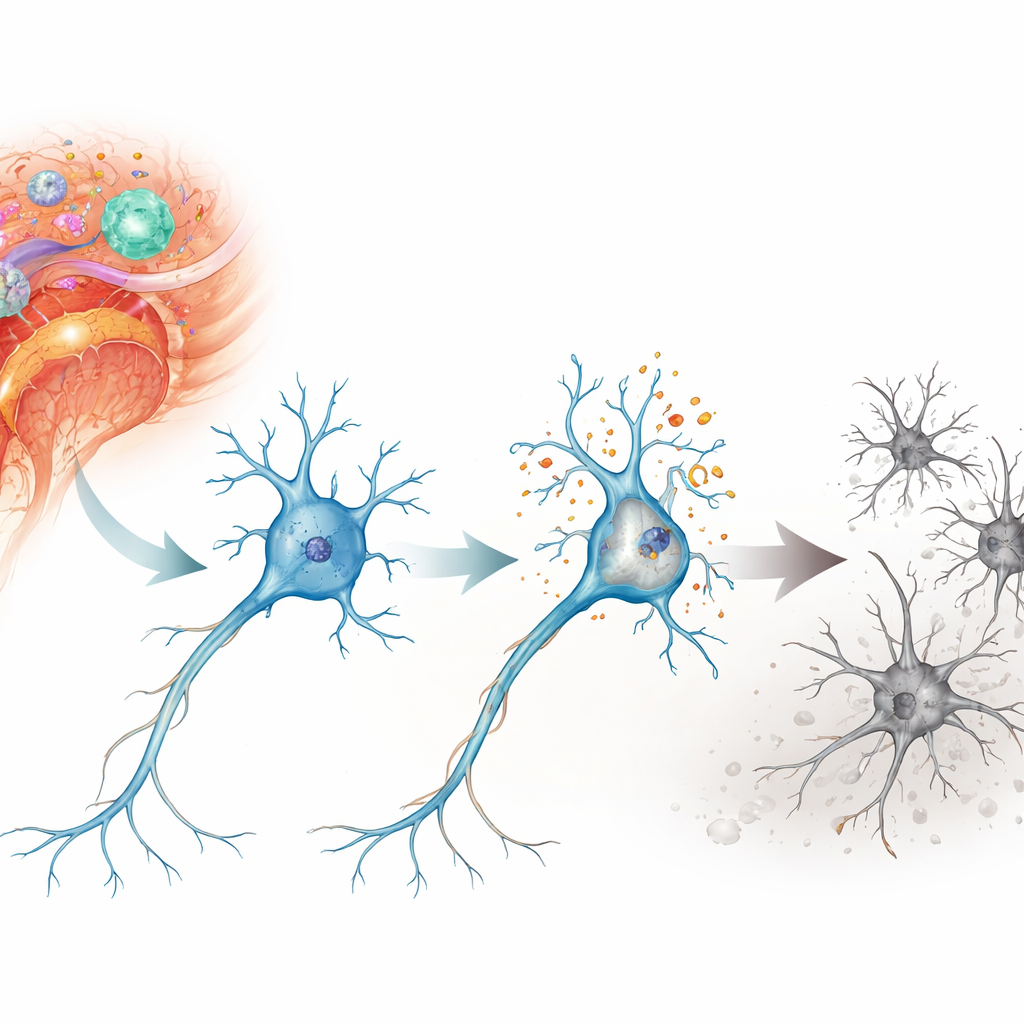
A damaging chain reaction inside neurons
The researchers focused on a form of cell death called parthanatos, a program that is triggered by severe DNA damage rather than by the more familiar suicide pathway of apoptosis. In a mouse model of autoimmune neuroinflammation known as experimental autoimmune encephalomyelitis, they examined neurons in the spinal cord and retina—regions that are injured in multiple sclerosis. They found strong signs of DNA breaks and oxidative stress in these neurons, especially in motor neurons of the spinal cord and in retinal ganglion cells that connect the eye to the brain. Over days to weeks, these cells were progressively lost, matching the time course of worsening neurological disability in the animals.
Following a death script called parthanatos
Looking more closely, the team traced the molecular steps of parthanatos inside affected neurons. When DNA was damaged, an enzyme in the nucleus called PARP1 became overactive and produced long chains of a molecule called PAR. These chains spilled out into the cell body, where they helped trigger the release of a mitochondrial protein (AIF) that partners with another protein, macrophage migration inhibitory factor (MIF). The AIF–MIF complex then moved back into the nucleus. There, MIF acted as a nuclease—an enzyme that cuts DNA—causing large-scale fragmentation of the genome and sealing the neuron’s fate. The authors detected each of these hallmarks, including excess PAR, AIF–MIF binding, and accumulation of MIF in neuronal nuclei, during peak disease in the mice and, to a more limited extent, in human brain tissue from a person with multiple sclerosis.
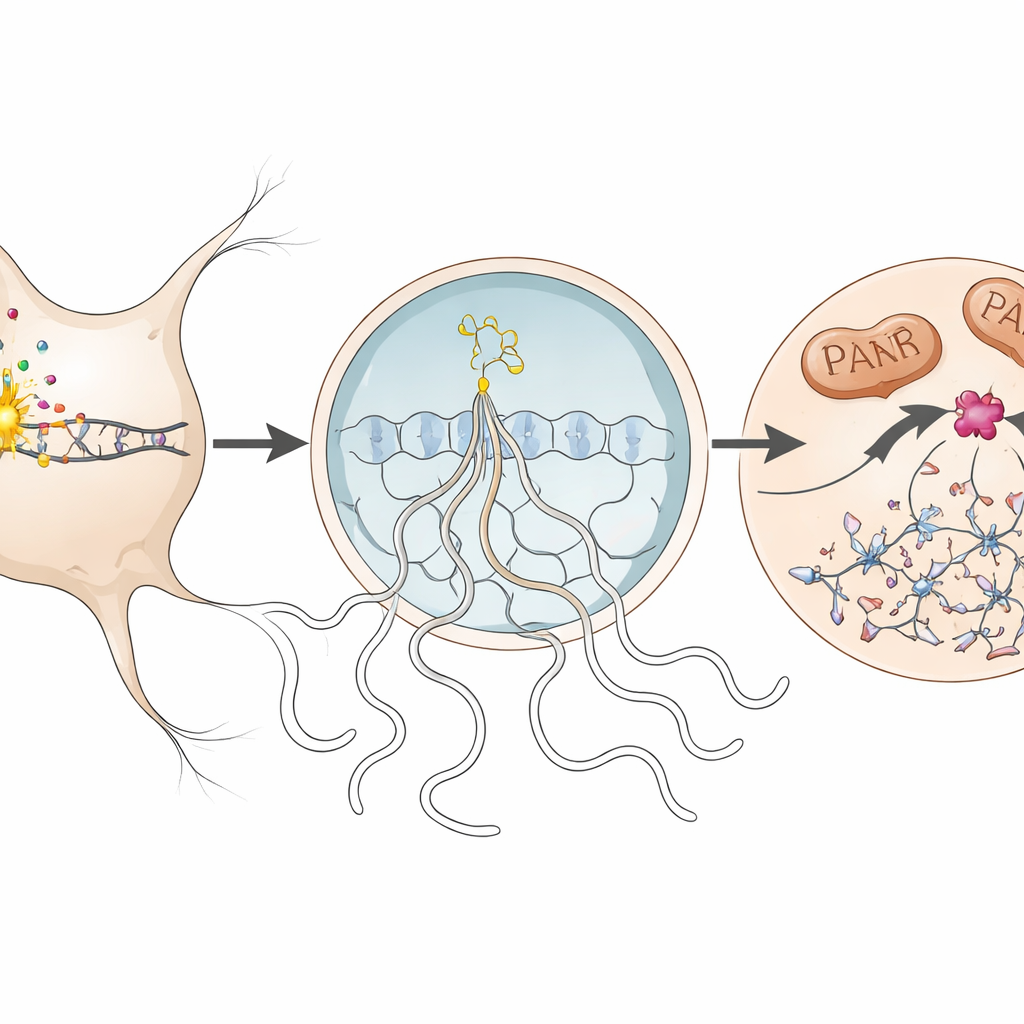
Protecting neurons by disarming a single enzyme
To test whether this pathway actually drives neuron loss, the investigators disabled MIF’s nuclease activity in two complementary ways. First, they used a knock-in mouse that carries a subtle mutation in MIF (E22Q) that leaves its other functions intact but prevents it from cutting DNA. In these animals, the autoimmune attack on myelin and the influx of immune cells into the spinal cord were unchanged, yet neurons in the spinal cord and retina were significantly better preserved, and long-term disability scores were lower than in normal mice. Second, they treated mice with PAANIB-1, a small molecule that selectively blocks MIF’s nuclease activity. Whether given preventively or after symptoms began, this drug reduced neuron loss in the chronic phase of disease without altering immune cell infiltration, glial activation, or the degree of demyelination.
Neurons respond differently when parthanatos is blocked
The team then asked how surviving neurons differ at the level of gene activity. Using single-nucleus RNA sequencing of spinal cord tissue, they profiled tens of thousands of neurons from normal and MIF-mutant mice with and without disease. In standard mice with autoimmune inflammation, neurons turned on many immune-related genes, including pathways that respond to interferon signals and help present antigens, while dialing down genes important for normal electrical signaling, neurotransmitter communication, and production of protective growth factors. In contrast, neurons in MIF-mutant mice maintained much of this core functional gene expression, even though some inflammatory programs were even more strongly activated. A re-analysis of human multiple sclerosis brain data showed a similar long-term suppression of basic neuronal function genes, suggesting that the mouse findings echo changes in people.
What this means for future treatments
Taken together, the results point to parthanatos—specifically the final DNA-cutting step carried out by MIF—as a key route by which inflammation kills neurons in autoimmune disease. Importantly, blocking MIF’s nuclease activity spared neurons without dampening the broader immune response or altering myelin loss, and the protected neurons appeared to retain healthier patterns of gene activity. For a lay reader, the bottom line is that this work identifies a concrete molecular “off switch” for a destructive death program inside neurons. Targeted drugs that flip this switch could, in principle, add true neuroprotection to existing immune-focused therapies for multiple sclerosis and other inflammatory conditions of the brain and spinal cord.
Citation: Mace, J.W., Gadani, S.P., Smith, M.D. et al. Autoimmune neuroinflammation leads to neuronal death via MIF nuclease-mediated parthanatos. Nat Neurosci 29, 796–809 (2026). https://doi.org/10.1038/s41593-026-02201-7
Keywords: multiple sclerosis, neuroinflammation, neuronal cell death, parthanatos, MIF nuclease