Clear Sky Science · en
Immunopeptidome profiling in pulmonary fibrosis provides a platform for identifying therapeutic targets
Turning the Immune System Against Lung Scars
Pulmonary fibrosis, especially idiopathic pulmonary fibrosis, is a deadly lung disease in which normal tissue is slowly replaced by stiff scar. Current drugs can slow this process but rarely stop it, let alone reverse the damage. This study asks a bold question: can we train the body’s own immune system to recognize and selectively remove the cells that drive scarring, much like modern cancer immunotherapies do for tumors?
Why Lung Scarring Is So Hard to Treat
Fibrosis is not a niche problem: roughly one in four people worldwide develop some form of organ scarring, and up to 45% of deaths in industrialized countries are linked to fibrotic disease. In idiopathic pulmonary fibrosis, microscopic wounds in the lung never fully heal. Instead, specialized cells called myofibroblasts and certain types of macrophages lay down excessive connective tissue, turning soft, airy lung into a stiff honeycomb of scar. Approved medicines can dampen inflammation and slow further damage, but they do little to remove the entrenched scar-forming cells already in place. The authors reasoned that if these key culprits display distinctive molecular “flags” on their surface, the immune system might be coaxed to recognize and eliminate them.
Reading the Hidden Flags on Diseased Cells
Every cell constantly breaks down its internal proteins into short fragments and displays a subset of these fragments on its surface using molecules called MHC class I. Together, these fragments form an “immunopeptidome” that T cells scan for signs of danger. The researchers used advanced mass spectrometry to catalog these peptide fragments from scarred lung tissue of patients with idiopathic pulmonary fibrosis and from a standard mouse model of lung fibrosis induced by the drug bleomycin. They found that fibrotic lungs present a much broader and distinct set of peptides than healthy lungs, with many peptide sources highly active in fibroblasts and macrophages involved in tissue remodeling.
Computers Help Pick the Most Promising Targets
Because each person carries a unique mix of MHC molecules and thousands of peptides can be displayed at once, the team built a computational pipeline called Fib-SCORE to prioritize the most promising peptide targets in mice. Fib-SCORE filtered peptides by how confidently they were detected, how strongly they were predicted to bind MHC, how consistently they appeared in fibrotic lungs, and whether their source genes were switched on specifically in the scar-driving cell types. This process distilled thousands of candidates down to a focused shortlist. Three peptides—derived from the proteins MAF, APBB2 and TNS3—stood out for being strongly linked to fibrosis-associated myofibroblasts and macrophages in both mouse and human lungs.
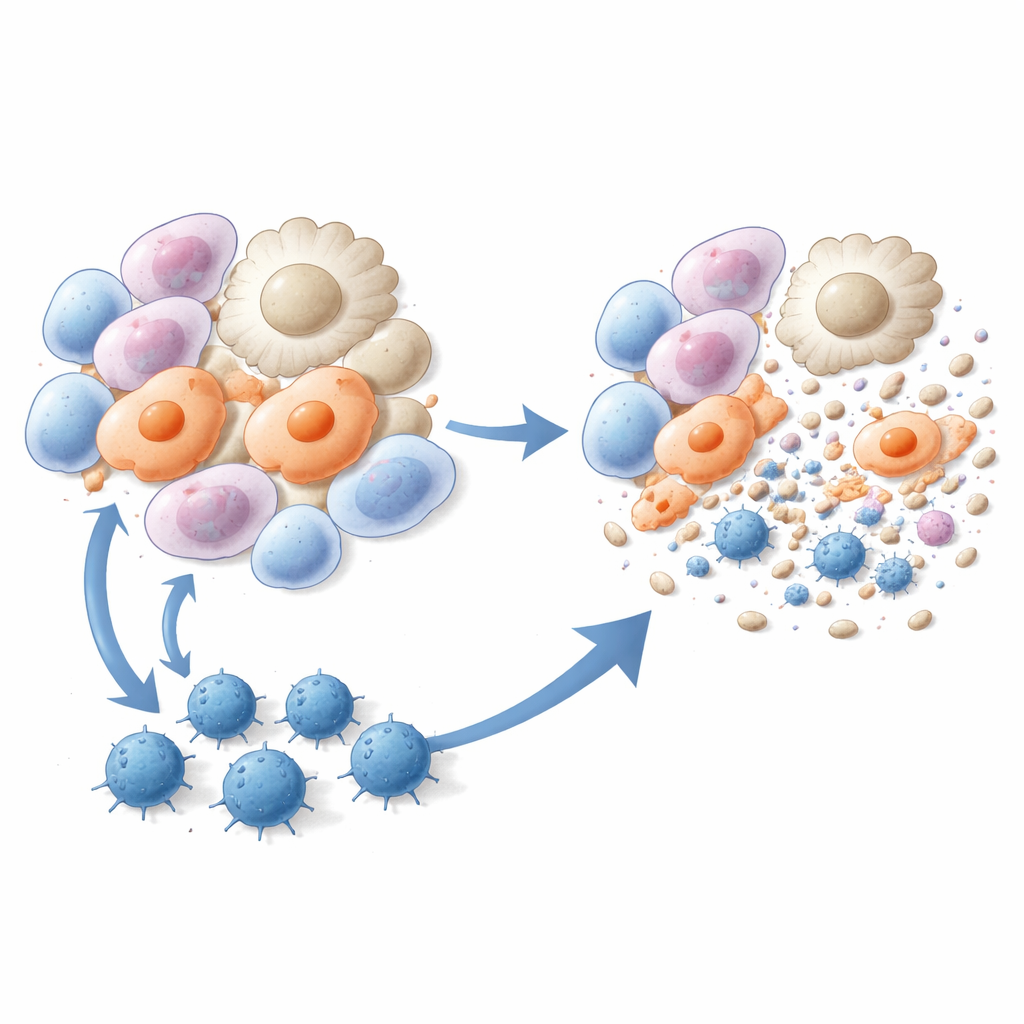
Training Killer T Cells to Clear Fibrotic Cells
The researchers then turned these three peptides into vaccines. Mice were given liposome-based injections containing one of the peptides plus an immune-boosting ingredient, either before or shortly after bleomycin exposure. Compared with untreated animals, vaccinated mice developed fewer fibrotic lesions, had less collagen deposited in the lung, and showed markedly improved lung tissue structure under the microscope. Single-cell RNA sequencing revealed that two particularly harmful cell populations—scar-associated macrophages and alveolar myofibroblasts—were strongly reduced after vaccination. Immune tests confirmed that peptide vaccination generated robust, peptide-specific CD8 T cells that proliferated, produced interferon-gamma, and efficiently killed myofibroblasts and pro-fibrotic macrophages in culture and in living animals.
A Lead Peptide With Human Potential
Among the three candidates, the MAF-derived peptide (called MAF116–124 in the study) showed the strongest overall benefit. In mice, vaccination with this peptide reshaped the lung environment: harmful scar-associated cells shrank in number, memory CD8 T cells accumulated in the lung, and killer T cells were seen in direct contact with myofibroblasts that were undergoing cell death. Because the MAF peptide sequence is highly conserved between mice and humans, the team tested whether it could also activate human T cells. Using blood from healthy donors carrying a compatible HLA type, they generated human cytotoxic T cells specific for the MAF peptide. These human T cells proliferated in response to peptide-presenting cells and selectively destroyed patient-derived myofibroblasts and M2-like macrophages in vitro, mirroring the mouse results.
What This Could Mean for Future Treatments
This work introduces a new concept: using detailed maps of peptide fragments on the surface of fibrotic cells to design vaccines that direct killer T cells against the main drivers of scarring. In mice, such vaccines did not just slow fibrosis—they reduced scar tissue and depleted the cell types that maintain it. While the study has limitations, including small human tissue numbers and reliance on a mouse injury model, it offers a blueprint for transforming fibrotic diseases into conditions that can be targeted by precision immunotherapy. If similar peptide vaccines prove safe and effective in people with idiopathic pulmonary fibrosis, they could eventually complement or even surpass current drugs by harnessing the immune system’s ability to seek out and remove the cells that turn healing into harm.
Citation: Bai, Z., Lan, T., Hong, W. et al. Immunopeptidome profiling in pulmonary fibrosis provides a platform for identifying therapeutic targets. Nat Immunol 27, 923–936 (2026). https://doi.org/10.1038/s41590-026-02501-x
Keywords: pulmonary fibrosis, immunotherapy, peptide vaccine, CD8 T cells, myofibroblasts