Clear Sky Science · en
A prophage-encoded abortive infection protein preserves host and prophage spread
How bacteria use hidden passengers to fight viruses
Bacteria are constantly under attack from viruses called phages, yet many survive thanks to tiny genetic hitchhikers known as prophages—dormant viruses built into their DNA. This study reveals how one such prophage equips Salmonella bacteria with a powerful self-destruct switch that stops invading viruses, all while letting the prophage itself continue to spread. Understanding this molecular tug-of-war helps explain how dangerous pathogens stay one step ahead of their own predators.
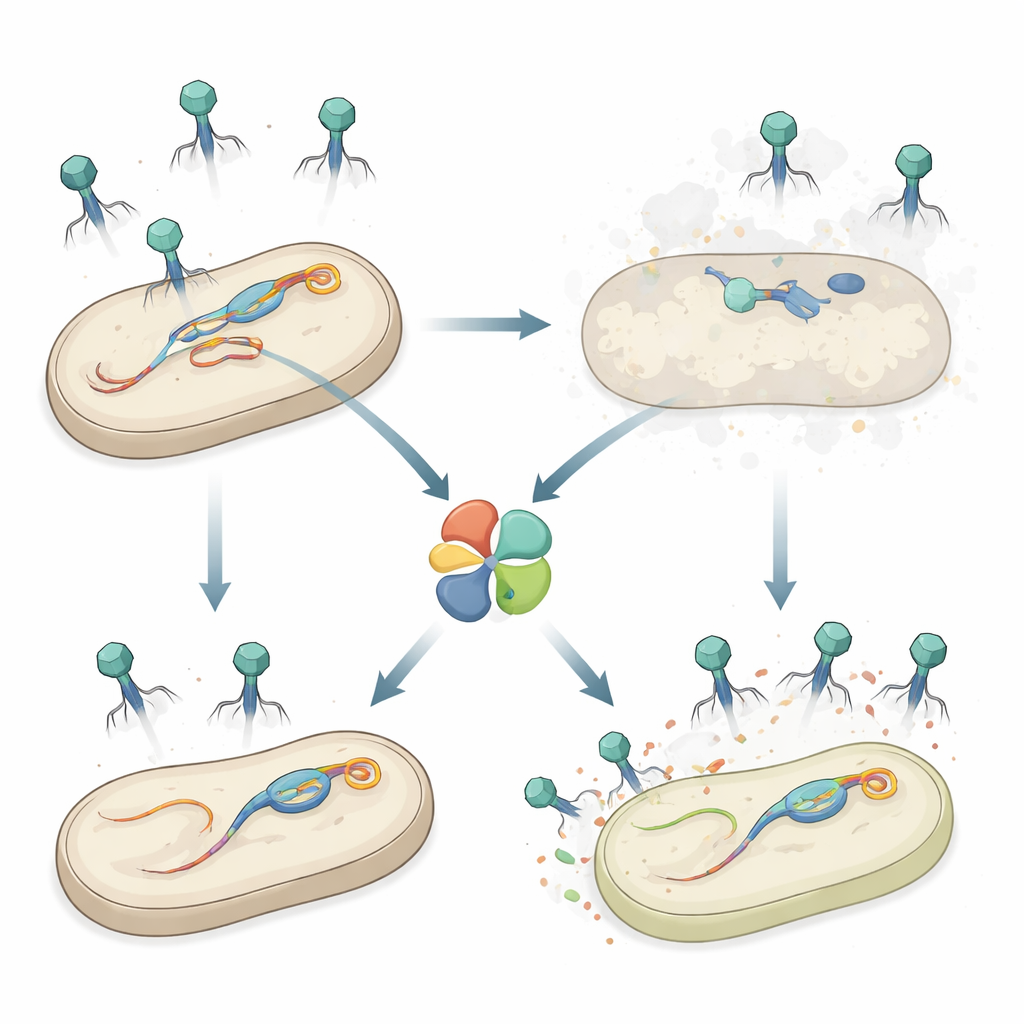
A secret weapon inside Salmonella
The authors studied Salmonella enterica serovar Typhimurium, a food‑borne pathogen that, like many bacteria, carries several prophages in its chromosome. Earlier work showed that one prophage, called Gifsy‑1, helps Salmonella resist attack from certain phages. Here, the team discovered an additional defense gene on Gifsy‑1, named hepS, that makes the bacteria far less vulnerable to a group of long‑tailed phages known as Siphoviridae. When hepS was deleted, invading phages formed many more plaques—clear spots of dead cells—revealing that HepS strongly reduces successful infections, and that it acts independently of other known defenses in the same prophage.
A self-sacrifice system that only wakes on attack
HepS belongs to a family of proteins that often serve as molecular “last stands”: when they sense infection, they shut down essential cell processes so that both the cell and the virus die. The researchers showed that HepS behaves in this way—an abortive infection system—protecting the overall bacterial population by sacrificing individual infected cells. Structural studies revealed that HepS naturally assembles into a four‑part complex (a tetramer) whose active centers are buried and poorly aligned, suggesting an “off” state that is safe for the host. Overproducing HepS in bacteria did not hinder growth, supporting the idea that the protein remains harmless until precisely triggered.
How phage tail tips flip the molecular switch
To learn what activates HepS, the team challenged bacteria carrying hepS with many different phages and found that most susceptible viruses shared similar tail tip proteins, the parts that recognize and pierce the bacterial surface. By selecting rare “escape” phages that could grow despite HepS, they traced resistance to single amino‑acid changes in a short segment of the tail tip protein, called J. Expressing this J fragment inside cells suddenly made HepS toxic, but the escape variant no longer did. Biochemical experiments confirmed that HepS binds this small J peptide directly. High‑resolution crystal structures showed that, once the peptide docks onto each subunit of the HepS tetramer, the whole complex rearranges: the active sites swing open and align, converting HepS into a potent RNA‑cutting enzyme.
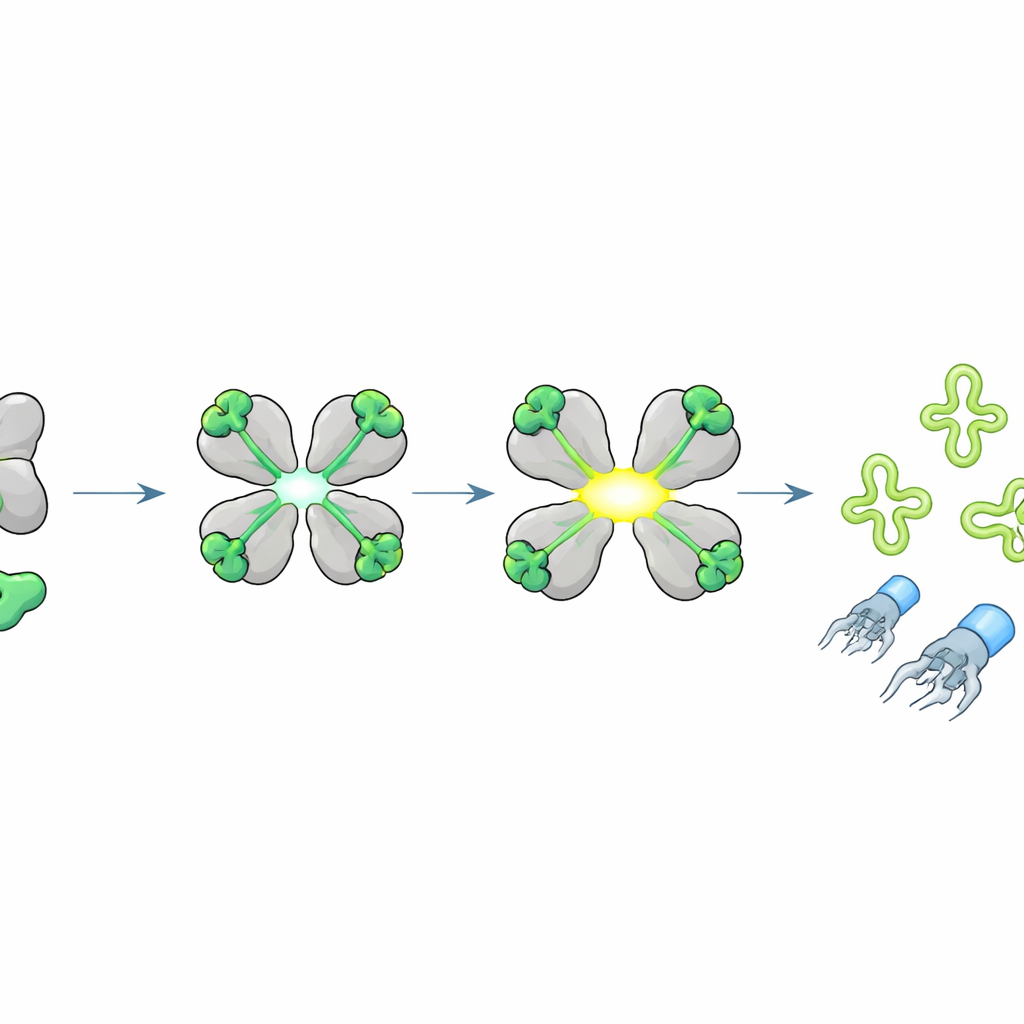
Cutting key tRNAs to stall virus production
What does activated HepS cut? The researchers observed that, when the J peptide turned HepS on, the cell’s RNA broke into characteristic fragments. Using specialized sequencing focused on transfer RNAs (tRNAs)—the adapters that read the genetic code during protein synthesis—they found that HepS slices specific tRNAs exactly at their anticodon loops, especially those for the amino acids threonine, isoleucine, and serine. Structural modeling suggested how an opened HepS tetramer can cradle a target tRNA and position its catalytic residues right at the anticodon. Destroying these tRNAs starves the cell of key building blocks for protein synthesis, abruptly halting production of viral proteins and blocking assembly of complete phage particles, even though viral DNA replication can still occur.
How the resident prophage avoids friendly fire
A central puzzle is why Gifsy‑1, which encodes HepS, is not killed by its own defense. The answer lies again in the tail tip protein. The three resident Gifsy prophages in Salmonella all have J proteins closely related to the HepS‑triggering versions, but at the critical position where activating phages carry a bulky phenylalanine, the Gifsy J proteins carry a milder isoleucine. Experiments showed that wild‑type Gifsy J does not activate HepS, whereas engineered versions that swap in phenylalanine suddenly do, leading to near‑complete loss of Gifsy‑1 particles—but only when hepS is present. In mixed infections, when Gifsy‑1 coexists with an activating prophage like ES18, HepS preferentially cripples ES18 while only modestly affecting Gifsy‑1, giving the HepS‑encoding prophage a competitive edge in producing new particles and spreading to fresh hosts.
What this means for bacteria, viruses, and us
Altogether, this work uncovers a finely tuned survival strategy: a prophage arms its Salmonella host with a booby‑trap protein that senses a hallmark of related invading viruses, then destroys the cell’s own protein‑making machinery by slicing select tRNAs. This sacrificial move halts the invader’s life cycle and protects the broader bacterial community, while subtle changes in the prophage’s own tail tip protein let it slip past its own trap and continue to replicate. The study highlights how viruses embedded in bacterial genomes do more than lie dormant—they actively shape battles against other viruses, influencing which strains of both bacteria and phages thrive in microbial ecosystems.
Citation: Sargen, M.R., Antine, S.P., Grabe, G.J. et al. A prophage-encoded abortive infection protein preserves host and prophage spread. Nature 652, 201–208 (2026). https://doi.org/10.1038/s41586-025-10070-6
Keywords: bacteriophage defense, prophage, abortive infection, tRNA nuclease, Salmonella phage interactions