Clear Sky Science · en
Metronidazole and ether derivatives target Helicobacter pylori via simultaneous stress induction and inhibition
Why stomach germs and old drugs still matter
Helicobacter pylori is a spiral-shaped germ that quietly colonizes the stomachs of almost half the world’s population. Most people never feel a thing, but long-term infection can lead to ulcers and even stomach cancer. Doctors have relied for decades on an older drug called metronidazole to help clear this bug, yet rising resistance means higher doses and multi-drug cocktails are now needed—regimens that can upset the gut’s friendly microbes. This study asks a simple question with big consequences: can we redesign this old workhorse so it hits H. pylori harder, at lower doses, and with less collateral damage?
An upgraded medicine with surprising strength
The researchers started from metronidazole’s core chemical structure and attached small “handles” called ether groups, creating a series of new compounds that behave like close cousins of the original drug. When they tested these variants against H. pylori in the lab, some were up to 60 times more potent than metronidazole itself, stopping bacterial growth at tiny concentrations. Importantly, this boost did not simply come from turning the drug “on” more easily through standard redox chemistry, suggesting that the modified molecules must be doing something qualitatively different inside the bacterial cell.
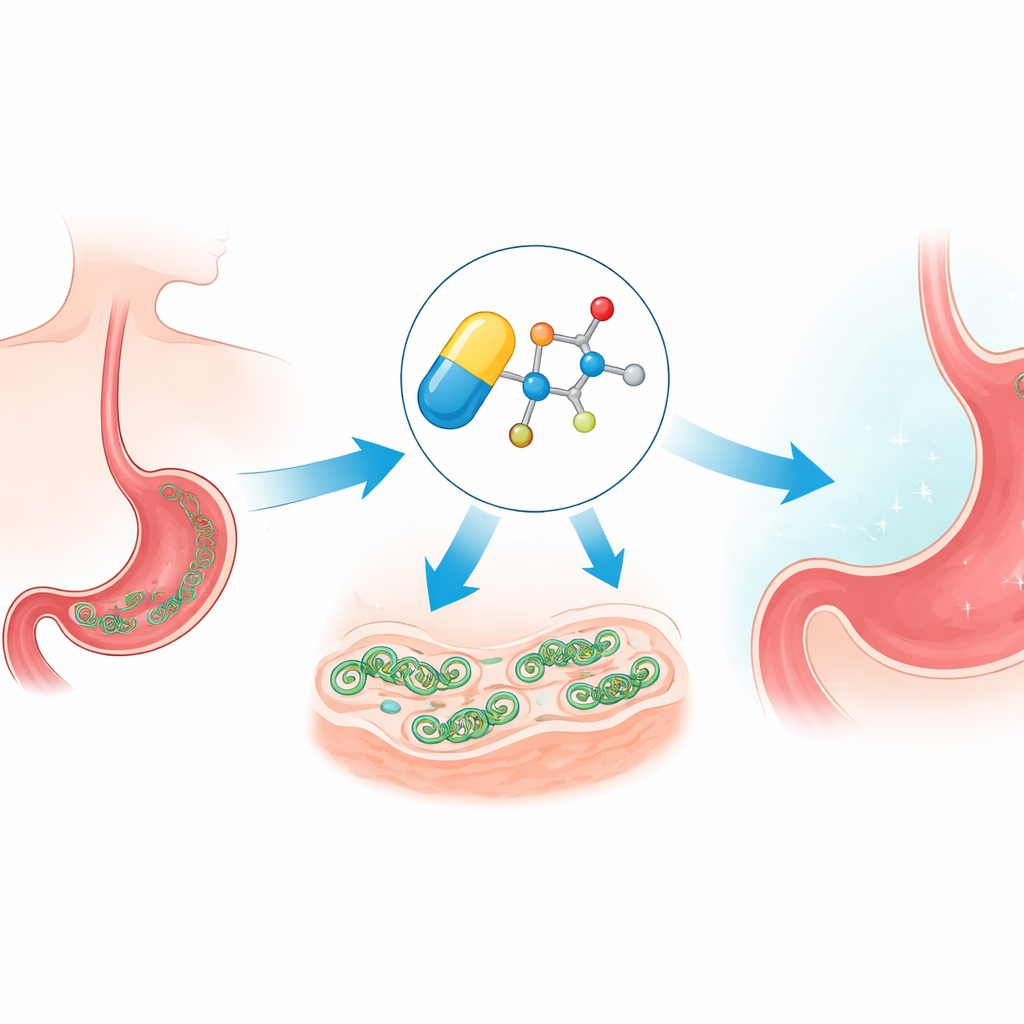
How the drug sneaks in and overloads the cell
Metronidazole is a so‑called prodrug: once it drifts into a microbe, its nitro group is chemically reduced to form aggressive radical species. These reactive fragments attack vital molecules such as DNA and proteins, creating oxidative stress. The team confirmed that the new ether versions still follow this path, generating stress within H. pylori. But because these reactions depend on low-oxygen conditions, the compounds remain largely inactive in well‑oxygenated human tissues, which helps explain why this drug family has long been useful against microbes yet relatively safe for patients.
Hitting the germ’s emergency repair crews
To find out what else the modified drugs were doing, the scientists used activity‑based protein profiling, a chemical fishing method where drug-like probes latch onto their targets and can be pulled out and identified. This revealed two main protein “catches” in H. pylori: a folding helper called HpGroEL, which acts like a chaperone to refold damaged proteins, and an enzyme called HpTpx, which neutralizes harmful peroxides and protects DNA from oxidative damage. The ether-based compounds bound especially strongly to HpTpx, forming permanent attachments at a critical cysteine in its active site and sharply reducing its ability to detoxify peroxides. In effect, the drugs both create oxidative chaos and disable the cell’s fire brigade at the same time.
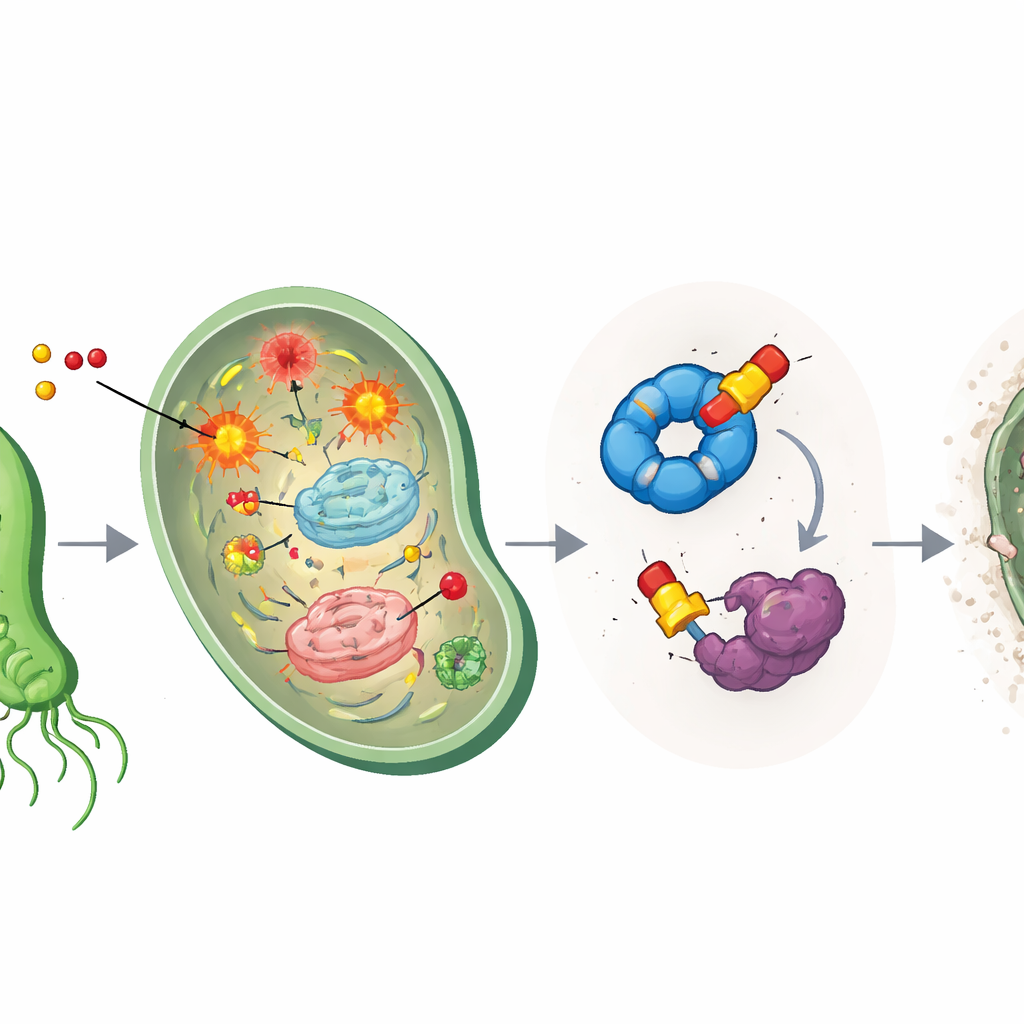
From test tube to mouse stomach
Potent antibiotics are useful only if they behave well in the body, so the team examined toxicity and how the new molecules move through animal systems. In human cell cultures, the ether derivatives showed very low toxicity, and chemical stability tests indicated they survived well in blood plasma. Pharmacokinetic studies in mice highlighted one standout compound, MF‑01, which reached favorable levels in the gut while maintaining good stability. In an H. pylori infection model that mimics human treatment—using a combination with clarithromycin and a stomach acid blocker—MF‑01 completely cleared the bacteria at about one‑fiftieth the standard metronidazole dose. At such low doses, the overall impact on the normal gut microbiome was milder than with conventional triple therapy, and common beneficial mouse gut bacteria were less sensitive to MF‑01 than to metronidazole.
What this means for patients and future drugs
Taken together, the work shows that metronidazole and its ether relatives act through a powerful one‑two punch: they trigger oxidative stress inside H. pylori while simultaneously blocking key stress‑response systems that would normally rescue the cell. This dual attack explains the dramatic increase in potency and suggests that carefully tuned derivatives could revive and improve an aging antibiotic class. Because these new molecules grow out of a long‑approved drug, they start with a head‑start on safety profiling. If future clinical studies confirm the mouse results, compounds like MF‑01 could enable shorter, lower‑dose regimens that more reliably cure H. pylori infections while being kinder to the rest of the gut ecosystem.
Citation: Fiedler, M.K., Pandler, M.S.I., Gong, R. et al. Metronidazole and ether derivatives target Helicobacter pylori via simultaneous stress induction and inhibition. Nat Microbiol 11, 1049–1063 (2026). https://doi.org/10.1038/s41564-026-02291-w
Keywords: Helicobacter pylori, metronidazole, antibiotic resistance, oxidative stress, gut microbiome