Clear Sky Science · en
Collective asymmetric synthesis of the Strychnos alkaloids via thiophene S,S-dioxide cycloadditions
Why tiny poison-inspired molecules matter
The Strychnos alkaloids are a family of natural compounds famous for their extreme potency—strychnine being the notorious example—and for their knotty, three‑dimensional shapes. These molecules have long fascinated chemists because they are both dangerous and potentially useful, with some showing promise against drug‑resistant cancers. Yet their architectural complexity makes them very hard to build in the lab. This paper unveils a new, streamlined way to construct many different Strychnos molecules in a controlled, efficient manner, opening doors to safer study and possible new medicines derived from these once‑fearsome poisons.
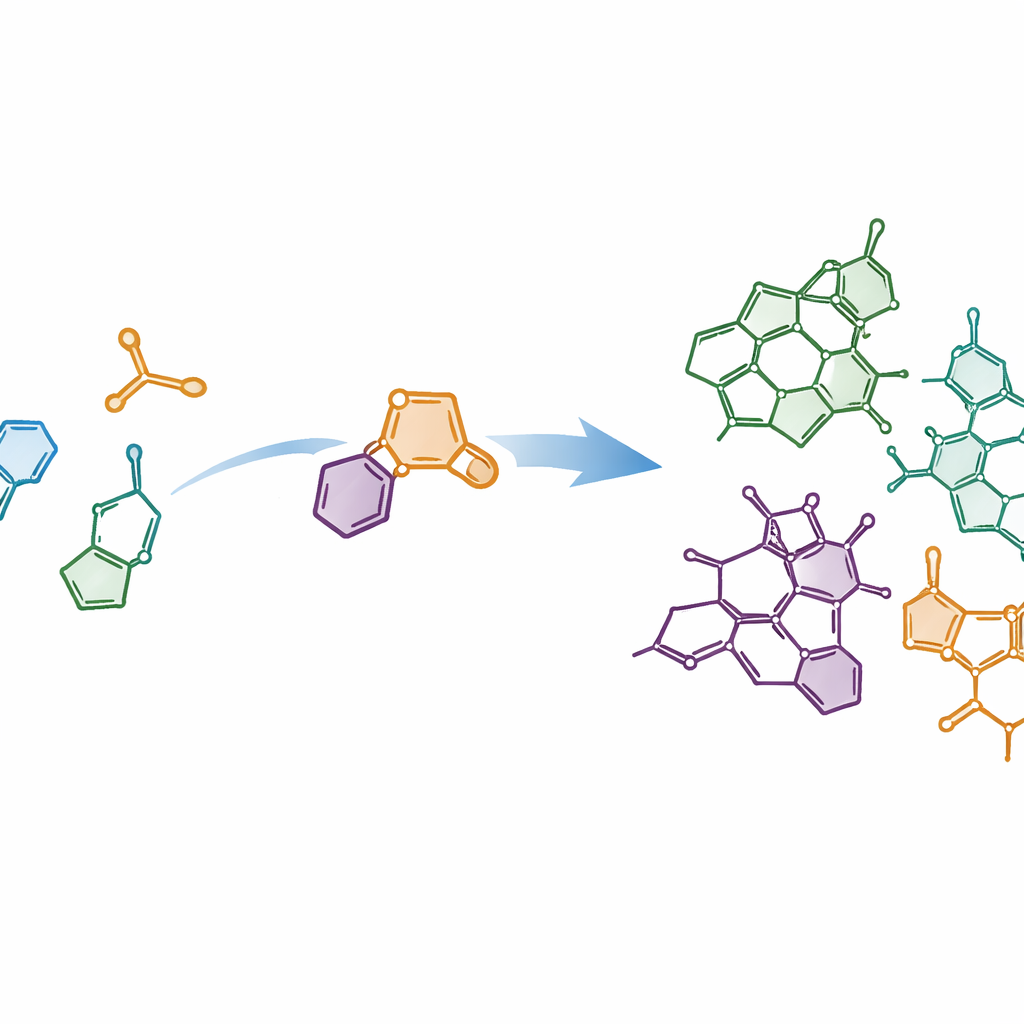
Building complex shapes from simple pieces
The authors set out to meet modern ideals of chemical synthesis: reactions that are short, waste‑lean and able to produce multiple related molecules from a common pathway. They focused on the dense, ring‑packed “core” shared across Strychnos alkaloids and asked whether this framework could be snapped together from simpler building blocks in just a few steps. Their plan relied on joining an indole fragment—a common scaffold in biology—to a special partner called a thiophene S,S‑dioxide. When these two meet, they undergo a powerful ring‑forming event called a Diels–Alder cycloaddition, followed by the clean loss of sulfur dioxide gas. This cascade converts relatively flat starting pieces into complex, three‑dimensional ring systems in essentially one stroke.
A versatile chemical shortcut
To turn this idea into a practical toolbox, the team first developed highly selective versions of these cycloaddition reactions. By attaching a cheap, chiral “handle” (a camphorsultam group) to the thiophene S,S‑dioxide, they could steer the reaction to form almost exclusively one mirror‑image product—a crucial feature for bioactive molecules, which often behave very differently from their mirror twins. They showed that a wide range of indole partners, bearing many different substituents, react smoothly under mild conditions to give tricyclic indoline products as single diastereomers. These products are not only stepping‑stones to Strychnos alkaloids but are also attractive frameworks for medicinal chemistry in their own right.
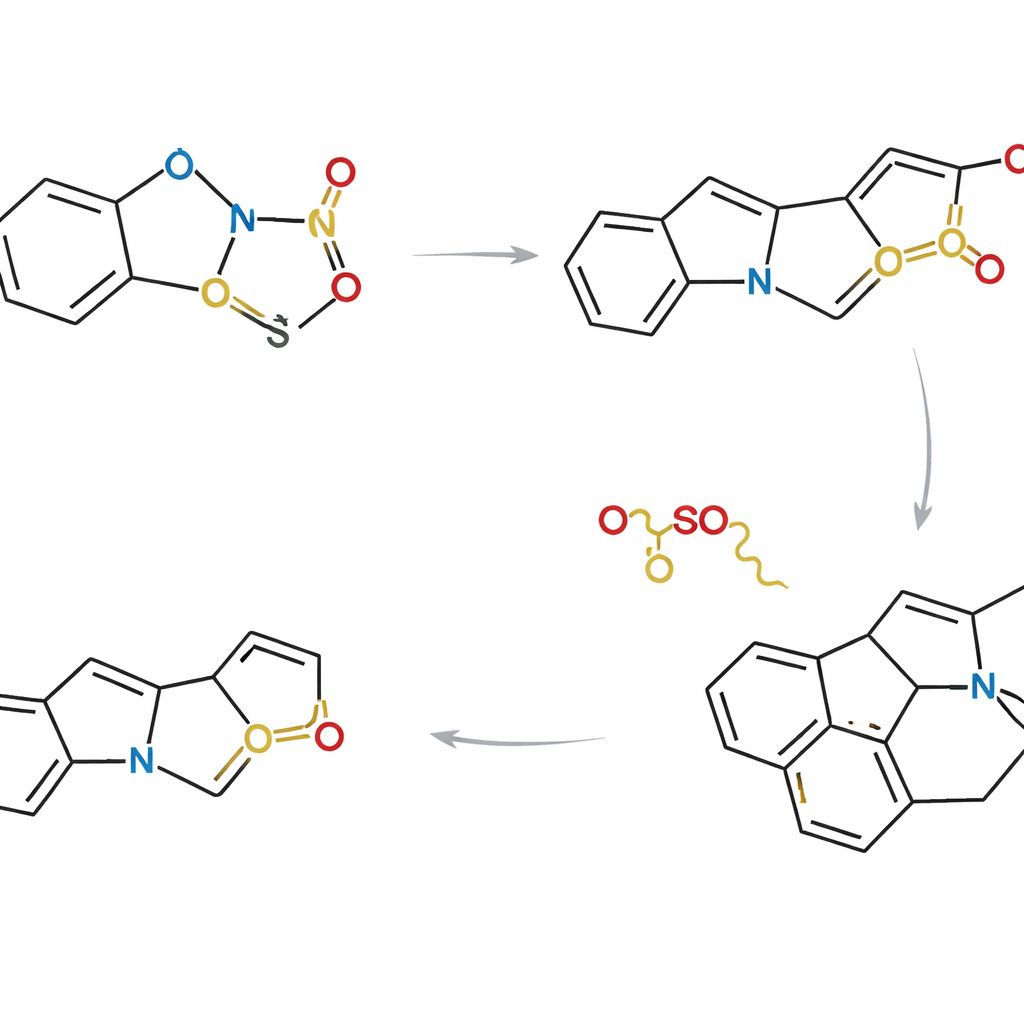
Collecting many natural products in one strategy
Armed with this control, the researchers designed three complementary routes that differ in how and when the indole and thiophene partners are linked. In one approach, the two fragments are tethered together before the cycloaddition, so the reaction happens within a single molecule. In others, they first react as separate molecules and then undergo internal ring closure. By carefully choosing substituents and reaction sequences, the team could converge on a common advanced intermediate that sits at the branching point of the Strychnos family tree. From this shared scaffold, they completed asymmetric syntheses of eight natural products, including akuammicine, lagumicine, norfluorocurarine, several alstolucines and echitamidine—often in the shortest and most atom‑efficient routes reported to date.
Cracking a century‑old synthetic challenge
One of the most striking demonstrations is the first total synthesis of brucine, a close chemical cousin of strychnine that has been known for more than 200 years and widely used as a chiral resolving agent, yet had never been fully built from scratch. Brucine’s indoline ring is unusually electron‑rich and prone to degradation, which has discouraged previous attempts. By tailoring their thiophene‑based cascade and carefully managing reaction conditions, the authors were able to forge brucine’s delicate framework in only nine linear steps from a simple commercially available starting material. They also achieved a ten‑step asymmetric synthesis of strychnine itself, the most concise chiral route reported so far.
Watching reactions with machine learning
To understand why their reactions give such precise outcomes, the team combined high‑level quantum chemistry with modern machine‑learning simulations. These calculations revealed that, in some cases, the reaction proceeds not in one smooth motion but through an uneven, stepwise dance: one carbon–carbon bond forms first, a shallow intermediate is briefly visited, and then sulfur dioxide is expelled almost spontaneously as the system slides down the energy landscape. The chiral camphorsultam side chain subtly biases this pathway, making one three‑dimensional arrangement much easier to reach than the other. This mechanistic insight provides guidelines for designing new thiophene‑based cascades beyond the Strychnos family.
From deadly toxins to useful tools
Overall, the study shows that thiophene S,S‑dioxides can act as powerful, programmable “spring‑loaded” pieces for snapping together intricate molecular architectures. By harnessing their built‑in drive to lose sulfur dioxide, the authors engineered cascade reactions that rapidly convert simple fragments into the tangled ring systems of Strychnos alkaloids with high selectivity and minimal wasted steps. For non‑specialists, the takeaway is that chemists are learning to turn even notorious poisons into versatile platforms for discovery, using clever reaction design and computational insight to explore complex chemical space more efficiently than ever before.
Citation: Park, K.H.‘., Park, J., Frank, N. et al. Collective asymmetric synthesis of the Strychnos alkaloids via thiophene S,S-dioxide cycloadditions. Nat. Chem. 18, 782–789 (2026). https://doi.org/10.1038/s41557-025-02041-1
Keywords: Strychnos alkaloids, asymmetric synthesis, cycloaddition cascades, thiophene S,S-dioxides, natural product chemistry