Clear Sky Science · en
Engineered outer membrane vesicles enhance solid tumour CAR-T cell therapy
Rebooting the Body’s Cancer Fighters
Many of the most dramatic cancer remissions in recent years have come from CAR-T cell therapy, in which a patient’s own immune cells are reprogrammed to hunt cancer. Yet for solid tumors like breast and liver cancers, this approach often stalls: the tumor’s protective neighborhood and shifting molecular “ID tags” allow malignant cells to hide or fight back. This study introduces a clever helper system based on tiny bubbles from bacteria that can both open up the tumor’s defenses and paint clear targets on tumor cells, so CAR-T cells can find and destroy them more effectively.
Why Supercharged T Cells Struggle in Solid Tumors
CAR-T cells recognize cancer cells by latching onto specific markers on their surface. Blood cancers usually display those markers consistently, but solid tumors are patchy: some cells carry the target, others do not, and surviving cells can simply stop showing it. At the same time, solid tumors are surrounded by a hostile environment packed with cells and signals that shut T cells down. One key brake is a molecular handshake between PD-L1 on tumor cells and PD-1 on T cells, which tells the immune system to stand down. As CAR-T cells start attacking, they release inflammatory molecules that paradoxically can drive PD-L1 levels even higher, further blunting their own impact.
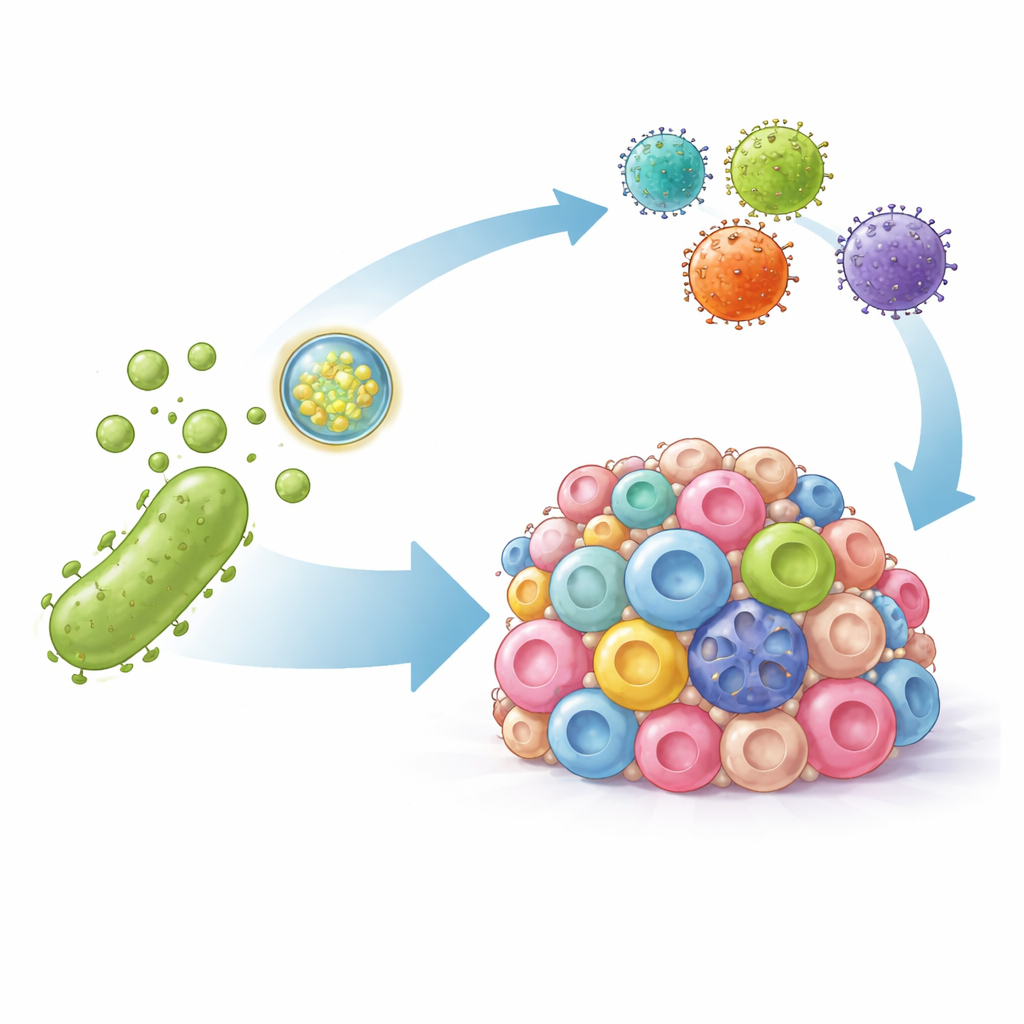
Turning Bacterial Bubbles into Precision Couriers
The researchers turned to bacterial outer membrane vesicles—naturally occurring nanoscale bubbles shed by bacteria that can stir up immune responses. They engineered these vesicles in two ways. First, they decorated their surface with a fragment of an antibody that sticks tightly to PD-L1, so the vesicles home in on tumor cells and physically block this immune-dampening signal. Second, they packed the vesicles with DNA instructions encoding an artificial version of the HER2 marker, a well-known target for CAR-T cells. Once delivered into tumor cells, this DNA makes the cells temporarily display HER2 on their surface, effectively tagging even previously invisible cancer cells for attack.
Rewriting the Tumor Neighborhood
In mouse models of breast cancer, the modified vesicles accumulated efficiently in tumors and reshaped the local immune landscape. Tumors treated with these vesicles showed fewer suppressive cell types, such as regulatory T cells, certain macrophages, and myeloid-derived suppressor cells, and more aggressive defenders like killer T cells and natural killer cells. These T cells produced more perforin and granzyme—molecules that punch holes in cancer cells—and more inflammatory messengers such as interferon-gamma and tumor necrosis factor. By blocking PD-L1 while also activating innate immunity, the vesicles turned a cold, protective tumor neighborhood into one that welcomes and sustains an immune assault.
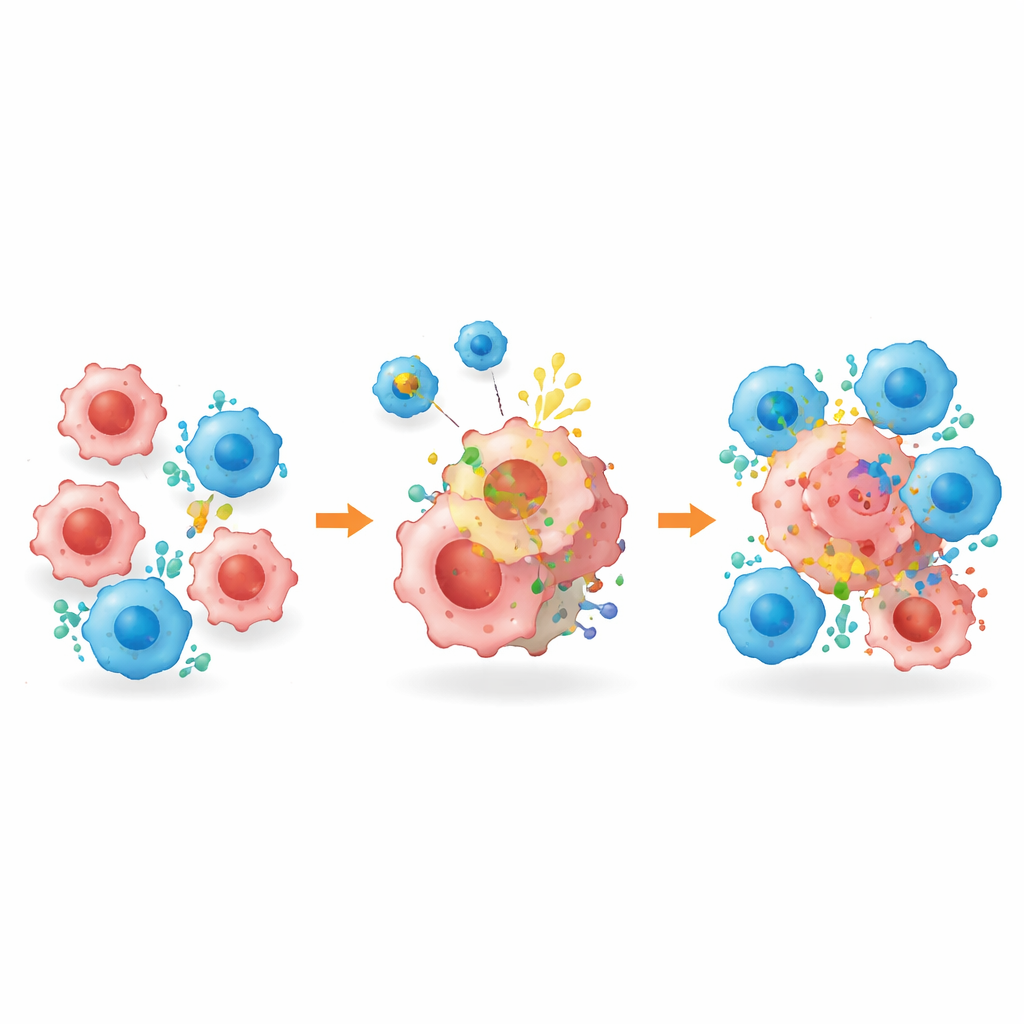
Tagging Hidden Tumor Cells for Elimination
Beyond remolding the tumor environment, the platform—named BROAD-CAR—directly tackled the problem of missing targets. In mixed tumors containing both HER2-positive and HER2-negative cells, conventional HER2-directed CAR-T therapy initially shrank tumors but left behind cells that had lost the target, leading to regrowth. When combined with BROAD-CAR, however, tumors were nearly wiped out: the vesicles caused HER2 to appear on formerly negative cells, allowing CAR-T cells to recognize and kill them. The same strategy worked in liver tumors that had no HER2 to begin with. Importantly, detailed analyses showed that HER2 decoration remained confined to tumor tissue, with no detectable expression in healthy organs or immune cells, and the treatment caused no obvious organ damage or weight loss in mice.
Lasting Protection Against Return and Spread
The benefits extended beyond the initial tumor. In breast cancer models, the combined BROAD-CAR and CAR-T treatment not only shrank primary tumors but also sharply reduced lung metastases. Mice that were fully cured resisted later re-challenge with cancer cells, whether or not those cells expressed HER2, suggesting that the intense initial battle triggered a broader, long-lived immune memory against the tumor. This hints that the platform may not just boost a single engineered T-cell product, but also help the wider immune system learn to recognize the cancer more completely.
What This Could Mean for Future Cancer Care
Overall, the study shows that bacterial vesicles can be turned into safe, adaptable helpers that make CAR-T therapy work far better against stubborn solid tumors. By both lifting immune brakes and painting clear, temporary targets on tumor cells, the BROAD-CAR system let engineered T cells clear diverse and even targetless cancers in mice, while avoiding healthy tissues. If similar results hold in humans, this approach could expand CAR-T treatment to many more patients with solid tumors and make relapses from antigen loss less likely, offering a powerful new way to help the immune system finish what it starts.
Citation: Li, X., Li, X., Shi, J. et al. Engineered outer membrane vesicles enhance solid tumour CAR-T cell therapy. Nat. Biomed. Eng 10, 660–675 (2026). https://doi.org/10.1038/s41551-025-01575-6
Keywords: CAR-T cell therapy, solid tumors, tumor microenvironment, bacterial outer membrane vesicles, cancer immunotherapy