Clear Sky Science · en
Extracellular matrix-driven patient stratification and network modeling reveal distinct molecular grades with potential clinical implications
Why the tumor’s surroundings matter
When we think about cancer, we often picture rogue cells growing out of control. But those cells live in a complex neighborhood made of proteins and sugars that form a support mesh called the extracellular matrix. This mesh is far from passive. It can stiffen, thicken, and send signals that help tumors grow or resist treatment. In this study, researchers asked whether the makeup of that mesh in lung tumors could be used to sort patients into meaningful groups and guide therapy choices.
Sorting tumors by their structural “scaffolding”
The team focused on lung adenocarcinoma, a common type of lung cancer, and analyzed 101 patients whose tumors had been deeply profiled at the DNA, RNA, and protein levels. They used a curated list of genes that build and regulate the matrix to create an “ECM barcode” for each patient. This barcode captured how much each tumor’s matrix differed from that patient’s nearby normal lung tissue. When they grouped patients using these barcodes, four distinct “ECM grades” emerged, ranging from almost normal-like matrix to tumors wrapped in a dense, complex mesh.
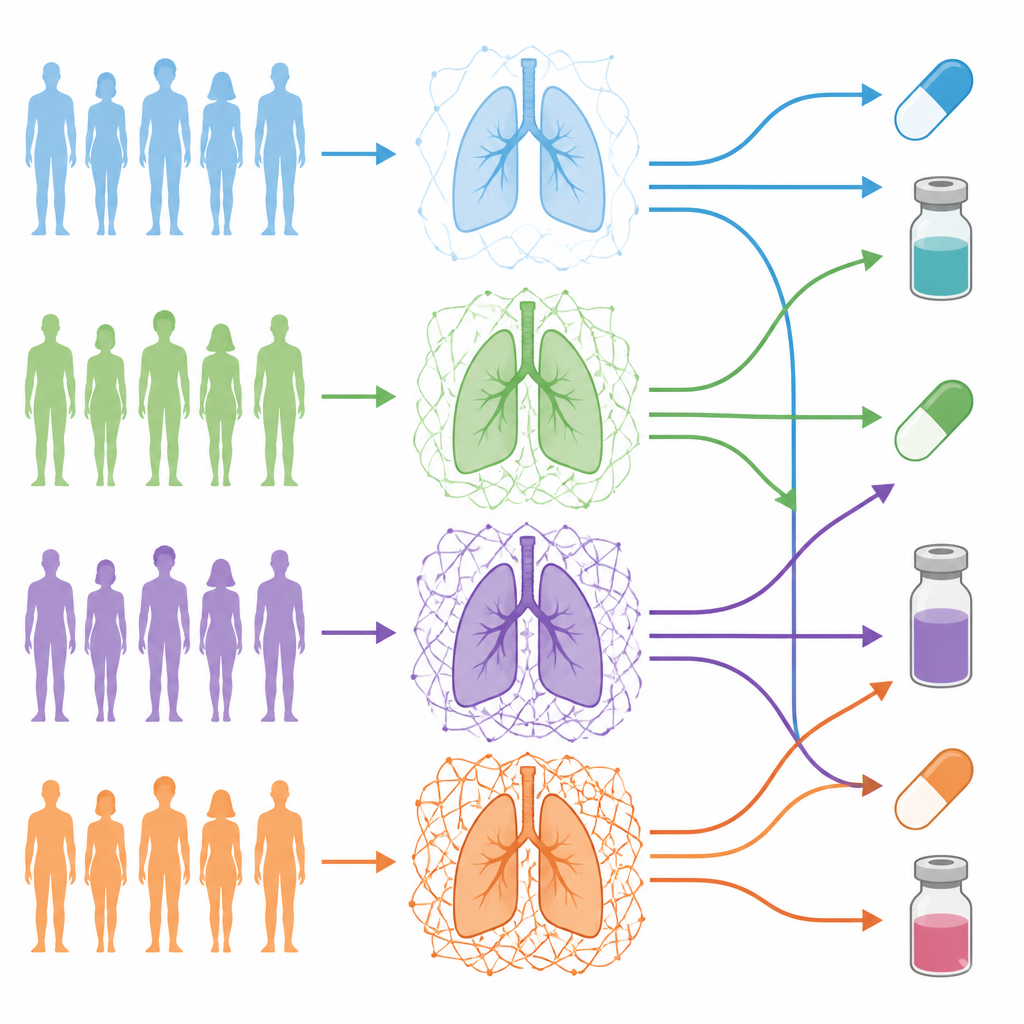
What high and low grades reveal about the tumor
Tumors with the highest ECM grades were packed with stromal cells such as cancer-associated fibroblasts, and had lower fractions of cancer cells themselves, a pattern linked to more advanced tumor microenvironments. These ECM-rich tumors showed stronger signals associated with epithelial to mesenchymal transition, invasiveness, cancer stem-like cells, and blood vessel growth. Patients in the highest ECM grade tended to have more aggressive clinical features, more tumor mutations, and a higher short-term risk of death or cancer coming back, compared with patients whose tumors had sparse matrix.
Hidden wiring between the matrix and cell behavior
To uncover how the outer matrix connects to inner cell behavior, the researchers built network models for each patient that linked altered matrix proteins to the signaling proteins and transcription factors they influence. Combining these networks showed that higher ECM grades were associated with stronger activity in pathways that regulate cell movement, survival, and adaptation to mechanical stress. Signals that encourage cells to loosen their attachments, migrate, and invade were more active, while protective factors that normally suppress invasion were weaker in ECM-rich tumors. The study also found that certain well known cancer mutations, such as those in KRAS and EGFR, tended to appear in different ECM grades, suggesting that the matrix environment and genetic changes may reinforce one another.
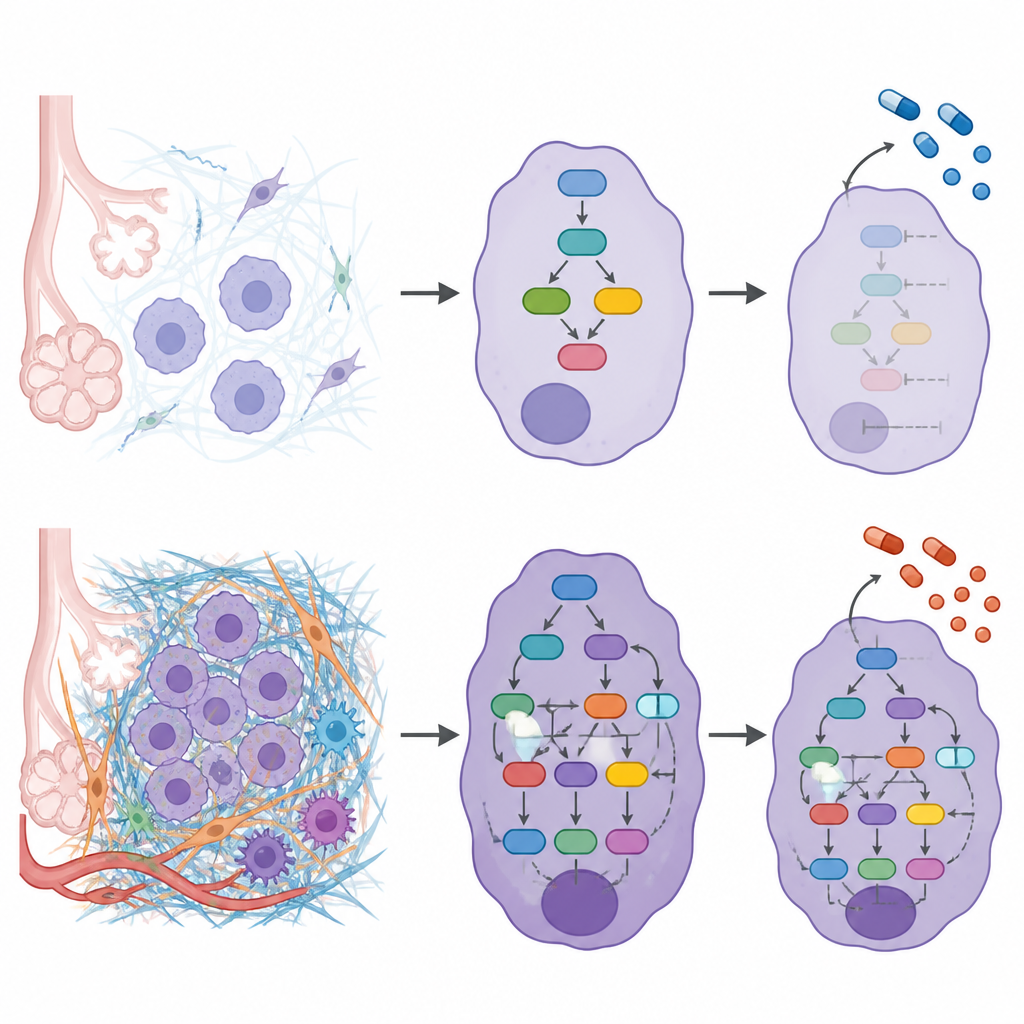
Linking the matrix to treatment choices
Because the matrix can shape how drugs reach and affect tumor cells, the team next asked whether different ECM grades might respond differently to existing cancer medicines. Using their network maps, they measured how “close” each drug’s targets were to the matrix-driven signaling hubs in each grade. This revealed medicines predicted to work best in specific ECM settings and others likely to act similarly across grades. One drug that blocks DNA repair enzymes, olaparib, appeared especially connected to low ECM grades, while an EGFR-targeting drug, erlotinib, looked broadly effective regardless of ECM grade.
Putting predictions to the test in the lab
The researchers then recreated simplified versions of low and high ECM environments in the lab using lung cancer cell lines carrying either KRAS or EGFR mutations. They grew the cells on healthy lung-derived matrix to mimic a low-grade environment and on a tumor-like basement membrane to mimic a richer matrix. Consistent with the network predictions, olaparib was more potent against EGFR-mutant cells on the healthy-like matrix than on the tumor-like one, while its effect on KRAS-mutant cells did not depend much on the matrix. In contrast, erlotinib reduced cell viability to a similar degree in both matrix conditions, matching its ECM-insensitive profile from the network analysis.
What this means for patients
This work suggests that the tumor’s supporting scaffold is not just a backdrop but a key feature that can be measured, grouped, and linked to how tumors grow and respond to drugs. By classifying lung cancers into ECM grades and mapping the signaling networks that connect the matrix to cell behavior, clinicians may eventually be able to select therapies that fit both the genetic makeup of the cancer and its physical surroundings. While more validation is needed before this approach can guide routine care, it points toward a future in which the structure around the tumor helps decide which treatments are most likely to work.
Citation: Dansık, A., Sarıca, S., Öztürk, E. et al. Extracellular matrix-driven patient stratification and network modeling reveal distinct molecular grades with potential clinical implications. npj Syst Biol Appl 12, 66 (2026). https://doi.org/10.1038/s41540-026-00697-0
Keywords: extracellular matrix, lung adenocarcinoma, tumor microenvironment, cancer networks, drug response