Clear Sky Science · en
Cell loss disrupts mechanical homeostasis to drive retinal pigment epithelium ageing-like phenotype in vitro
Why aging eye cells struggle to keep vision clear
As we age, many people develop problems with central vision, often tied to damage in a thin layer of cells at the back of the eye called the retinal pigment epithelium (RPE). These cells quietly work every day to support the light‑sensing photoreceptors above them, yet how simple cell loss with age changes their physical state and undermines their job has been hard to pin down. This study recreates an aging‑like scenario in the lab and shows that losing even a modest number of RPE cells can mechanically "rewire" the remaining tissue in ways that make it stiffer, less flexible, and worse at its core task of cleaning up visual waste.
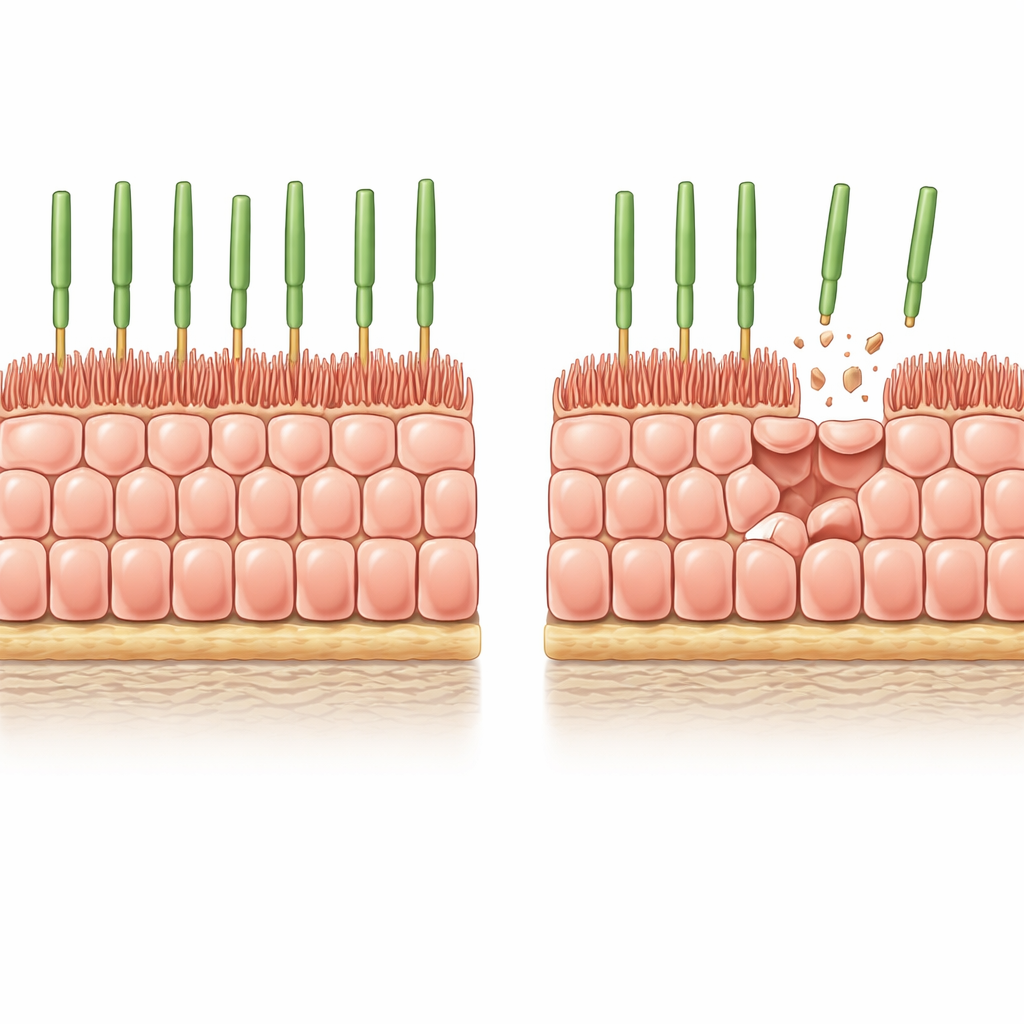
A careful laboratory model of aging eye tissue
The researchers began with human stem‑cell‑derived RPE grown as a uniform, tightly packed sheet on a soft, biologically realistic gel. Under these conditions the cells stop dividing, adopt the classic honeycomb pattern seen in healthy RPE, and develop finger‑like microvilli on their top surface that interact with photoreceptors. To mimic the gradual cell loss that happens in the aging eye, the team introduced a controllable "suicide" switch into the cells and then triggered widespread but partial cell death. Over a few days, about 8% of the cells disappeared, and the survivors had to stretch to cover the gaps without help from new cell division.
How the cell layer reshapes itself after loss
After this induced thinning, the remaining RPE cells grew wider but became noticeably shorter, preserving their volume while spreading sideways to seal the tissue. Their apical microvilli shrank and became more disorganized, and some cells developed thick actin stress fibers running straight across them—features also seen in aged human RPE. Measurements confirmed that key microvilli‑associated proteins, such as ezrin and its active phosphorylated form, were reduced. In other words, simple loss of neighbors was enough to push structurally young cells into an architecture that strongly resembles that of old RPE in the eye.
When stiffness trades off against cleaning ability
Structure in this tissue is not just about looks; it underpins function. RPE cells must ingest and digest countless fragments from photoreceptor outer segments every day. Using fluorescently labeled fragments from pig retinas, the team measured this cleanup activity. Density‑reduced RPE sheets internalized fewer fragments overall, yet those they did engulf tended to be larger. High‑resolution imaging showed why: in healthy sheets, the apical surface bulged upward to form well‑defined cups around each fragment, supported by dynamic actin and myosin rearrangements. In density‑reduced sheets, the surface looked smoother and less deformable, with fewer clear cup shapes and the appearance of large actin‑rich protrusions that did not seem to help engulf particles.
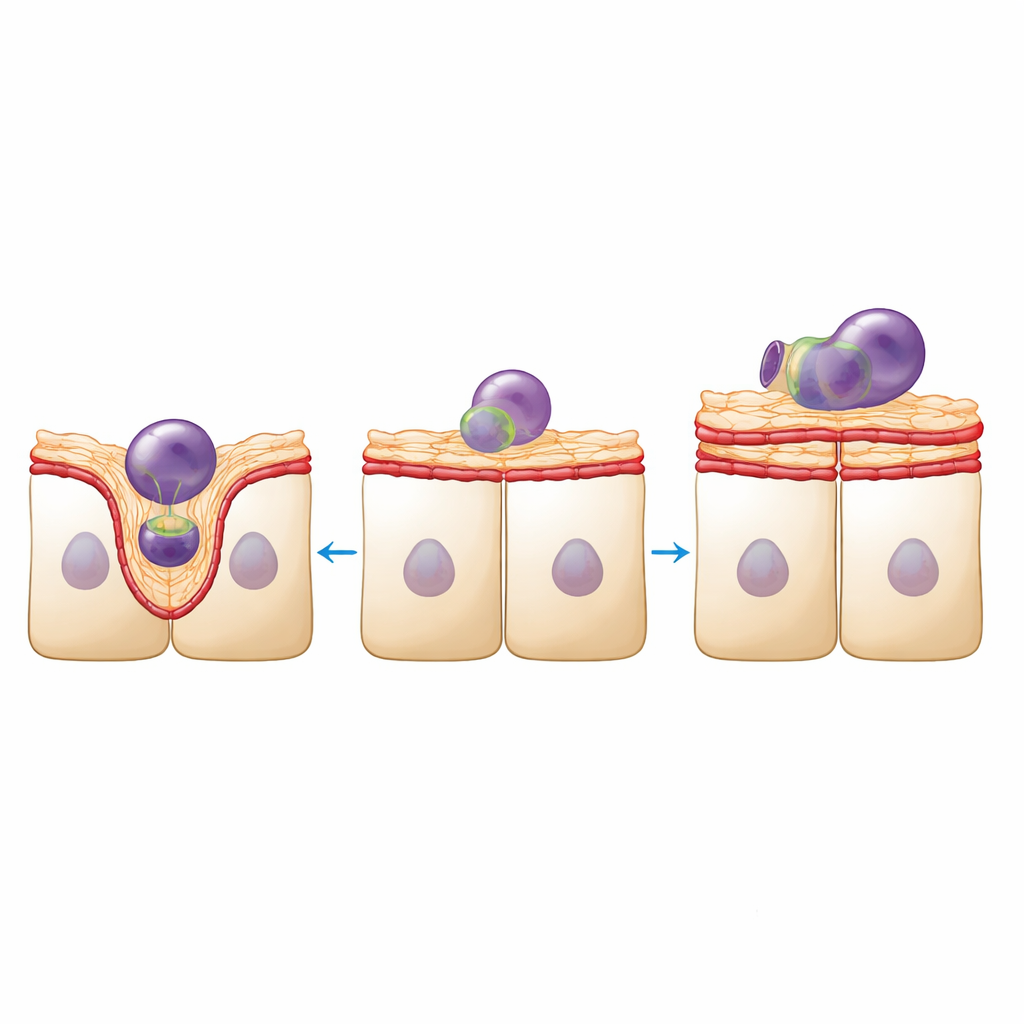
Mechanical homeostasis shifts toward rigidity
To link these visual changes to mechanics, the authors probed the tissue’s physical properties at two scales. Nanoindentation, which presses a tiny spherical tip into the apical surface, revealed that the density‑reduced RPE became roughly one‑third stiffer and more elastic as a multicellular sheet. At the same time, molecular markers of contractility and junction strength—phosphorylated myosin light chain and the adhesion protein vinculin—were enriched along cell‑cell borders. Yet Brillouin microscopy, a light‑based method that senses local stiffness and water content inside cells, showed that the apical cortex itself actually became mechanically distinct and, in some respects, softer. Together, these findings suggest that the monolayer reinforces its lateral connections to keep the sheet intact while the apical surface loses the finely tuned microstructure that normally allows it to bend and cup around particles.
Actin regulators steer the balance between strength and flexibility
Gene expression analysis pointed to broad rewiring of the actin machinery in density‑reduced RPE. Proteins that promote branched actin networks and crosslinking were upregulated, while several formins, which favor long, linear filaments, and ezrin were downregulated. The team then pharmacologically nudged these systems. Inhibiting formins in normal RPE stiffened the sheet and made engulfed fragments larger, partially mimicking the aging‑like state. In density‑reduced RPE, blocking the Arp2/3 complex, a key branched‑actin nucleator, increased the number of fragments internalized and restored more pronounced bulging of the apical surface. These interventions show that tweaking actin nucleation can shift the tissue back and forth between a more rigid, protective configuration and a more deformable, phagocytosis‑friendly one.
What this means for aging eyes
Taken together, the work shows that modest cell loss alone can push RPE tissue into a new mechanical equilibrium: the sheet becomes more strongly stitched together and stiffer as a whole, but its top surface loses the plasticity needed to efficiently engulf photoreceptor debris. In effect, the cells prioritize holding the tissue together over performing their specialized cleaning duty. This structural trade‑off, driven by changes in the actin cytoskeleton and junctional tension rather than by classical biochemical aging damage, may help explain why aged RPE often looks intact yet functions poorly. Understanding and potentially tuning this balance between stability and flexibility could open new paths to preserving vision in disorders such as age‑related macular degeneration.
Citation: Piskova, T., Kozyrina, A.N., Astrauskaitė, G. et al. Cell loss disrupts mechanical homeostasis to drive retinal pigment epithelium ageing-like phenotype in vitro. Nat Commun 17, 3404 (2026). https://doi.org/10.1038/s41467-026-71493-x
Keywords: retinal pigment epithelium, cell mechanics, age-related macular degeneration, actin cytoskeleton, phagocytosis