Clear Sky Science · en
Quantitative live imaging reveals PRICKLE1 controls junctional neural tube morphogenesis independent of Planar Cell Polarity
How Early Nerve Formation Shapes Our Brains and Spines
Before a human embryo is even the size of a grain of rice, a flat sheet of cells must fold and seal into a hollow tube that will become the brain and spinal cord. When this process goes wrong, babies can be born with serious spinal defects. This study uses cutting-edge live imaging in tiny quail embryos to watch this transformation in real time, revealing a surprising cell behavior and a key control protein that together help prevent a specific class of spinal problems.
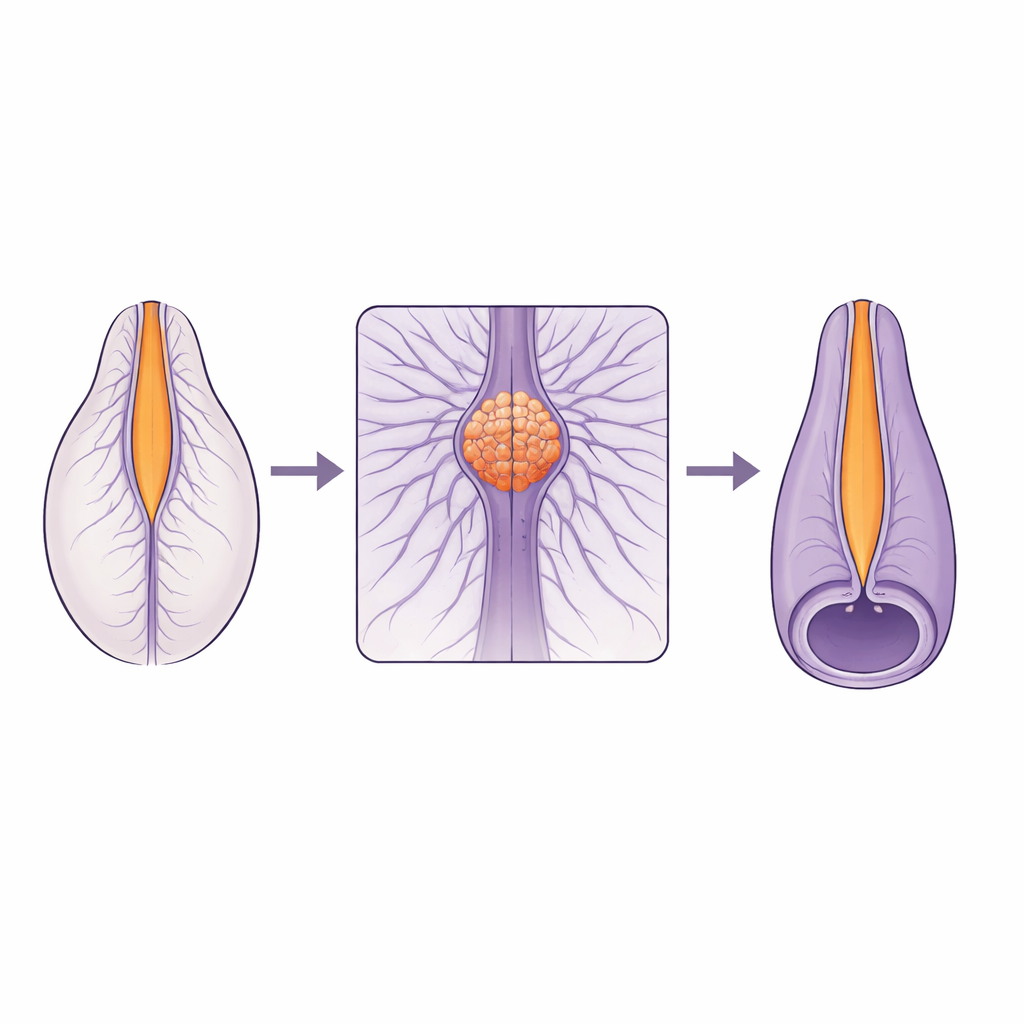
A Delicate Join in the Growing Spine
The spinal cord does not form in a single way along its length. The upper part arises by folding and closing of a flat tissue, while the lower part forms from a solid rod of cells that hollows out from within. These two construction programs must meet and merge smoothly at a small area called the junctional zone. Many human spinal defects cluster near this junction, suggesting it is especially fragile. Yet until now, scientists lacked the live, high‑resolution views needed to understand how cells actually move and change shape in this zone as the tube closes.
Watching Cells Move and Dive Beneath the Surface
The researchers turned to transgenic quail embryos whose cells glow under the microscope, allowing them to track hundreds of individual cells over hours. They found that the junctional neural tube forms through two coordinated movements. First, cells from the sides crawl toward the midline, a motion called mediolateral convergence that narrows the tissue. Second, a stripe of cells right at the center does something more dramatic: these cells shrink their top (apical) surfaces, then drop beneath the surface into deeper tissue, a behavior known as ingression. This ingression resembles an epithelial‑to‑mesenchymal transition, in which orderly sheet‑like cells become more free‑moving, but here it happens in cells that still retain neural identity and go on to contribute mainly to the spinal cord rather than to other tissues.
A Surprising Role for a Known Polarity Protein
Earlier work had implicated a protein called PRICKLE1 in this junctional region. PRICKLE1 is usually known as part of the planar cell polarity system, which orients cells side‑to‑side and helps tissues elongate. One might expect that removing PRICKLE1 would scramble this polarity. Instead, when the team reduced PRICKLE1 levels specifically in the junctional zone, the usual polarity patterns and long actin cables that guide convergence remained largely intact. What changed was the behavior of the medial cells: they failed to constrict their apical surfaces, did not ingress properly, and the posterior opening of the neural tube remained unclosed, producing junctional neural tube defects in the embryos.
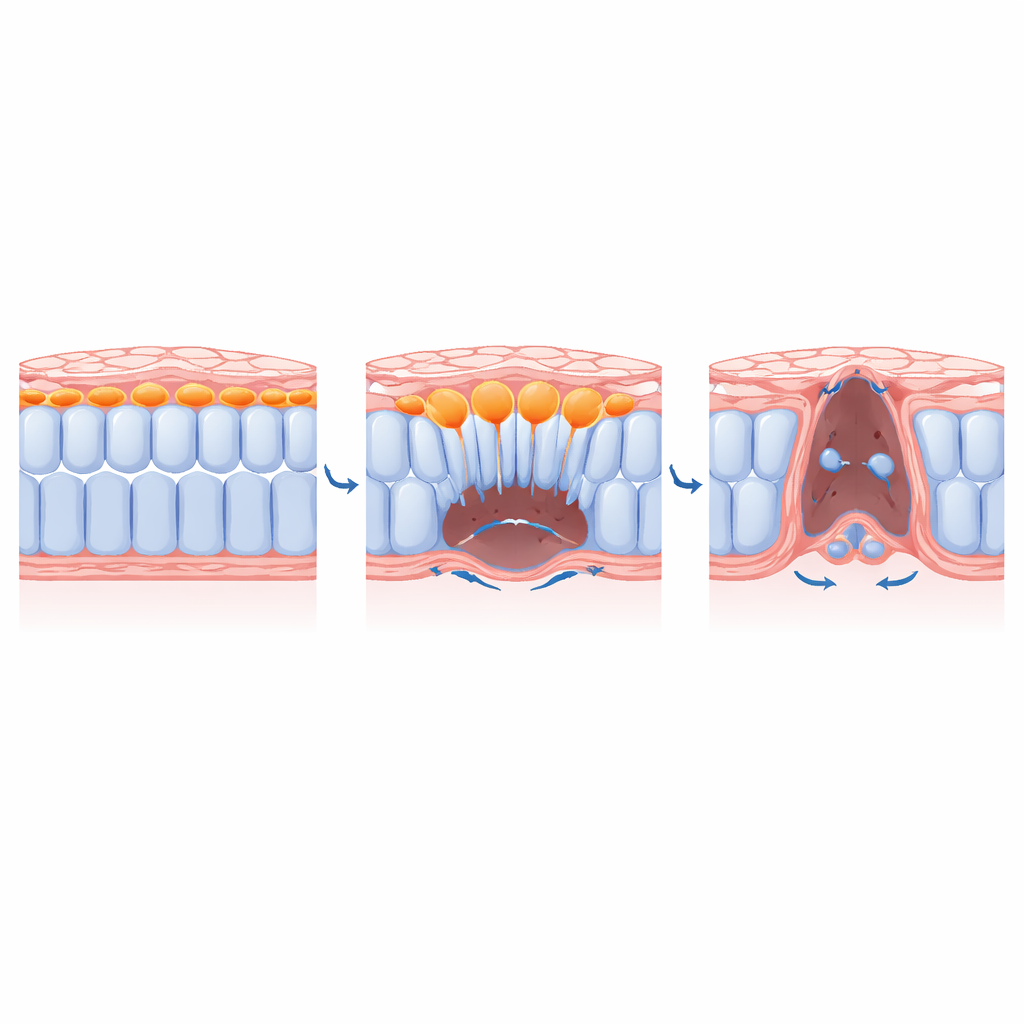
How Central Cells Make Space for Their Neighbors
By combining genetic tools, drugs, and even light‑activated inhibitors of myosin (the motor that powers contraction), the team dissected how ingression is controlled. They showed that key EMT regulators such as the transcription factor SLUG and signals from fibroblast growth factors are required for medial cells to adopt a protrusive, migratory shape and to break down the supporting matrix beneath them. Blocking these cues trapped medial cells near the dorsal surface and prevented them from moving ventrally. Crucially, when the researchers disabled contraction only in the medial cells, those cells no longer ingressed, and lateral neighbors could not fully converge toward the midline. In other words, the act of central cells diving inward physically clears space that allows the remaining sheet to zipper shut.
Why This Matters for Human Spinal Defects
The work reveals that PRICKLE1 has a PCP‑independent job at the apical cortex of medial cells: it promotes the build‑up of contractile actin and myosin needed for apical constriction and ingression. Without this function, even though side‑to‑side polarity cues persist, the central EMT‑like process stalls, and the neural tube fails to close at the junction. Because human mutations in PRICKLE1 are most often linked to localized spinal dysraphisms—junctional neural tube defects—these findings offer a concrete cellular explanation. For a lay reader, the take‑home message is that a tiny group of cells must both pull their tops tight and dive beneath the surface at just the right place and time, and that the protein PRICKLE1 is a critical switch for this motion. When that switch fails, the join between upper and lower spinal cord cannot form properly, helping to explain a specific and serious class of birth defects.
Citation: Wang, J.X., Alvarez, Y.D., Tan, S.Z. et al. Quantitative live imaging reveals PRICKLE1 controls junctional neural tube morphogenesis independent of Planar Cell Polarity. Nat Commun 17, 3654 (2026). https://doi.org/10.1038/s41467-026-71242-0
Keywords: neural tube defects, PRICKLE1, epithelial-to-mesenchymal transition, embryonic development, cell migration