Clear Sky Science · en
Modelling synaptic dysfunction in childhood dementia using human iPSC-derived cortical networks
Why this research matters to families
Childhood dementia is a devastating group of brain diseases in which children slowly lose memory, thinking skills and independence. One of the most common forms, Sanfilippo syndrome type A, currently has no cure and limited treatment options. This study uses advanced stem-cell methods to grow tiny human brain networks in the lab, allowing scientists to watch how neurons from affected children behave over time. By revealing exactly how these cells go wrong, the work points to new drug strategies that might protect thinking and learning, even if a child is diagnosed late.
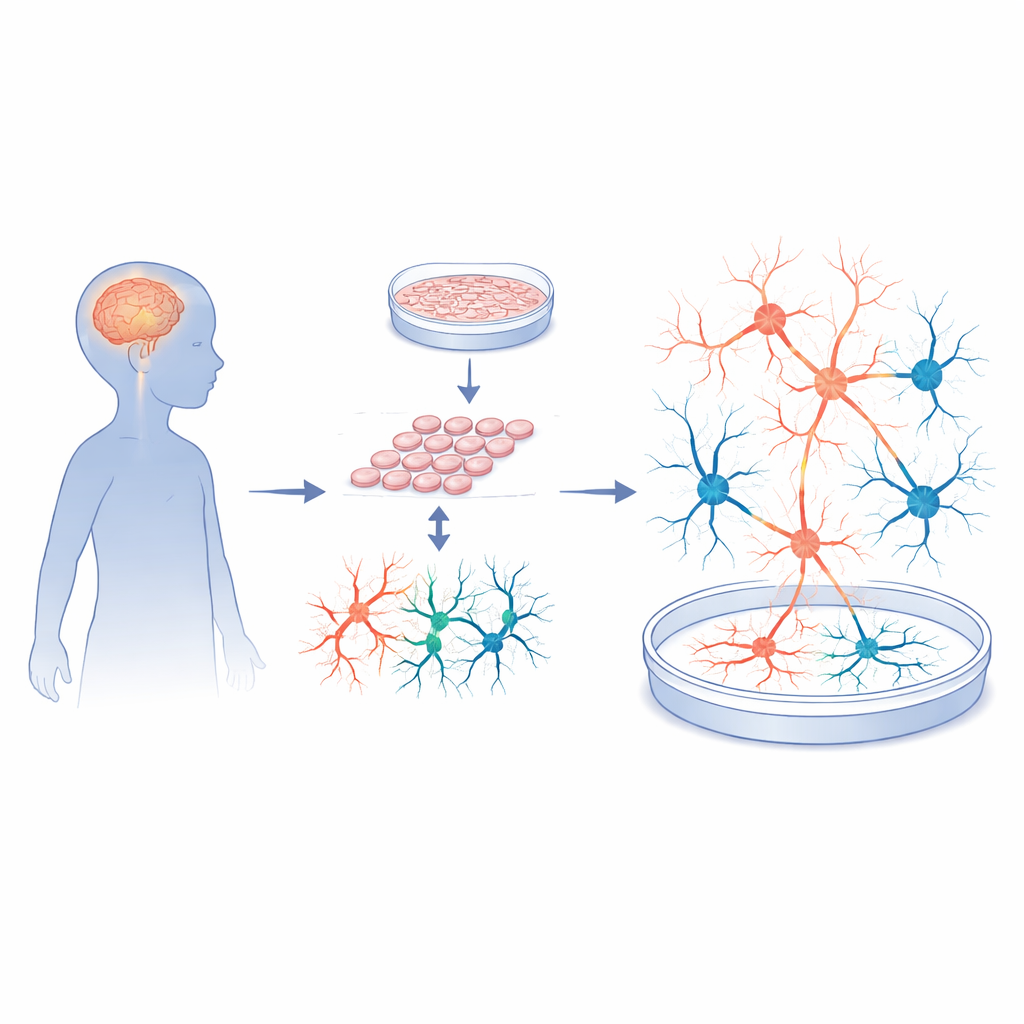
Turning skin cells into working brain circuits
The researchers started with skin biopsies from five children with Sanfilippo type A and five neurotypical, age-matched donors. They reprogrammed these skin cells into induced pluripotent stem cells, a flexible cell type that can become almost any tissue. These stem cells were then carefully guided to become cortical neurons and supporting cells, mimicking the region of the brain involved in learning, memory and behaviour. Rigorous quality checks showed that both patient and control stem cells grew well, had normal chromosomes, and could form high-quality neural progenitor cells. In other words, any later differences in brain-like activity were unlikely to be caused by flaws in the basic cell-making process.
Neurons that look and fire normally at first
Next, the team examined the shape and electrical behaviour of individual neurons. Using detailed imaging, they reconstructed hundreds of single cells and found that patient and control neurons had similarly complex branching patterns—suggesting that Sanfilippo does not stop these cells from building a rich network of connections. Electrical recordings showed that the neurons from both groups produced action potentials (the electrical spikes that carry information) with comparable strength, speed and thresholds. Key ion channels that generate these spikes also worked normally. Early in development, then, Sanfilippo neurons looked much like typical neurons and could fire just as well.
Hidden imbalance between “go” and “stop” signals
The problems appeared later, as the lab-grown networks matured. The team measured tiny currents flowing into neurons when neighbouring cells released chemical messengers. Over weeks, Sanfilippo neurons developed more excitatory contacts—synapses that send “go” signals—marked by increased clusters of the protein PSD95. Inhibitory contacts, which provide “stop” signals, lagged behind. This created an unhealthy tilt toward excitation. The excitatory currents in patient neurons became more frequent and larger, while inhibitory currents stayed largely unchanged. When the scientists introduced mild cellular stress by briefly removing growth factors and antioxidants, this excitatory bias in patient cultures became much more pronounced, whereas neurotypical networks were largely unaffected.
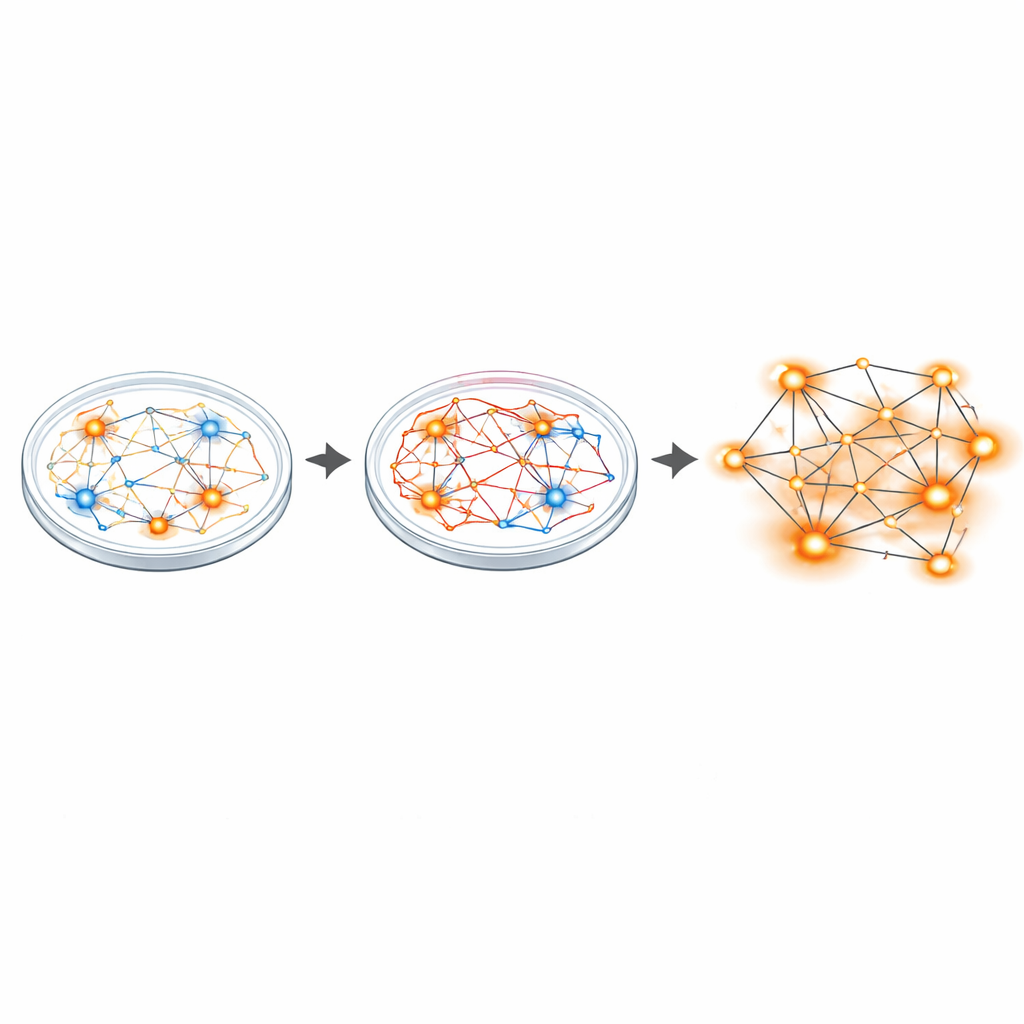
From overexcited synapses to hyperactive networks
To see how this imbalance plays out at the circuit level, the researchers used multielectrode arrays—tiny grids that record from many neurons at once. While single neurons from patients fired at about the same average rate as controls, the overall networks told a different story. Sanfilippo cultures had more active and bursting neurons and showed roughly twice as many synchronized network events, a pattern of coordinated firing that signals hyperactive circuits. Single-nucleus RNA sequencing of thousands of cells then revealed broad shifts in gene activity, especially in genes that build and strengthen glutamatergic (excitatory) synapses. Several of these genes have been linked to other brain conditions featuring imbalances between excitation and inhibition, such as autism and schizophrenia.
What this means for future treatments
For a non-specialist, the key message is that in this childhood dementia, the basic wiring of neurons is largely intact, but the balance between the brain’s accelerators and brakes goes awry over time. Neurons become driven too hard by excitatory input, leading to hyperactive networks that may set the stage for later degeneration and loss of cognitive abilities. Because the team could reproduce these changes in a dish using patient-derived cells, their model offers a powerful platform to search for drugs that restore balance between excitatory and inhibitory signals. Such treatments would not replace the missing enzyme that causes Sanfilippo, but they could help preserve thinking and behaviour—potentially improving quality of life for children living with this devastating disease.
Citation: Mazzachi, P., McDonald, E., Greenberg, Z. et al. Modelling synaptic dysfunction in childhood dementia using human iPSC-derived cortical networks. Nat Commun 17, 3161 (2026). https://doi.org/10.1038/s41467-026-71112-9
Keywords: childhood dementia, Sanfilippo syndrome, synaptic imbalance, induced pluripotent stem cells, neural network hyperexcitability