Clear Sky Science · en
Structural basis of the cyclin Y/14-3-3 protein-mediated activation of CDK16
Why this protein puzzle matters
Inside our cells, teams of proteins work together like tiny machines to control growth, survival, and even how cells respond to stress. One such protein, called CDK16, influences nerve cell wiring, recycling of cell contents, fertility, and cancer cell behavior. Yet CDK16 is oddly quiet unless two specific helper proteins, cyclin Y and 14-3-3, come together in just the right way. This study reveals, atom by atom, how that unusual trio assembles and how their handshake switches CDK16 from idle to active, offering new clues for designing future cancer drugs that target this control system.
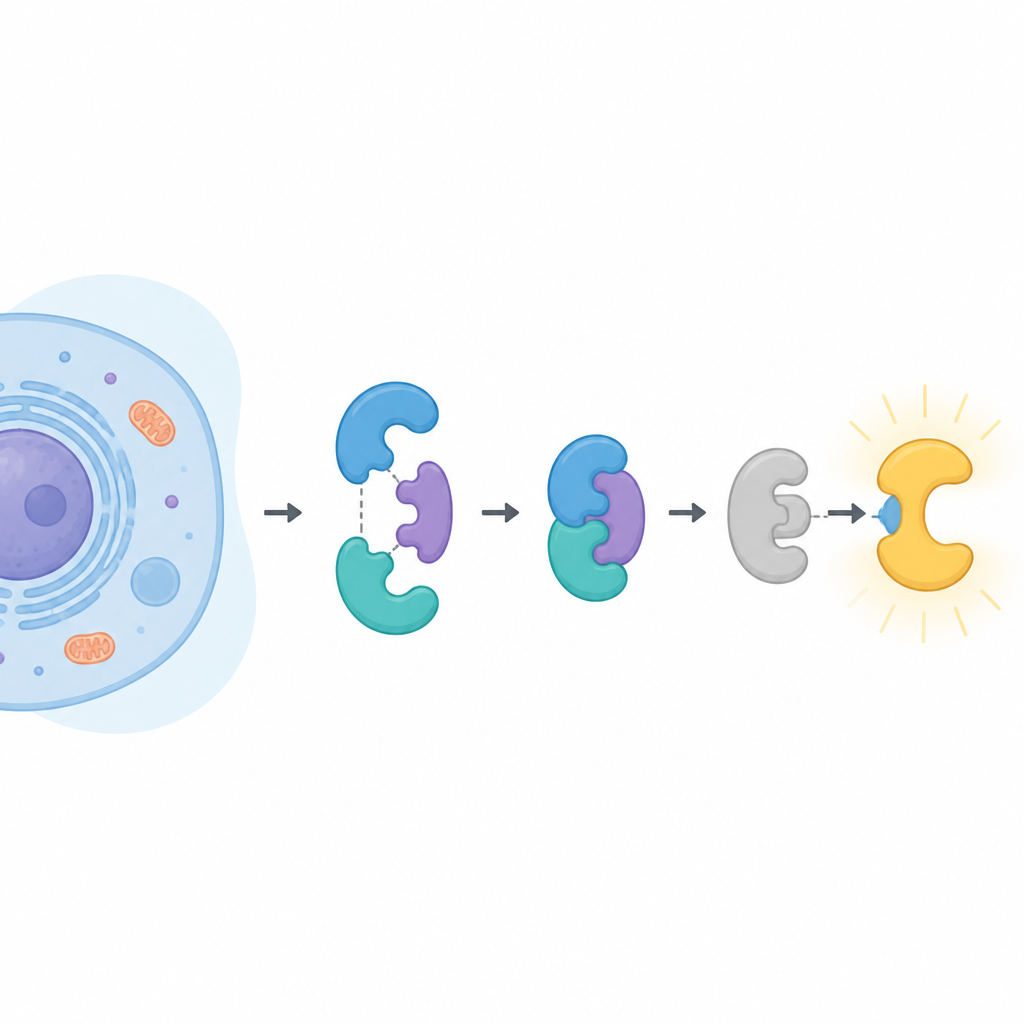
The key players in the cell
CDK16 belongs to a large family of enzymes that add small phosphate tags to other proteins, changing their activity. Most of its relatives are turned on simply by binding to a partner called a cyclin. CDK16 is different. It only wakes up when bound to cyclin Y, which itself must be decorated with phosphate tags and held in place by another protein pair known as 14-3-3. CDK16 has been linked to brain development, the final stages of sperm formation, the way cells recycle their own contents during starvation, and the growth and drug resistance of several cancers. Understanding exactly how CDK16 is controlled could therefore illuminate both normal biology and disease.
How the helper pair reshapes cyclin Y
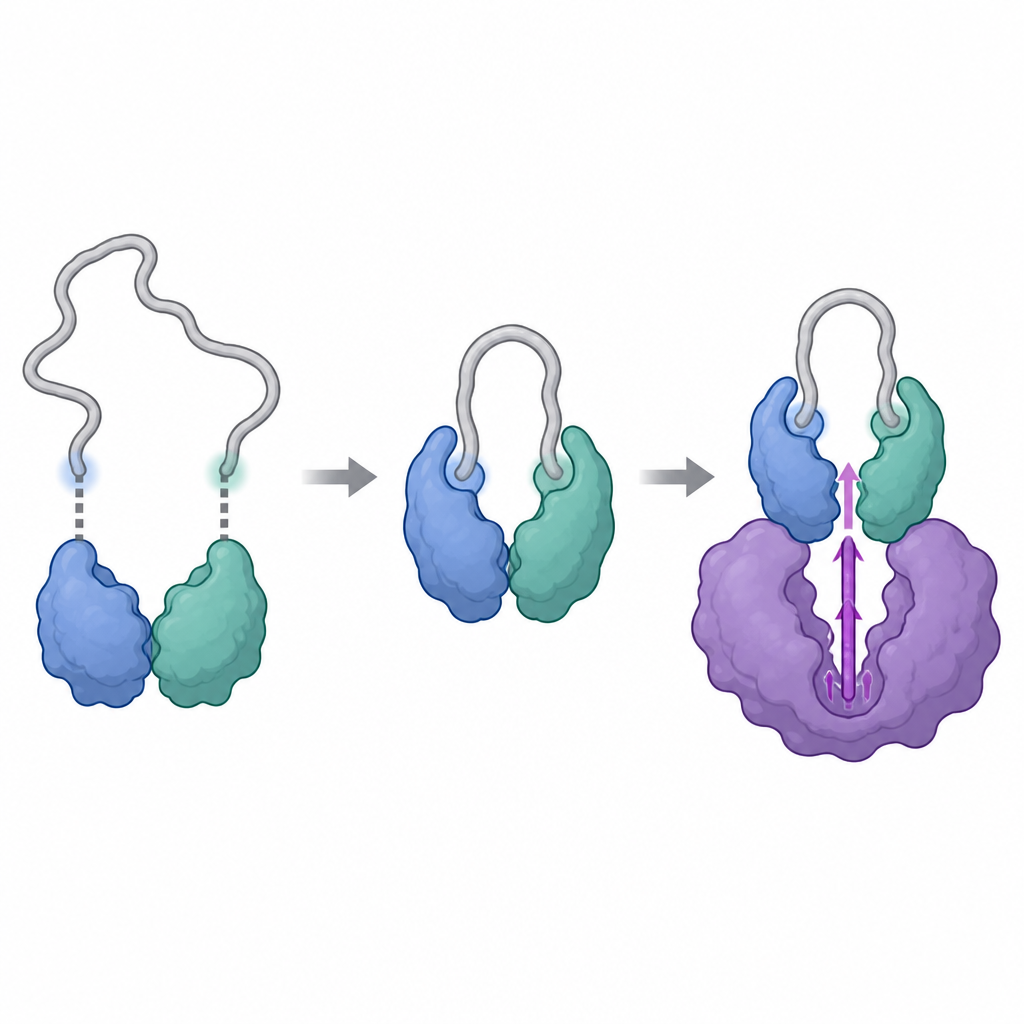
The researchers used powerful structural tools, including cryo-electron microscopy and a technique that tracks how hydrogen atoms in a protein swap with heavy water, to visualize the complexes formed by cyclin Y and 14-3-3, with and without CDK16. They found that cyclin Y carries two specific spots that can each latch onto one half of the 14-3-3 pair, like two buttons fastening into a clasp. When both spots are phosphorylated and engaged, 14-3-3 grips cyclin Y and subtly reshapes a crucial surface region. This reshaping exposes and stiffens the area that will later interact with CDK16, turning cyclin Y into a more suitable docking partner than it would be on its own.
How CDK16 is switched on
Once cyclin Y is properly held by 14-3-3, CDK16 can bind and complete the assembly. The structural snapshots show that CDK16 embraces cyclin Y with two main contact zones: a central region that resembles the typical docking site seen in other cyclin-dependent enzymes, and an extra, flexible tail at the front of CDK16 that is unique to this subfamily. When CDK16 binds, this floppy tail folds and nestles against cyclin Y, helping lock the complex together. At the same time, the activation loop of CDK16, a mobile segment near its chemical center, becomes ordered and held in an "on" position through direct touches from both cyclin Y and the end of the 14-3-3 pair. Mutating key amino acids at these interfaces weakens the complex and sharply reduces CDK16’s activity, confirming that these structural contacts are essential for turning the enzyme on.
Fine-tuning the control switch
The study also reveals how CDK16 itself can be tuned by additional phosphate tags on its own front-end tail. In earlier work, certain sites in this tail were known to affect how well cyclin Y binds. The new structural and dynamic data suggest that adding or removing these tags can alter how the tail folds and whether it can engage with the cyclin Y and 14-3-3 complex. In some cases, the tags may even recruit 14-3-3 directly to CDK16, blocking access to cyclin Y or shielding those regulatory sites from being reset. In this way, cells appear to use a layered control scheme in which both cyclin Y and CDK16 must carry the right phosphate marks before a fully active complex can form.
What this means for health and disease
By laying out the three-way embrace between CDK16, cyclin Y, and 14-3-3 in molecular detail, this work explains why CDK16 refuses to turn on without its helpers and how their interaction nudges the enzyme into its active shape. For non-specialists, the takeaway is that CDK16 is not a lone switch but part of a carefully wired safety circuit that depends on multiple protein contacts and chemical tags. Because CDK16 contributes to cancer cell survival and resistance to treatment, the newly revealed contact surfaces offer specific targets where future drugs might wedge in and disrupt the complex, selectively dimming CDK16’s activity while leaving related enzymes less affected.
Citation: Kohoutova, K., Kosek, D., Brzezina, A. et al. Structural basis of the cyclin Y/14-3-3 protein-mediated activation of CDK16. Nat Commun 17, 4262 (2026). https://doi.org/10.1038/s41467-026-70778-5
Keywords: CDK16, cyclin Y, 14-3-3 proteins, protein complexes, cancer signaling