Clear Sky Science · en
IVNS1ABP mutation drives cellular senescence in newly identified progeroid neuropathy
When Nerves Grow Old Too Soon
Some children show signs of aging long before their time, developing gray hair, fragile skin, and difficulty walking while still in school. This study investigates one such newly identified condition, tracing it from a single faulty gene to aging brain cells, and offering a window into how our nervous system may grow old in all of us.
A New Kind of Early Aging Disorder
The researchers describe a family in which several adolescents developed a rare “progeroid neuropathy.” These young people had mottled skin, premature graying of hair, walking problems, and intellectual disability, along with thinning of key brain structures. By scanning their DNA, scientists found that the affected siblings all carried two copies of a rare change in a gene called IVNS1ABP. This gene was previously known mainly for its role in how cells handle flu virus, not for aging or nerve disease. The team set out to uncover how this mutation could make cells and the nervous system appear old before their time. 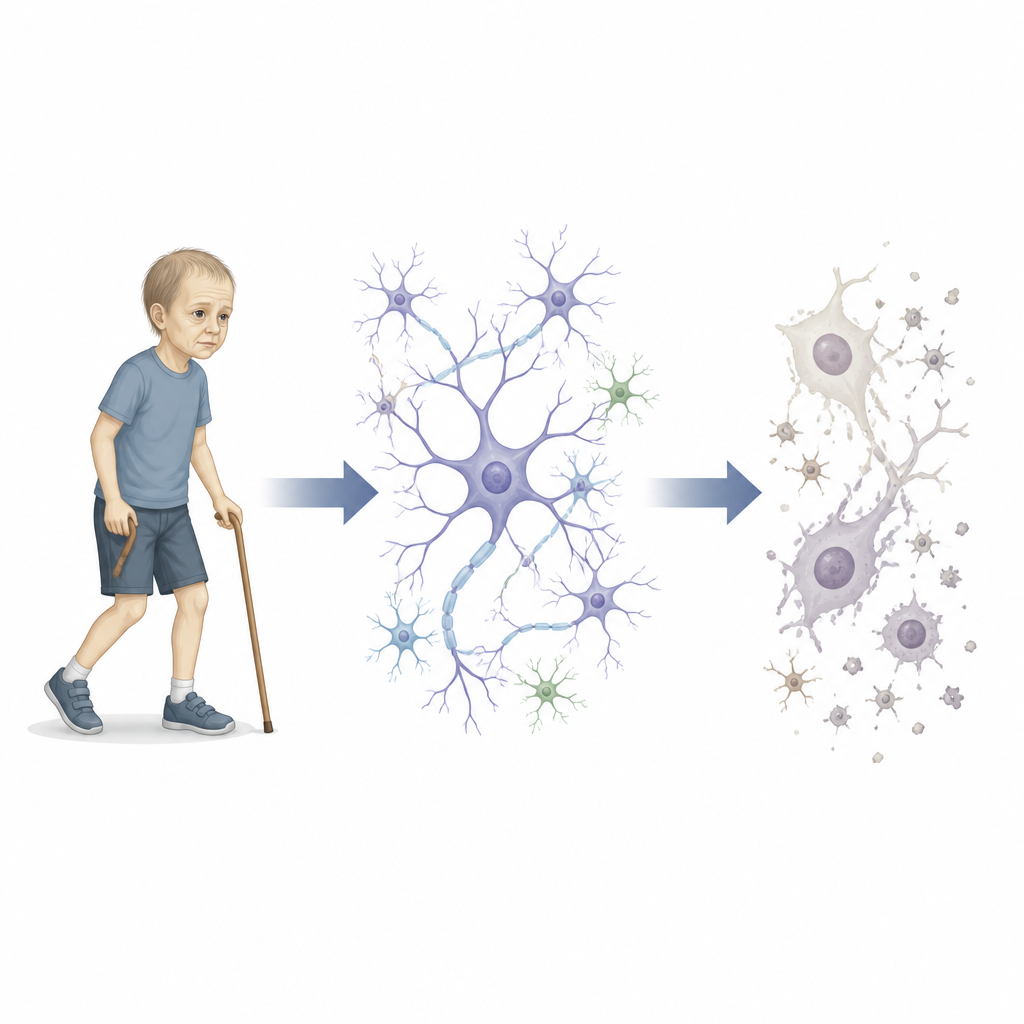
Recreating the Disease in a Dish
To study the disorder safely and in detail, the team reprogrammed skin cells from patients and unaffected relatives into induced pluripotent stem cells, then guided these into neural progenitor cells, the precursors of brain cells. They also used gene editing to create “isogenic” pairs of cells that were genetically identical except for the single IVNS1ABP mutation, and even made complete gene knockouts. Across patient cells, edited stem cells, and their neural descendants, the same pattern appeared: cells divided more slowly, spent longer in each phase of the cell cycle, and frequently failed at splitting cleanly into two daughters.
Cells Showing Signs of Old Age
The faulty cells bore classic hallmarks of cellular aging. They accumulated DNA damage, as shown by molecular flags that mark broken or stressed chromosomes. Protective chromatin marks that help keep DNA organized were reduced, while the nucleus itself often grew larger. Many cells turned positive for a widely used aging stain and switched on p16 and related genes that enforce permanent growth arrest. When grown as three-dimensional “mini-brains” called cerebral organoids, mutant neural progenitors exited the cell cycle early and turned into neurons ahead of schedule. These organoids were smaller and had thinner zones of dividing precursor cells but thicker layers of more mature neurons, suggesting that the pool of “seed” cells that should build the brain was being used up too fast.
How a Cytoskeletal Helper Goes Wrong
Digging deeper, the scientists asked which proteins normally partner with IVNS1ABP. Using pull-down experiments and mass spectrometry, they found that the healthy protein strongly interacts with actin and a group of actin-associated proteins that help build and remodel the cell’s internal scaffolding. In contrast, the mutant form bound these partners poorly. In test-tube assays, normal IVNS1ABP promoted the assembly of actin filaments efficiently, whereas the mutant version was much weaker and failed to bind well even at higher amounts. Inside mutant neural progenitors, the balance tipped away from sturdy filamentous actin toward its loose, single-unit form, and the ring of actin that normally tightens to pinch a dividing cell in two became thin and uneven. 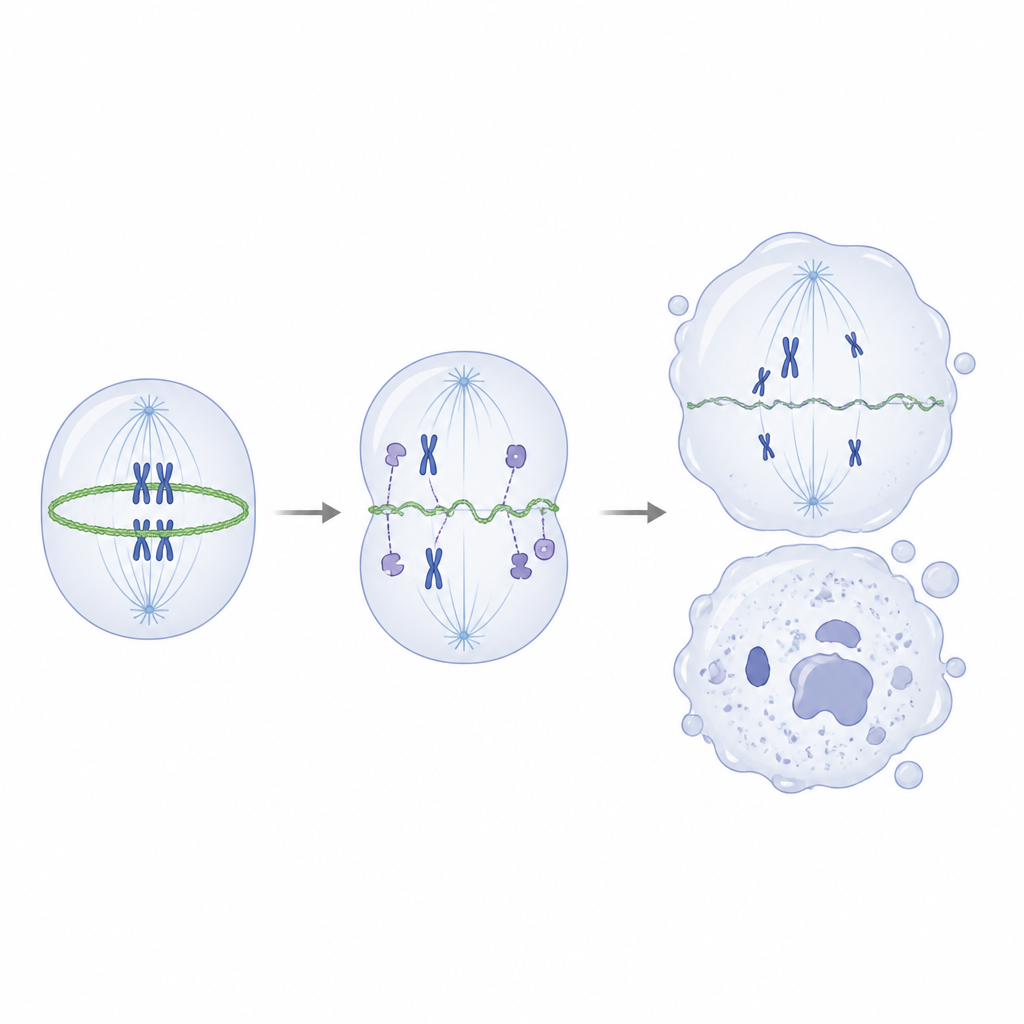
From Broken Cell Splits to Early Nerve Aging
This faulty scaffolding during division led to misaligned chromosomes, abnormal spindles, and uneven cell splits, which in turn produced DNA damage and pushed cells into a senescent, non-dividing state. Importantly, when the researchers used a low dose of a drug that promotes actin assembly, they could partially restore the actin balance, improve the shape of the contractile ring, reduce division errors, and lessen markers of DNA damage and cellular aging, even though the gene mutation remained. In cerebral organoids, the same treatment partly rescued growth and the pool of neural progenitors. For a layperson, the conclusion is that this rare childhood condition stems from a single gene that normally helps manage the cell’s inner scaffolding during division. When that helper fails, developing nerve cells divide poorly, burn out early, and age too soon, leading to fewer and weaker neurons and, ultimately, to both early aging features and problems with movement and thinking.
Citation: Yuan, F., Tan, Y.S., Wang, H. et al. IVNS1ABP mutation drives cellular senescence in newly identified progeroid neuropathy. Nat Commun 17, 4159 (2026). https://doi.org/10.1038/s41467-026-70756-x
Keywords: cellular senescence, progeroid neuropathy, IVNS1ABP, actin dynamics, neural progenitor cells