Clear Sky Science · en
Cross-ketone deacylative coupling via oxidative SH2 homolytic substitution
Turning common chemicals into flexible building blocks
Chemists rely on a small set of simple ingredients to build the complex molecules found in medicines and materials. This study shows how to coax one of the most common ingredients, ketones, into forming new connections that were previously very hard to achieve, opening fresh routes to design drug-like molecules more quickly and precisely.
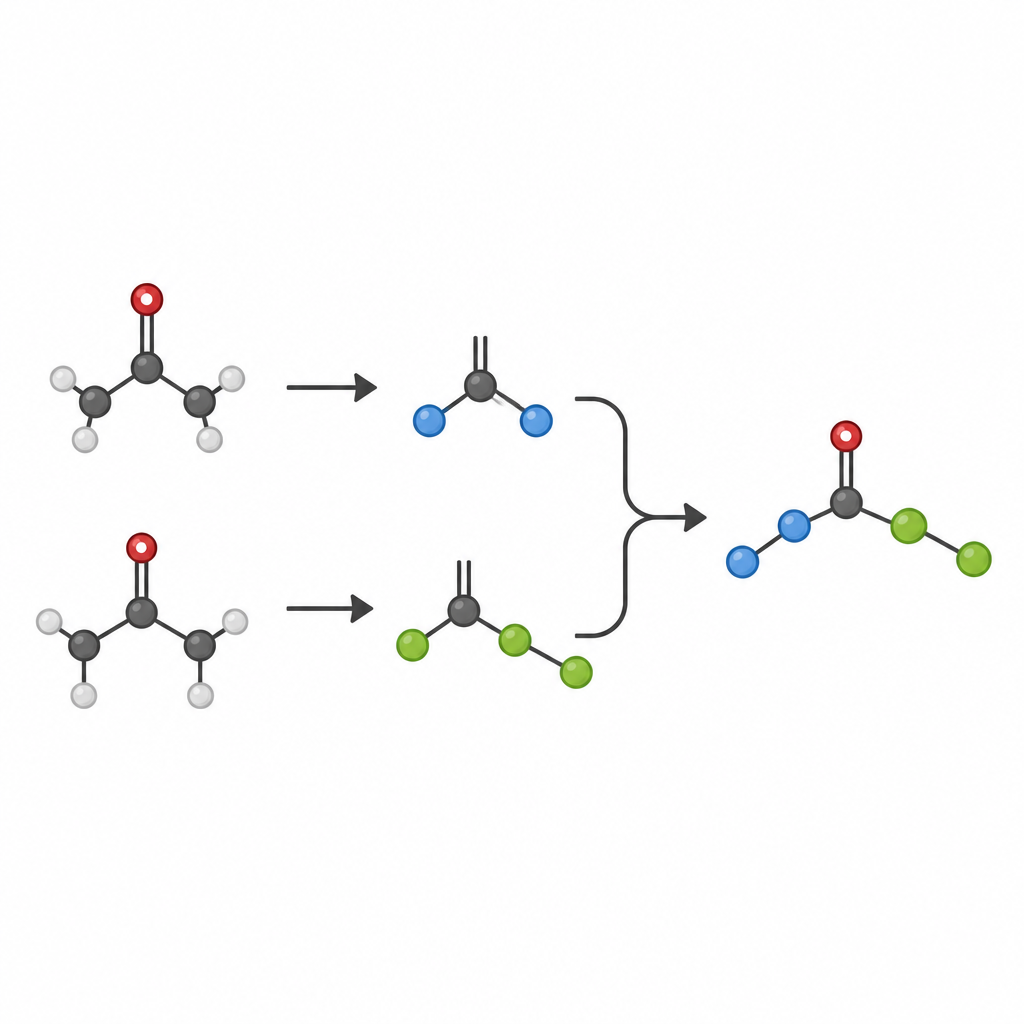
Why everyday ketones matter
Ketones are workhorse molecules in organic chemistry, prized because they are easy to make, store, and modify. Classic reactions such as the aldol reaction and McMurry coupling already turn ketones into more complex products, but these methods often need large amounts of metal reagents and struggle to join two different partners in a controlled way. In particular, forming sturdy links between two saturated carbon atoms, the so-called C–C single bonds in crowded environments, has remained a major challenge, even though such crowded carbon centers are central features in many modern medicines.
A new way to make two radicals work together
The authors set out to get two different ketones to talk to each other in a clean and selective manner. Instead of activating the usual carbonyl portion of the ketone, they focus on breaking a nearby carbon–carbon bond to release small, short-lived fragments known as radicals. These radicals can, in principle, recombine in many ways, including unwanted self-pairing or side reactions. To tame this chaos, the team uses a strategy called SH2, a form of homolytic substitution in which a metal complex briefly holds one radical and then is attacked by a second radical from the outside. This outer-sphere pathway helps guide which two pieces actually join.
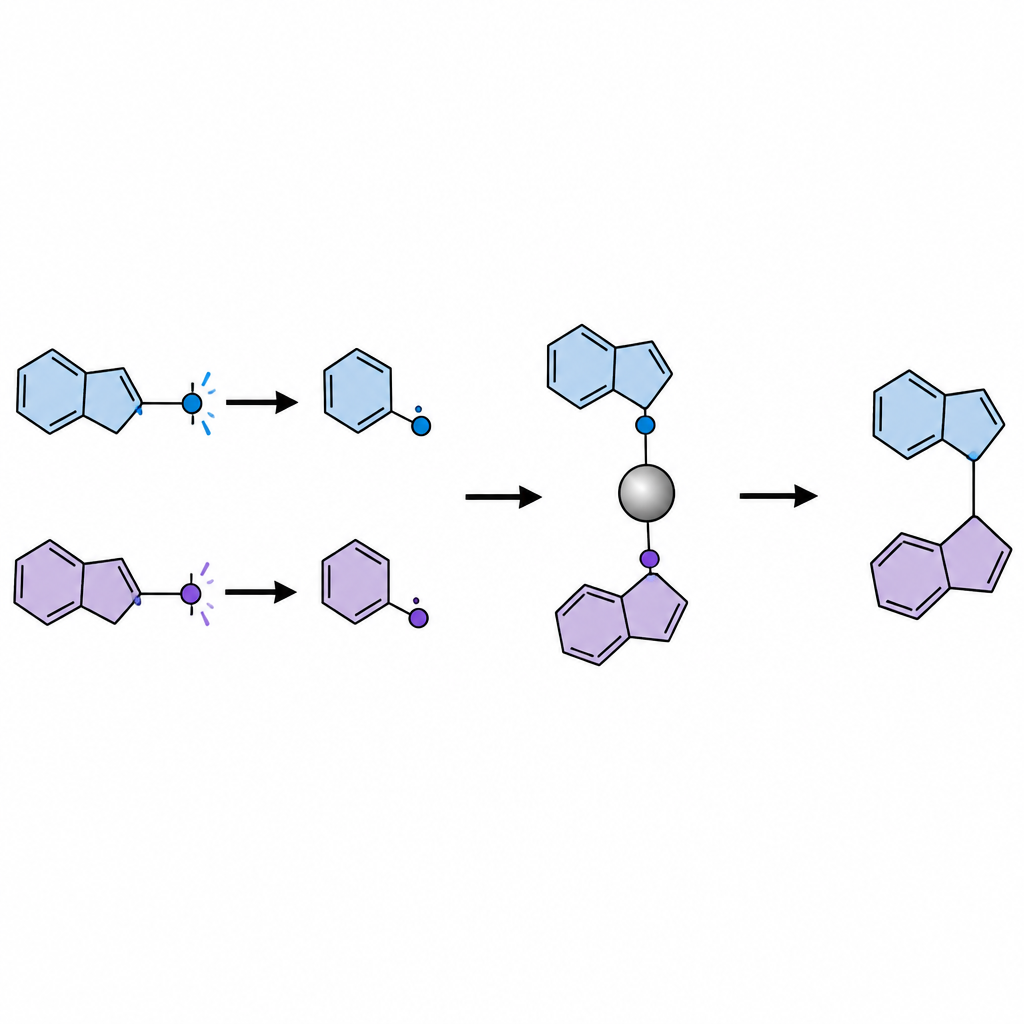
Masking ketones to control their behavior
Directly shining light on ketones often generates both useful and harmful radical fragments. To bypass this problem, the researchers convert each ketone into a bench-stable “masked” form called a dihydroquinazolinone in one simple step before the main reaction. Under blue light, an organic photocatalyst oxidizes these masked ketones, causing them to snap apart and release two distinct radicals: a smaller, less hindered primary fragment and a bulkier secondary or tertiary fragment. A nickel catalyst selectively grabs the smaller radical to form a persistent nickel–carbon complex, while the bulkier radical stays free in solution. Because the nickel–primary complex forms a stronger bond than the nickel–tertiary complex would, the system naturally sorts the radicals into their different roles.
Building crowded centers for drug-like molecules
Once assembled, the nickel–primary complex is attacked by the bulkier radical in an SH2 step, forging a new carbon–carbon bond and regenerating the nickel catalyst. Using this approach, the team constructs a wide range of products that contain a crowded, four-way carbon center next to a nitrogen, oxygen, or sulfur atom. These include β-quaternary amines, β-amino alcohols, β-diamines, β-aminothiols, and related ethers, all motifs that are common in bioactive compounds. The reaction tolerates many functional groups, such as alkenes, esters, halides, and ring systems, and operates under mild conditions with visible light. The authors also show that the method can be applied late in the synthesis of known drug molecules, gently modifying their structures without disturbing other sensitive parts.
Peeking under the hood of the reaction
To understand how the process works, the researchers trap the fleeting radicals, study how the light-absorbing catalyst is quenched, and compute the relative speeds and energies of key steps. Their evidence points to a mechanism where both radicals arise from oxidation of the masked ketones rather than from the peroxide oxidant, and where nickel remains in a higher oxidation state, avoiding the typical inner-sphere cross-coupling pathway that would react with aryl halides. Calculations show that formation of the nickel–primary complex is energetically preferred, and that both radicals are produced at similar rates, conditions that suppress unwanted self-coupling and favor the desired cross-pairing.
What this means for future molecule making
In simple terms, this work teaches ketones a new trick: how to break apart in a controlled way so that the right fragments recombine into new, more complex structures. By combining visible light, a nickel catalyst, and cleverly masked ketones, the authors create a general and flexible route to crowded carbon centers that are highly valued in drug discovery. This strategy adds a powerful tool to the chemist’s toolbox for editing and diversifying molecules, particularly at late stages where small changes can have large effects on biological activity.
Citation: Yang, JX., Zhang, MY., Wen, Q. et al. Cross-ketone deacylative coupling via oxidative SH2 homolytic substitution. Nat Commun 17, 4248 (2026). https://doi.org/10.1038/s41467-026-70619-5
Keywords: ketone coupling, radical chemistry, nickel catalysis, photoredox, quaternary carbon