Clear Sky Science · en
Multimodal imaging reveals a lysosomal drug reservoir that drives heterogeneous distribution of PARP inhibitors
Why this research matters for cancer treatment
Many modern cancer drugs are designed with exquisite precision, yet they still fail some patients. One underexplored reason is that drugs may not reach the same concentration inside every cancer cell, even within a single tumor. This paper investigates how three widely used ovarian cancer drugs, called PARP inhibitors, spread through tumors and individual cells, and reveals that tiny recycling compartments inside cells, known as lysosomes, can act as a hidden drug reservoir that shapes whether treatment succeeds or fails.
Uneven drug levels inside the same tumor
The researchers focused on high-grade serous ovarian carcinoma, an aggressive and highly variable form of ovarian cancer. Using fresh tumor samples removed during surgery, they kept thin slices of patient tissue alive in the lab and soaked them with three clinically used PARP inhibitor drugs. With advanced mass spectrometry imaging, they could see where the drugs accumulated across each slice. Even though every region was exposed to the same dose, drug distribution was strikingly uneven – some areas were drug "hotspots" while nearby zones had very little. This patchiness appeared between different patients, between different tumors in the same patient, and even within a single tumor sample, suggesting that cell-intrinsic properties strongly influence how much drug each area absorbs. 
Drug-rich regions respond better but reveal a cellular clue
To find out what distinguished hotspots from low-drug regions, the team used spatial transcriptomics, a technology that reads gene activity while preserving tissue layout. Comparing regions with high versus low levels of two PARP inhibitors (rucaparib and niraparib), they found that drug-rich areas showed stronger signatures of DNA damage, cell cycle arrest, and programmed cell death – exactly what these drugs are meant to trigger. At the same time, these high-drug regions consistently expressed more genes linked to lysosomes, the acidic compartments that digest cellular waste and recycle components. Multiple lysosome-associated genes showed a tight correlation with local drug concentration, hinting that differences in lysosomal content might help determine how much drug a cell can hold and how strongly it responds.
Single cells show non-genetic differences in drug uptake
To zoom in further, the scientists turned to ovarian cancer cell lines grown in culture. One of the drugs, rucaparib, glows under the microscope, allowing them to measure its level inside thousands of individual cells. They saw up to a fivefold difference in intracellular drug signal between the highest- and lowest-accumulating cells, even though all were exposed to the same concentration. Sorting out the brightest and dimmest cells and growing them for several weeks showed that these differences were not fixed by genetics: over time, the populations reverted to a similar distribution. This suggested that non-genetic factors – such as variable organelle content or metabolic state – underlie the diversity in drug uptake.
How lysosomes become a useful drug reservoir
Proteomic analyses of cells with high versus low rucaparib levels showed that the high-drug cells were enriched in lysosomal proteins. Imaging confirmed that rucaparib rapidly entered cells and then accumulated in punctate structures that overlapped with lysosome-specific dyes. Cells with more lysosomal signal had more intracellular drug, and manipulating lysosomes changed drug levels: boosting lysosome formation or acidification increased rucaparib accumulation, while neutralizing lysosomal acidity reduced it. Importantly, the drug did not remain harmlessly trapped. Lysosome-rich cells had more DNA damage and slower growth after treatment, and disrupting lysosomal function lowered nuclear drug levels and weakened the drug’s long-term killing effect after a short exposure pulse. 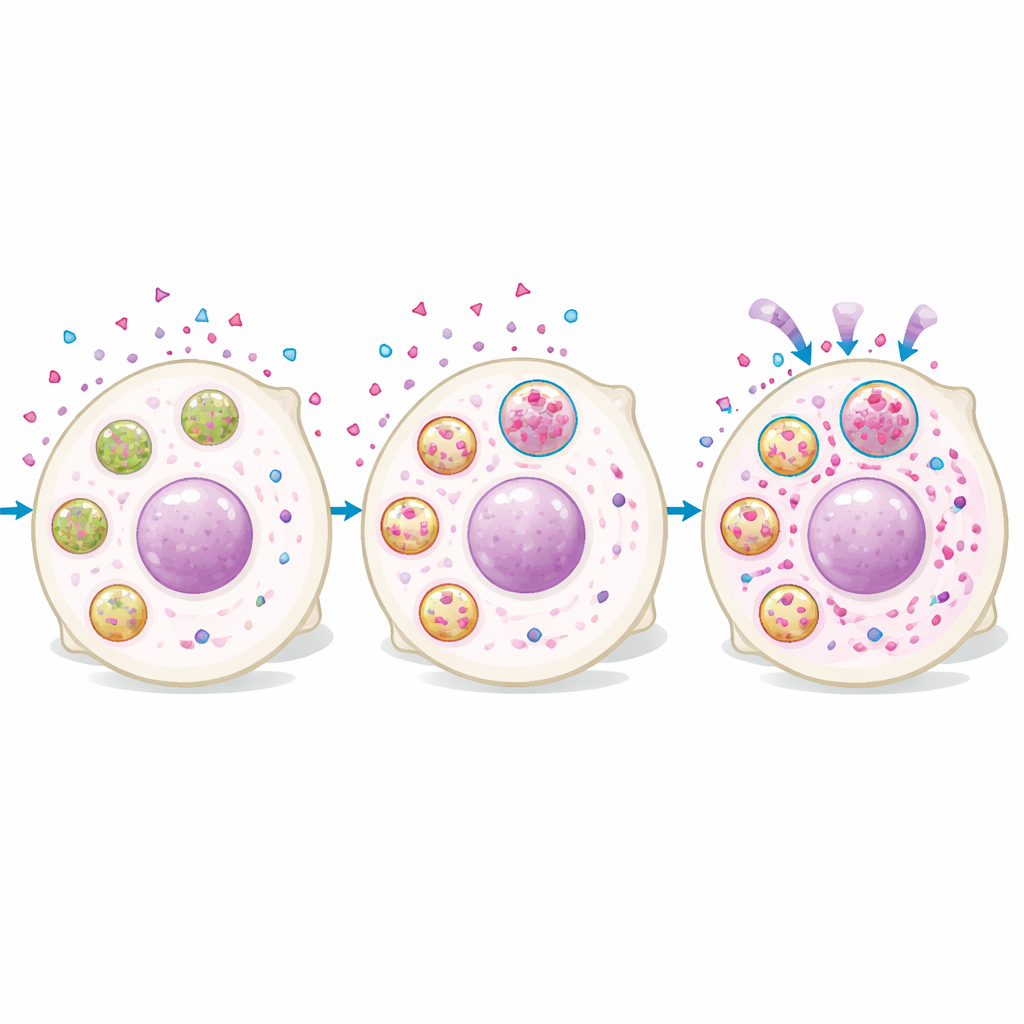
Why only some PARP drugs behave this way
Not all PARP inhibitors behaved alike. Rucaparib and niraparib are weak bases, meaning they gain charge and become less able to cross membranes in the acidic environment of lysosomes, favoring their buildup there. Another widely used PARP inhibitor, olaparib, has very different chemical properties and did not show this kind of pH-dependent lysosomal accumulation. When the team measured drug content by mass spectrometry, neutralizing lysosomes reduced the intracellular levels of rucaparib and niraparib but left olaparib unchanged. Likewise, altering lysosomal content only changed the DNA damage response to the weak-base drugs, not to olaparib. This implies that patients whose tumors naturally have more lysosomes could respond better to some PARP inhibitors than others.
What this means for patients and future therapies
This study reveals that lysosomes in cancer cells can act not as a dead-end trap, but as a reservoir that stores certain drugs and slowly feeds them back to where they act, in this case the nucleus. Because tumors are highly heterogeneous, regions with many lysosomes may accumulate more of specific PARP inhibitors, leading to better local killing of cancer cells, while nearby regions with fewer lysosomes may be underdosed despite receiving the same treatment. In practical terms, this work suggests that matching the chemical properties of a drug to the cellular makeup of a patient’s tumor – such as lysosomal content – could improve responses and overcome some cases of apparent drug resistance. It also points toward future diagnostic strategies where tiny tumor samples are tested ex vivo to identify which drug will distribute and work best for each individual.
Citation: R. Moncayo, C., Restuadi, R., Zhang, G. et al. Multimodal imaging reveals a lysosomal drug reservoir that drives heterogeneous distribution of PARP inhibitors. Nat Commun 17, 4086 (2026). https://doi.org/10.1038/s41467-026-70558-1
Keywords: ovarian cancer, PARP inhibitors, lysosomes, drug resistance, tumor heterogeneity