Clear Sky Science · en
Butyrate extends health and lifespan in mice with mitochondrial deficiency
Why the Gut Matters for Failing Cell Engines
Mitochondrial diseases are rare disorders in which the tiny “power plants” inside our cells begin to fail, causing a cascade of problems across many organs and often shortening life. Treatments today are mostly limited to managing symptoms. This study in mice uncovers an unexpected ally living in the gut: certain bacteria that produce a fatty acid called butyrate. The work shows that restoring this gut-derived molecule can shore up a leaky intestine, ease multiple age‑like diseases, and significantly extend lifespan in animals with severe mitochondrial defects.
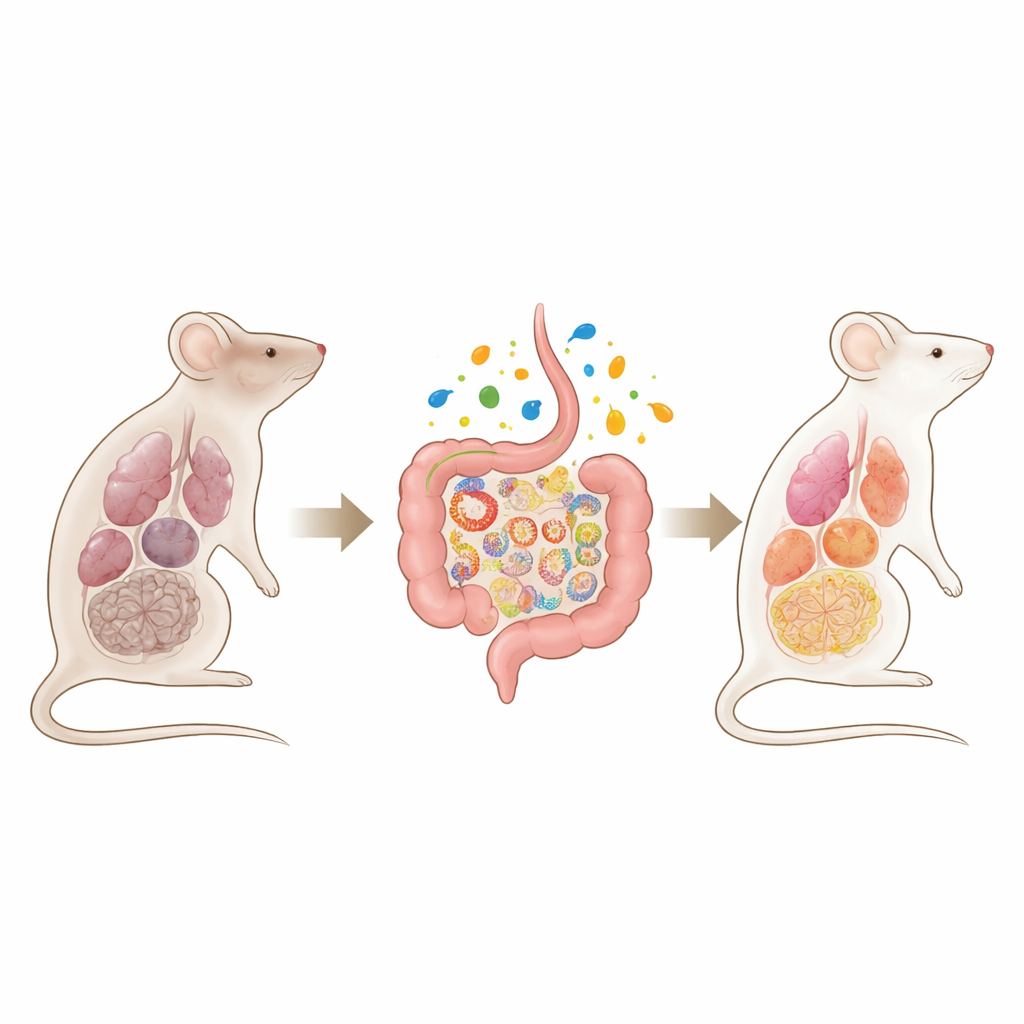
When Cell Power Plants Break Down
The researchers first engineered a mouse in which a key mitochondrial regulator, TFAM, can be switched off throughout the body in adulthood. Once this molecular “guardian” of mitochondrial DNA was removed, the animals rapidly developed a syndrome resembling accelerated aging: loss of fat and muscle, weak grip, poor blood sugar control, lung scarring, heart and vessel abnormalities, kidney failure, anemia, and signs of brain degeneration including movement problems and impaired nesting behavior. Many organs showed inflammation and molecular markers of cellular senescence, and the mice died prematurely, indicating that widespread mitochondrial failure alone can drive multi‑organ breakdown.
A Leaky Gut and a Disturbed Microbial Community
Because mitochondria are crucial for the health of intestinal cells, the team examined the animals’ guts. They found shorter intestinal structures, fewer dividing cells, and a thinner protective mucus layer despite normal numbers of mucus‑producing goblet cells. More of these cells were “stuck” holding mucus instead of releasing it. Genes that help seal the spaces between cells and produce antimicrobial defenses were switched down, and the intestine became more permeable: fluorescent tracer leaked into the blood, and a blood marker of bacterial products rose sharply. At the same time, the mix of gut bacteria shifted. Microbes that usually thrive on dietary carbohydrates and make health‑promoting short‑chain fatty acids declined, while other groups expanded. Chemical analysis confirmed that levels of all major short‑chain fatty acids in feces, and several in blood, were markedly reduced.
A Common Pattern in Another Mitochondrial Disease Model
To see if this gut disruption was unique to one genetic trick, the scientists turned to a second mouse strain that accumulates random mutations in mitochondrial DNA as it ages and develops premature multimorbidity. These animals also had a leakier intestinal barrier, reduced expression of junction and antimicrobial genes, and a simpler, imbalanced community of gut microbes. In particular, bacteria that specialize in producing butyrate were depleted, and fecal butyrate levels were selectively lower, while other short‑chain fatty acids were relatively spared. This pointed to loss of butyrate‑producing partners as a shared hallmark of mitochondrial decline.
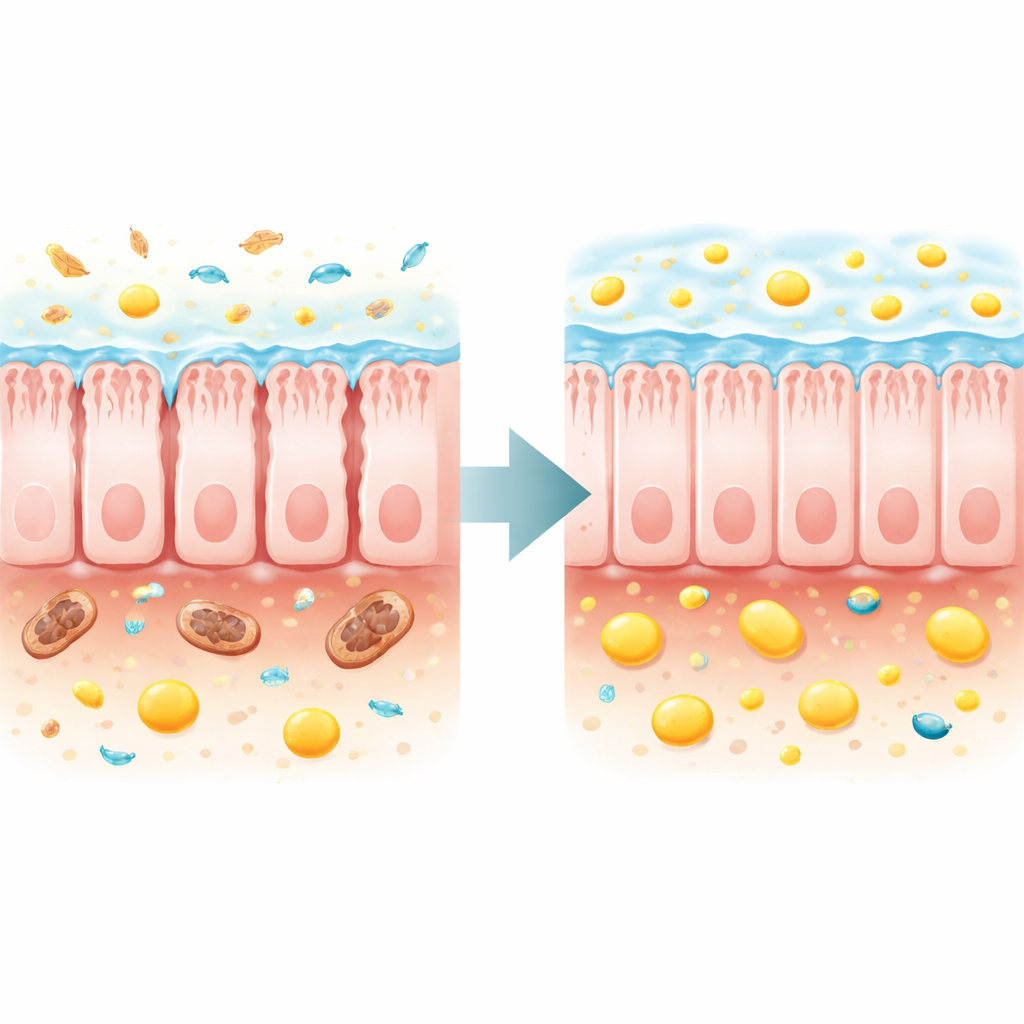
Replacing Lost Microbial Help and Rebuilding the Barrier
The authors then asked whether repairing the gut ecosystem or directly restoring butyrate could improve whole‑body health. Transferring fecal microbes from healthy donors into the TFAM‑deficient mice modestly slowed weight loss, improved muscle strength and constipation, raised fecal butyrate and related fatty acids, and extended maximum lifespan by about 70% compared with mice that received diseased microbiota. A more targeted approach used a dietary supplement, tributyrin, which is converted into butyrate in the gut. This treatment boosted fecal butyrate, delayed wasting, strengthened muscles, normalized fasting blood sugar, and reduced kidney damage. Remarkably, it extended median lifespan by roughly one quarter and maximum lifespan by more than three quarters in the sick mice.
How a Gut Molecule Talks to Genes
Butyrate is known not only as a fuel for intestinal cells but also as a chemical signal that can influence which genes are turned on or off. In the diseased mice, the researchers found that specific chemical tags on histone proteins—small acyl groups added to histone H3 at defined positions—were greatly reduced in intestinal tissue. These tags help open up DNA around genes that preserve the gut’s barrier and handle stress. Depleting normal mice of their microbiota with antibiotics produced a similar loss of these marks, linking them to microbial metabolites. Feeding the TFAM‑deficient mice tributyrin restored these histone modifications and shifted intestinal gene activity back toward a healthier pattern, including genes involved in cell‑to‑cell junctions, the scaffolding of the tissue, and responses to oxidative damage.
A New Way to Support Failing Mitochondria
In simple terms, this study suggests that when mitochondria falter, the intestine becomes leaky and its friendly microbes change, starving the body of butyrate. That loss, in turn, weakens the gut barrier and disturbs gene regulation, fueling systemic disease. Rebuilding the partnership between host and microbiota—either by transplanting a healthier microbial community or by supplying a butyrate‑releasing supplement—can ease many symptoms and extend life in mice with severe mitochondrial dysfunction. While much work is needed before such strategies can be applied to people, the findings highlight the gut and its microbial chemistry as promising new levers for treating disorders rooted in broken cellular power plants.
Citation: Gabandé-Rodríguez, E., Gómez de las Heras, M.M., Ramírez-Ruiz de Erenchun, P. et al. Butyrate extends health and lifespan in mice with mitochondrial deficiency. Nat Commun 17, 3909 (2026). https://doi.org/10.1038/s41467-026-70547-4
Keywords: mitochondrial disease, gut microbiome, butyrate, intestinal barrier, short-chain fatty acids