Clear Sky Science · en
Stereodivergent synthesis of chiral amines bearing vicinal stereocenters via hydroamination of trisubstituted alkenes
Why the Shape of a Molecule Matters
Many of today’s medicines owe their power to tiny differences in three-dimensional shape. Two molecules built from the same atoms can behave like left and right hands: almost identical, yet one may heal while the other harms. This study introduces a new way to sculpt such shapes in a key family of molecules called amines, which appear throughout drugs, natural products, and agrochemicals. By teaching simple, flat building blocks to fold into precise 3D arrangements, the work opens a more reliable route to medicines that are both more effective and safer.
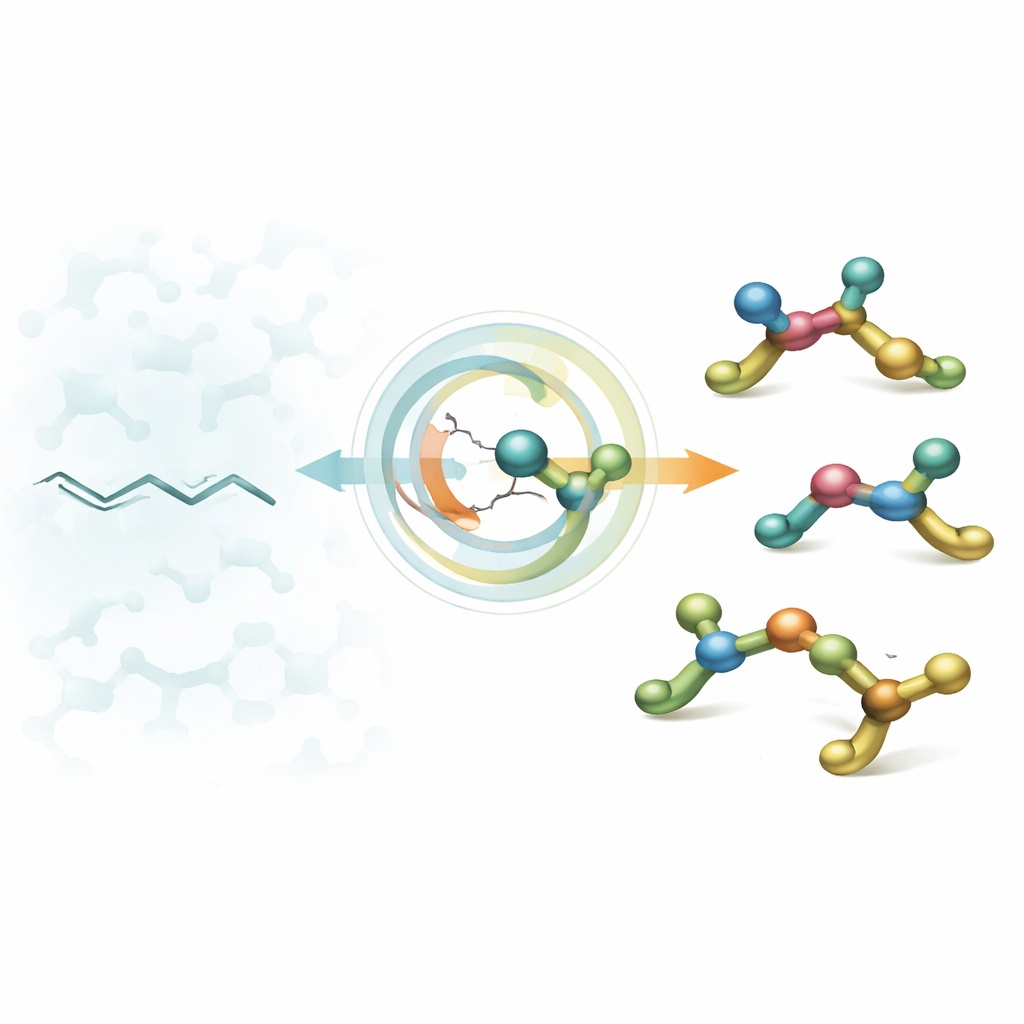
From Flat Chains to 3D Building Blocks
Drug designers often start from aliphatic amines – small nitrogen‑containing fragments that plug into biological targets. For decades, chemists have become adept at making amines with a single "twist point" (a stereocenter), yet real drugs frequently contain several such points right next to each other. These tightly packed chiral centers can dramatically improve how a molecule fits into an enzyme or receptor, but they are notoriously hard to build in a controlled way. Existing methods usually favor just one layout, leaving chemists without a simple, flexible strategy to reach all of the shapes they might want to test.
A Single Reaction That Can Aim Four Ways
The authors tackle this problem by converting readily available trisubstituted alkenes – essentially flat carbon chains with a crowded double bond – into 3D amines bearing two neighboring stereocenters. Their key move is a nickel‑catalyzed hydroamination: a nickel hydride species first adds across the double bond, then an amine fragment is attached, all in a carefully choreographed sequence. The clever twist is that the outcome depends in a predictable way on just two switches: whether the starting double bond is in an E or Z arrangement, and whether the nickel’s chiral ligand is its R or S version. By flipping these two settings, the same reaction setup can deliver any one of the four possible stereoisomers on demand.
Designed for Real‑World Molecules
Beyond the conceptual advance, the chemistry proves robust and practical. The reaction tolerates a wide range of amine partners, from simple secondary amines to more decorated variants that carry esters, carbamates, sulfones, and even boronic esters often used later in drug modification. Likewise, many different trisubstituted alkenes participate smoothly, including those derived from natural products and existing drug molecules. In most cases, the process delivers the desired amines in high yields and with excellent control over both which mirror image is formed and how the two neighboring stereocenters are arranged.
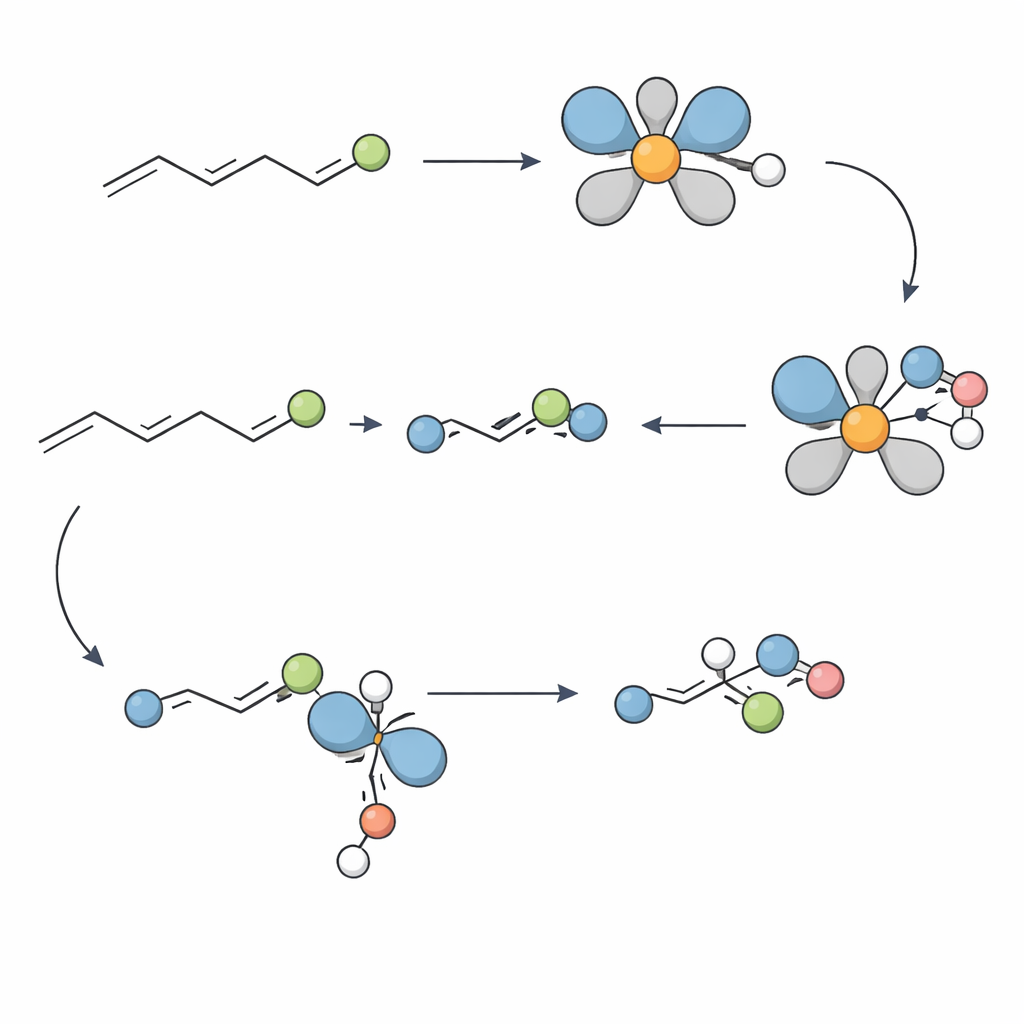
Probing How the Catalyst Does Its Job
To understand why the method works so well, the team carried out mechanistic experiments. Labeling studies showed that the hydride in the nickel complex adds to a specific position on the alkene in a single, non‑reversible step, setting up the two neighboring stereo‑centers with precise geometry. Tests with potential radical traps suggested that free‑radical pathways are unlikely. Instead, evidence from high‑resolution mass spectrometry and a crystallographically characterized nickel complex points to a cycle that shuttles between nickel(II) and nickel(I) species. A key step appears to be a hand‑off (transmetalation) between these two oxidation states, which ultimately leads to formation of the C–N bond that defines the amine product.
New Possibilities for Future Medicines
In practical demonstrations, the authors applied their strategy to modify advanced drug‑like molecules and to build stereo‑defined analogues of existing medicines that contain an N‑(2‑ethylamino)amide motif. They also showed that the reaction can be scaled up and that the resulting products can be further transformed without losing their carefully set stereochemistry. For non‑specialists, the key message is that chemists now have a programmable way to dial in not just one, but all four possible 3D arrangements around two neighboring chiral centers in valuable amine structures. This capability should greatly expand the menu of candidate molecules for drug discovery, enabling more systematic exploration of shape–activity relationships and, ultimately, better‑designed therapies.
Citation: Bai, H., Li, M., Wang, X. et al. Stereodivergent synthesis of chiral amines bearing vicinal stereocenters via hydroamination of trisubstituted alkenes. Nat Commun 17, 3431 (2026). https://doi.org/10.1038/s41467-026-70294-6
Keywords: chiral amines, nickel catalysis, hydroamination, stereodivergent synthesis, drug discovery