Clear Sky Science · en
Energy-sensing molecule RORγ regulates cholesterol metabolism and immune signaling in diabetic kidney disease and aging
Why this research matters for people with diabetes and aging kidneys
Diabetic kidney disease and the natural aging of the kidneys are two of the main reasons people eventually need dialysis or a transplant. This study uncovers a previously unrecognized “energy-sensing switch” inside kidney tubule cells that helps keep cholesterol and inflammation in check. When this switch, a molecule called RORγ, shuts down, fat and inflammatory damage build up in the kidneys. When it is turned back on, the kidneys are protected. Understanding this control system points toward new ways to slow kidney damage in both diabetes and aging.
The hidden link between energy use, fat, and kidney injury
Kidney tubules handle much of the body’s cleanup work and use a lot of energy to do it. In both diabetes and aging, these cells accumulate excess lipids, especially cholesterol, and become inflamed and scarred. The authors examined kidney tissue from diabetic mice, aged mice, and people with diabetes. Across all three, they found that RORγ—a nuclear protein that helps cells sense energy and regulate genes—was consistently reduced, especially in tubule cells. Lower RORγ levels were associated with worse kidney function, more scarring, and higher markers of cell aging, suggesting that RORγ normally acts as a guardian of tubular health. 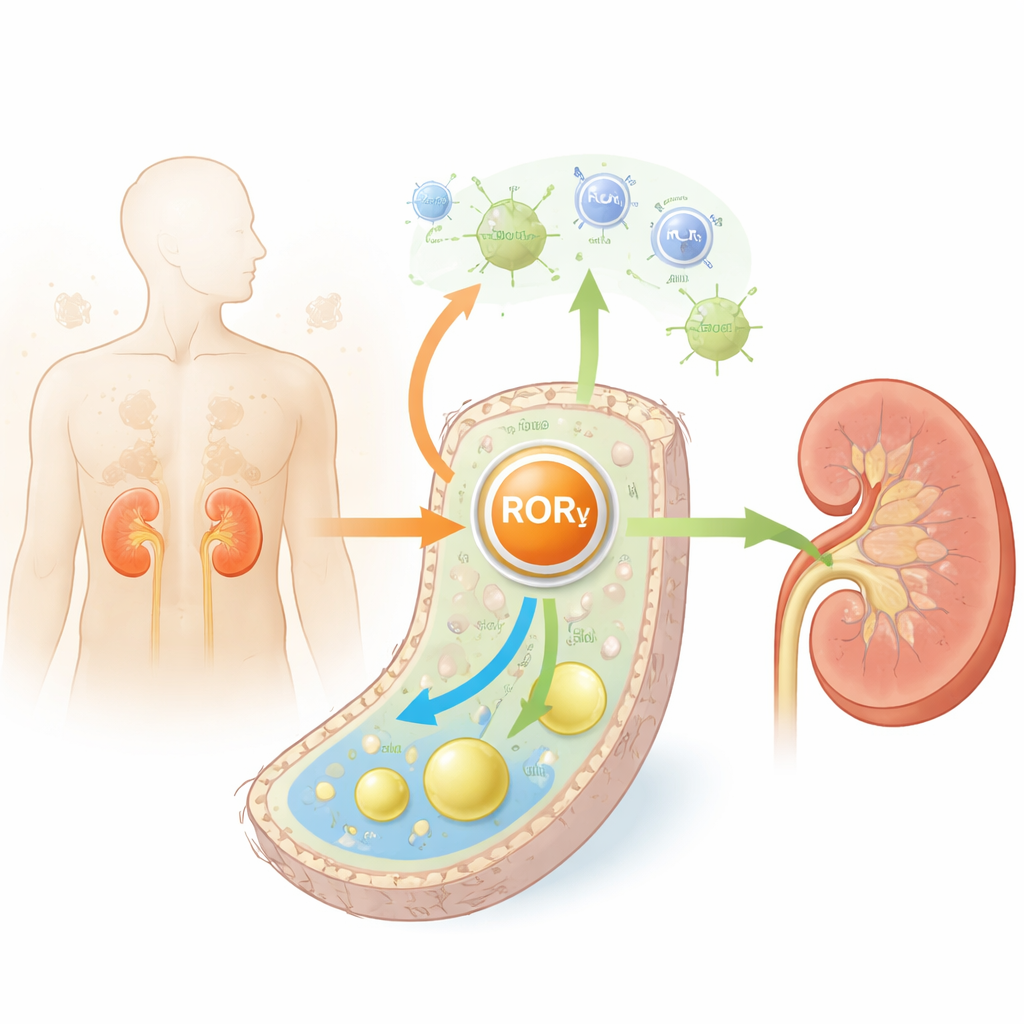
How RORγ keeps cholesterol production and immune alarms under control
The team discovered that RORγ protects tubule cells by stabilizing another protein, INSIG1, which sits in the membrane of an internal structure called the endoplasmic reticulum. INSIG1 normally acts as a gatekeeper, preventing a cholesterol-promoting factor (SREBP2) and an inflammation-triggering sensor (STING) from traveling to the cell’s shipping center, the Golgi apparatus, where they become fully active. In diabetes or aging, when RORγ is low, INSIG1 is rapidly tagged for destruction and broken down, the gate fails, and both cholesterol production and inflammatory signaling surge.
The two helper routes that RORγ uses to stabilize INSIG1
RORγ reinforces INSIG1 through two complementary helper systems. First, it switches on a protein called YOD1 that removes ubiquitin tags—molecular “dispose of me” signals—from INSIG1, slowing its degradation. Second, RORγ boosts the activity of an energy sensor enzyme, AMPK, by increasing levels of a scaffolding protein called CAB39 that assembles AMPK’s upstream activator. Active AMPK then adds a phosphate group to INSIG1 at a specific site, which weakens its interaction with an enzyme that would otherwise target it for destruction. Together, YOD1 and AMPK make INSIG1 last longer, keeping cholesterol synthesis and STING-driven immune activity under tight restraint. 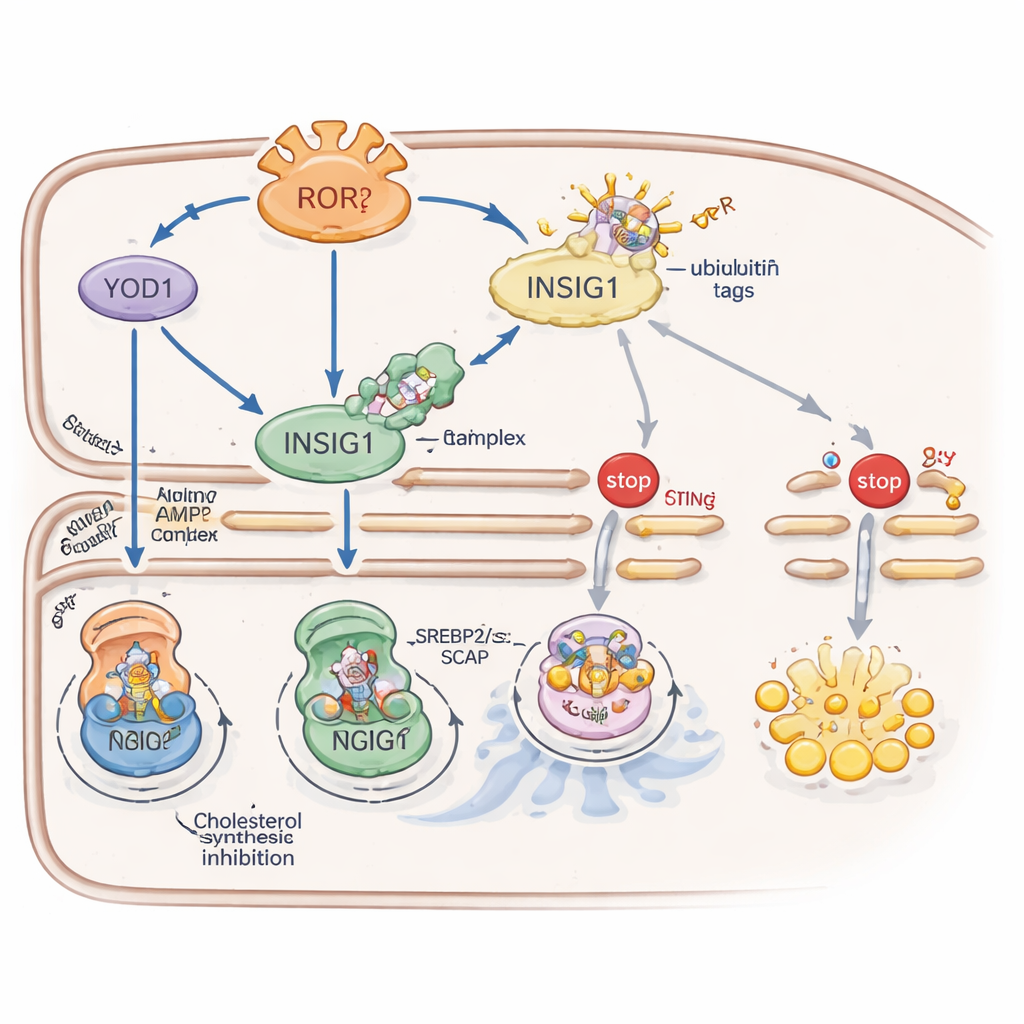
When the protective switch fails in diabetes and aging
In a diabetic environment—high sugar and fat, oxidative stress, and DNA damage—several mechanisms conspire to turn off RORγ. A DNA-binding protein called CTCF becomes more active and clamps down on the RORγ gene, lowering its production. At the same time, impaired AMPK signaling and reduced activity of another regulator, SIRT1, diminish RORγ’s movement into the nucleus and weaken its grip on target genes. In mice engineered to lack RORγ only in tubule cells, diabetes and aging caused much worse cholesterol buildup, inflammation, fibrosis, and markers of cellular senescence, confirming that loss of this switch directly accelerates kidney injury.
Turning RORγ back on as a potential therapy
Because RORγ is weakened but not completely absent in disease, the authors tested whether restoring its activity could protect kidneys. Delivering extra RORγ specifically to tubule cells, or providing exosomes loaded with RORγ protein, improved kidney structure and function in diabetic and aged mice: albumin leakage fell, scarring lessened, and cholesterol and inflammatory gene activity dropped. They also identified a natural compound from Ganoderma lucidum (a medicinal mushroom), called ganodermanontriol, that binds to RORγ and enhances its activity. In diabetic mice, this compound boosted RORγ’s downstream pathways, reined in cholesterol production and STING signaling, and eased kidney damage.
What this means for future kidney treatments
This work reveals RORγ as a central coordinator that links the kidney cell’s energy status to fat handling and innate immune responses. When active, it stabilizes INSIG1 and keeps both cholesterol synthesis and inflammatory alarms from running out of control. When suppressed, as in diabetes and aging, kidneys become vulnerable to lipid overload, chronic inflammation, and scarring. By showing that RORγ can be reactivated with gene delivery, exosome-based approaches, or small-molecule agonists, the study highlights a promising new strategy for slowing or preventing diabetic kidney disease and age-related kidney decline.
Citation: Liang, Z., Xiang, J., Yang, G. et al. Energy-sensing molecule RORγ regulates cholesterol metabolism and immune signaling in diabetic kidney disease and aging. Nat Commun 17, 2906 (2026). https://doi.org/10.1038/s41467-026-69724-2
Keywords: diabetic kidney disease, cholesterol metabolism, innate immunity, aging kidney, nuclear receptors