Clear Sky Science · en
Tuning aromatic cage occupancy in prenyltransferases enables selective and efficient production of rare c-prenylated flavonoids
Why smarter plant molecules matter
Many of today’s medicines and health supplements come from plant chemicals called flavonoids. When these molecules are decorated with greasy “tails” made from small carbon chains, their biological power often increases dramatically, showing promise against cancer, infections, diabetes, and inflammation. Yet these boosted versions, known as prenylated flavonoids, are hard to obtain in pure form and useful quantities from either plants or traditional chemical synthesis. This study describes a way to re‑engineer a natural enzyme so it can reliably and efficiently build rare, tailor‑made prenylated flavonoids, opening a path to greener production of future drugs and nutraceuticals. 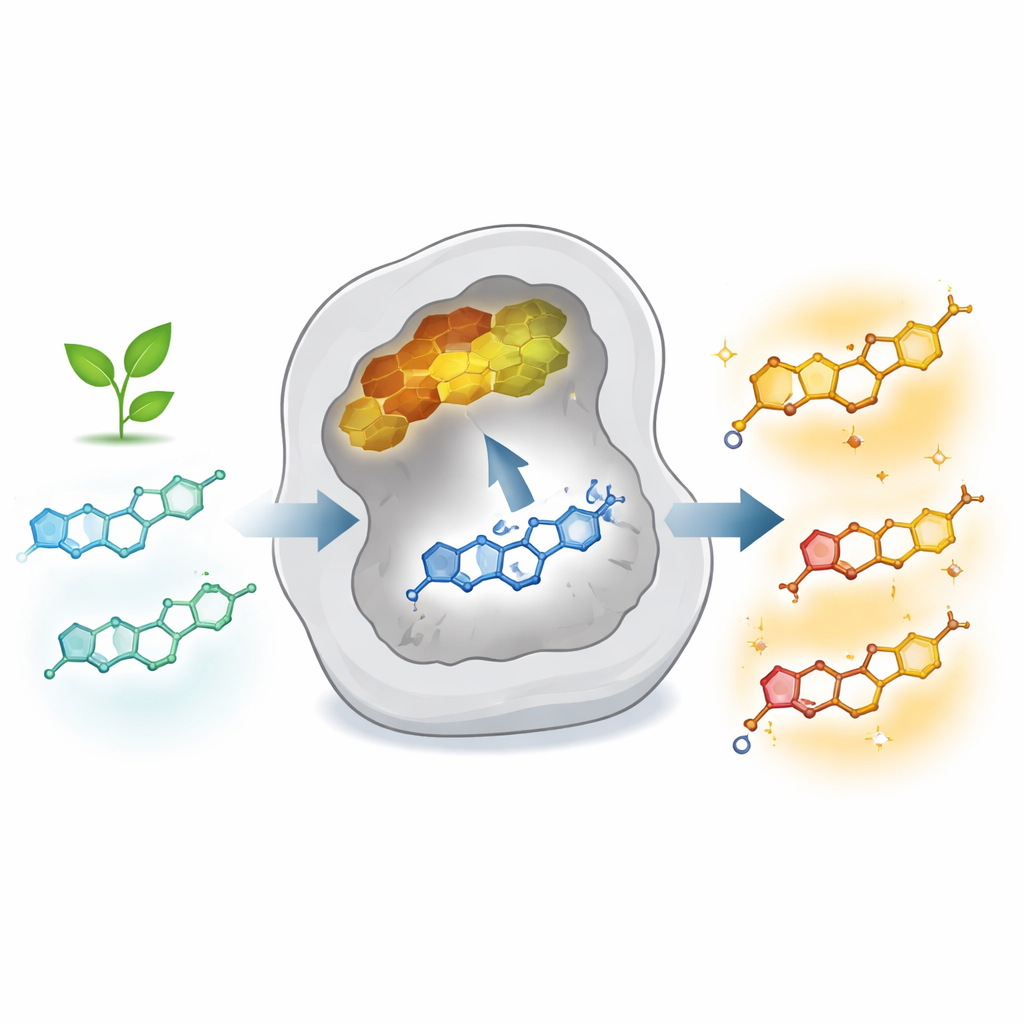
The challenge of fine-tuning plant chemistry
Flavonoids share a fragile, polyphenolic core that does not tolerate harsh chemical conditions. Attaching a prenyl tail at just one desired spot—while leaving the rest of the molecule untouched—is difficult for synthetic chemists. Nature already uses enzymes called aromatic prenyltransferases to do this job, but the known enzymes are often slow, poorly selective, and not well understood. As a result, more than a thousand naturally occurring prenylated flavonoids have been cataloged, yet many remain too scarce to study thoroughly, and the most interesting “C‑prenylated” varieties, where the tail is directly linked to a carbon atom, are especially challenging to make on demand.
Reprogramming a versatile enzyme
The authors focused on AtaPT, a fungal prenyltransferase that can act on dozens of aromatic compounds but normally shows modest activity and produces mixtures of products. Using rounds of directed evolution—systematically mutating and screening thousands of enzyme variants—they reshaped AtaPT’s active pocket to favor specific reactions on the flavonoid kaempferol. Through a combination of semi‑random mutagenesis and structure‑guided design, they identified three powerful mutants. One variant (M8) efficiently performs an unusual “reverse” prenylation at the 8‑position of kaempferol; another (G326W) directs a short prenyl group to the 3′‑position; and a third (M7) installs a longer geranyl chain at the 3′‑position with high yield and selectivity. Together, these enzymes transform a single starting flavonoid into several valuable, precisely modified products.
Discovering a molecular docking cage
To understand why these mutants worked so well, the team solved crystal structures of the engineered enzymes and ran molecular dynamics simulations. These studies revealed a key design feature: an “aromatic cage” formed by stacked ring‑shaped amino acids surrounding part of the flavonoid. In M8, a critical mutation replaces a rigid residue with histidine, helping to build this cage and lock the flavonoid into a pose where carbon 8 is perfectly aligned for attack by the incoming prenyl fragment. When a longer donor molecule such as geranyl pyrophosphate is used, its tail reaches into and fills this cage instead, pushing the flavonoid into an alternative conformation that exposes the 3′‑position. Thus, simply changing which part of the enzyme occupies the cage—and which donor is present—switches where the tail is attached and which kind of chain is preferred. 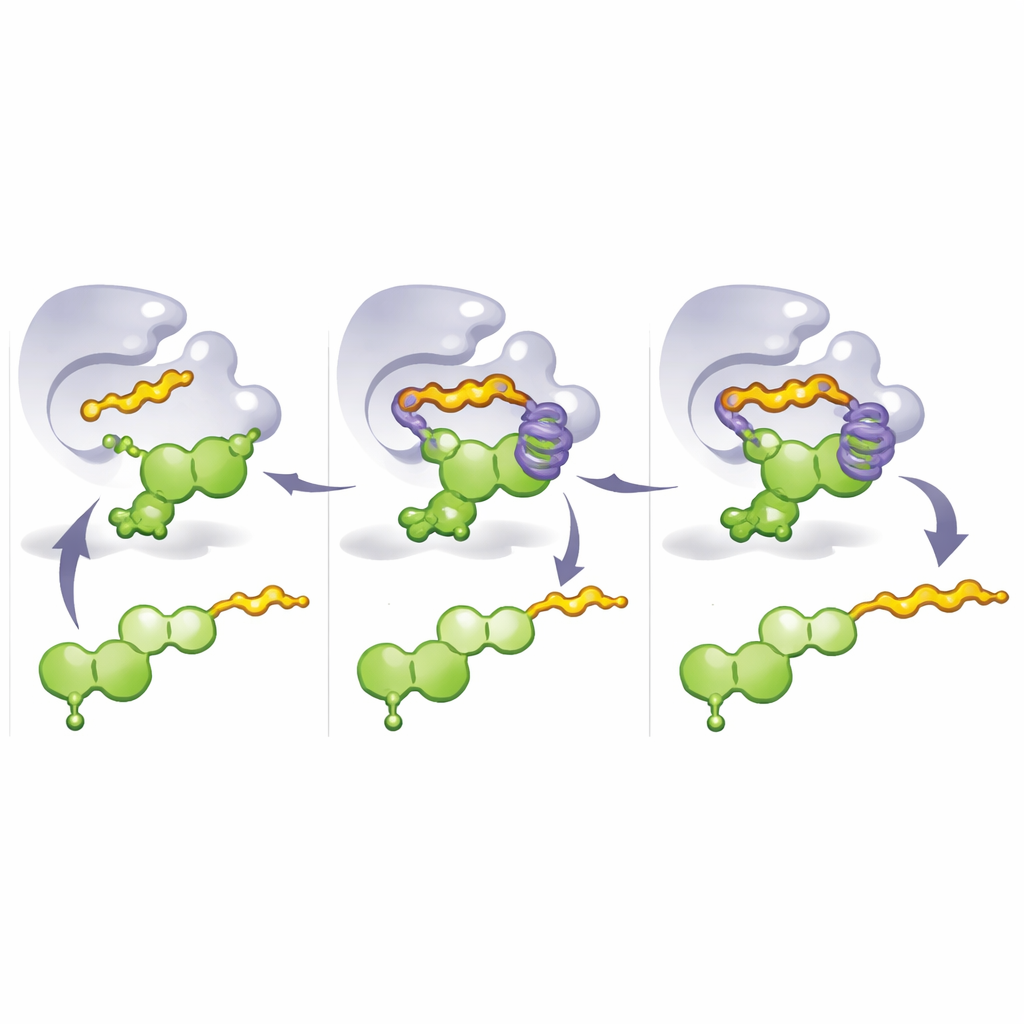
Extending the blueprint and broadening the catalog
Armed with this mechanistic picture, the researchers tested whether the aromatic cage concept could be exported to related enzymes. By introducing similar mutations into three homologous prenyltransferases from other microbes, they dramatically improved their activity and precision, confirming that the cage is a transferable design motif rather than a quirk of a single protein. They then explored a panel of 26 different flavonoids, showing that the engineered enzymes could prenylate most of them, often at defined positions. In total, the team prepared 31 prenylated flavonoids, including eight compounds never before reported, thereby enriching the palette of structures available for biological testing and drug discovery.
Building a practical, sustainable production line
To turn these reactions into a viable manufacturing route, the authors coupled their engineered enzymes with compact metabolic pathways that generate the reactive prenyl donors inside engineered E. coli cells from simple alcohols such as prenol or geraniol. In optimized lysate‑based systems, they achieved product titers up to 400 milligrams per liter on preparative scales, and successfully scaled several target molecules to liter‑volume reactions. Because the process runs in water, at mild temperatures, and avoids harsh reagents, it offers an environmentally friendly alternative to extracting trace amounts of product from tons of plant material.
What this means for future medicines
This work shows that cleverly reshaping a small region of an enzyme—the aromatic cage—can turn a sluggish, promiscuous catalyst into a precise and efficient molecular tool. By harnessing this principle, the researchers created a flexible biocatalytic platform for making rare C‑prenylated flavonoids in high purity and yield. For non‑specialists, the takeaway is that we are getting much better at asking living systems to build complex, tailor‑made plant‑like molecules on command, potentially speeding up the search for new treatments while reducing environmental impact.
Citation: Qiu, R., Huang, H., Chi, J. et al. Tuning aromatic cage occupancy in prenyltransferases enables selective and efficient production of rare c-prenylated flavonoids. Nat Commun 17, 2945 (2026). https://doi.org/10.1038/s41467-026-69706-4
Keywords: prenylated flavonoids, enzyme engineering, biocatalysis, directed evolution, green chemistry