Clear Sky Science · en
Dipeptidylpeptidase 4 inhibition attenuates gestational pathologies via immune homeostasis restoration in the pulmonary-uterine axis
Why Lung Infections in Pregnancy Matter
Respiratory infections like flu or coronavirus are usually thought of as problems confined to the lungs. But during pregnancy, these illnesses are linked to babies born too small, too early, or not surviving at all. This study in mice asks a crucial question: how can an infection in the airways disturb the womb, even when the virus never reaches the uterus or the baby? The researchers uncover a hidden communication line between lung and uterus and show that an existing drug can protect pregnancies by calming this immune cross-talk.
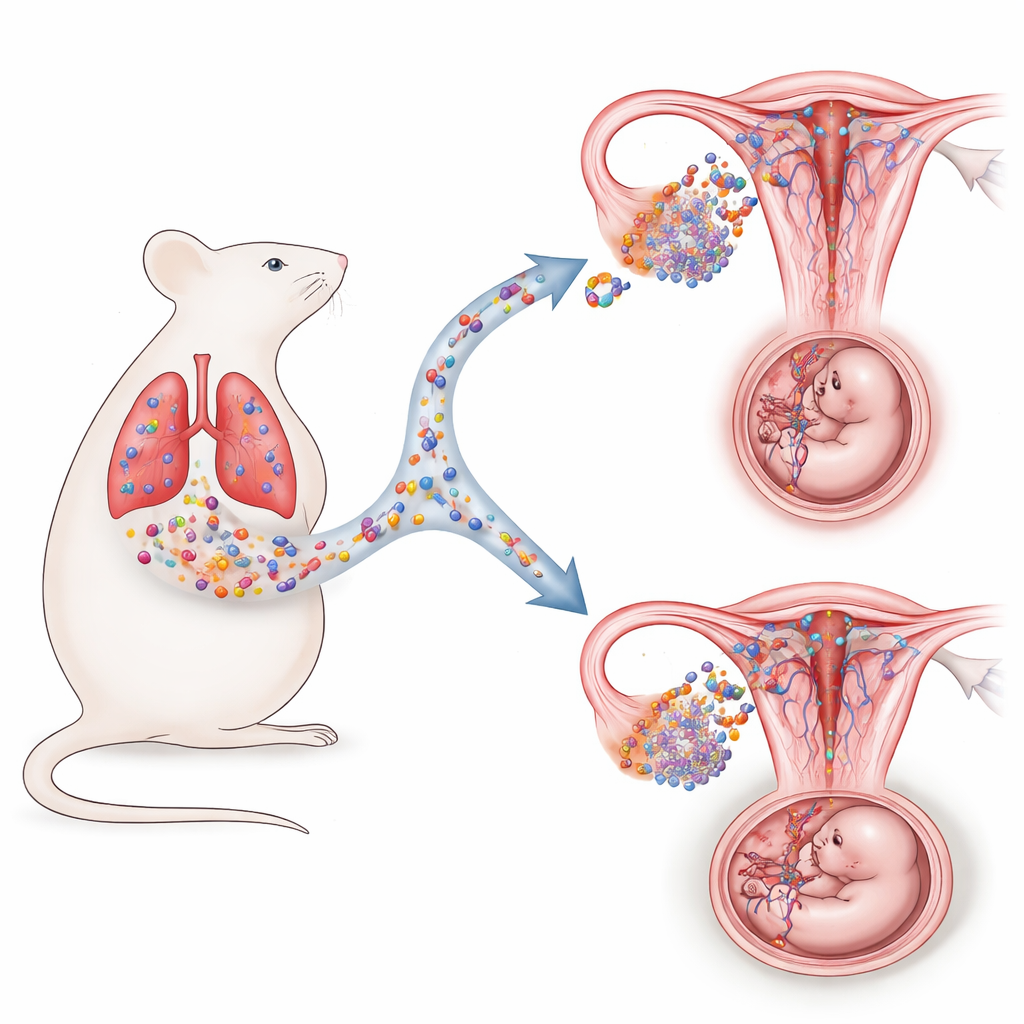
From Sick Lungs to Struggling Babies
The team began by infecting pregnant mice with an H1N1 influenza virus early in gestation. As expected, the mothers developed inflamed, damaged lungs. Surprisingly, the fetuses also showed clear signs of growth restriction: they were shorter, lighter, and had smaller placentas than those from uninfected mothers. Yet the virus itself stayed in the lungs; no viral genetic material was detected in the uterus or placenta. Instead, the uterine environment shifted into an abnormal low-alert state: immune signals that normally help build blood vessels and support placental invasion were dampened, and the arteries that feed the placenta became thick-walled and narrow, starving the growing embryos of nutrients and oxygen.
A Chemical Messenger Links Lung and Uterus
To understand how lung inflammation could quiet the immune system in the uterus, the researchers looked for molecules rising in the bloodstream after infection. They found elevated levels of DPP4, an enzyme with known roles in immunity and metabolism. More importantly, they discovered that a particular immune “brake” receptor called IL1R2 was strongly induced in the inflamed lungs and then accumulated in the uterus. IL1R2 acts as a decoy for the powerful alarm molecule IL-1, soaking it up and blocking its ability to trigger helpful inflammation and blood vessel growth. Detailed cell analyses and single-cell RNA sequencing revealed that a specific subset of myeloid cells—macrophage-like cells in the lung—began to overproduce IL1R2 after infection, then circulated in the blood and appeared in the uterine lining, where they blunted the local immune reactions needed for healthy placental remodeling.
Repurposing a Diabetes Drug to Protect Pregnancy
The study then tested whether blocking DPP4 could interrupt this harmful lung–uterus immune axis. Pregnant mice with respiratory influenza or a coronavirus-like infection were treated with sitagliptin, a widely used DPP4 inhibitor for type 2 diabetes. Sitagliptin did not reduce the amount of virus in the lungs, but it did soften the lung’s overactive inflammatory response and sharply lowered IL1R2 levels in both lung and uterus. In treated animals, the uterine immune environment resembled that of healthy pregnancies: blood vessels at the maternal–fetal interface regained normal thickness and diameter, trophoblast cells invaded properly, and fetuses and placentas grew back to near-normal size. These benefits persisted to birth, restoring litter sizes and offspring growth without detectable side effects in uninfected mothers.
Proving IL1R2 Is the Culprit
To confirm that IL1R2 itself drives the damage, the researchers engineered mice lacking the Il1r2 gene. When these females were infected with influenza during pregnancy, their lungs still became inflamed, but their uteruses did not accumulate excess IL1R2-bearing cells. As a result, placental arteries remodeled correctly, trophoblast invasion was preserved, and fetal growth remained normal, despite ongoing lung disease. A separate coronavirus model produced the same pattern: lung-restricted infection, fetal growth restriction, elevated IL1R2 in the uterus, and rescue of pregnancy outcomes when sitagliptin was given. Together, these experiments pinpoint IL1R2-positive myeloid cells as key messengers that carry the imprint of lung inflammation to the womb.
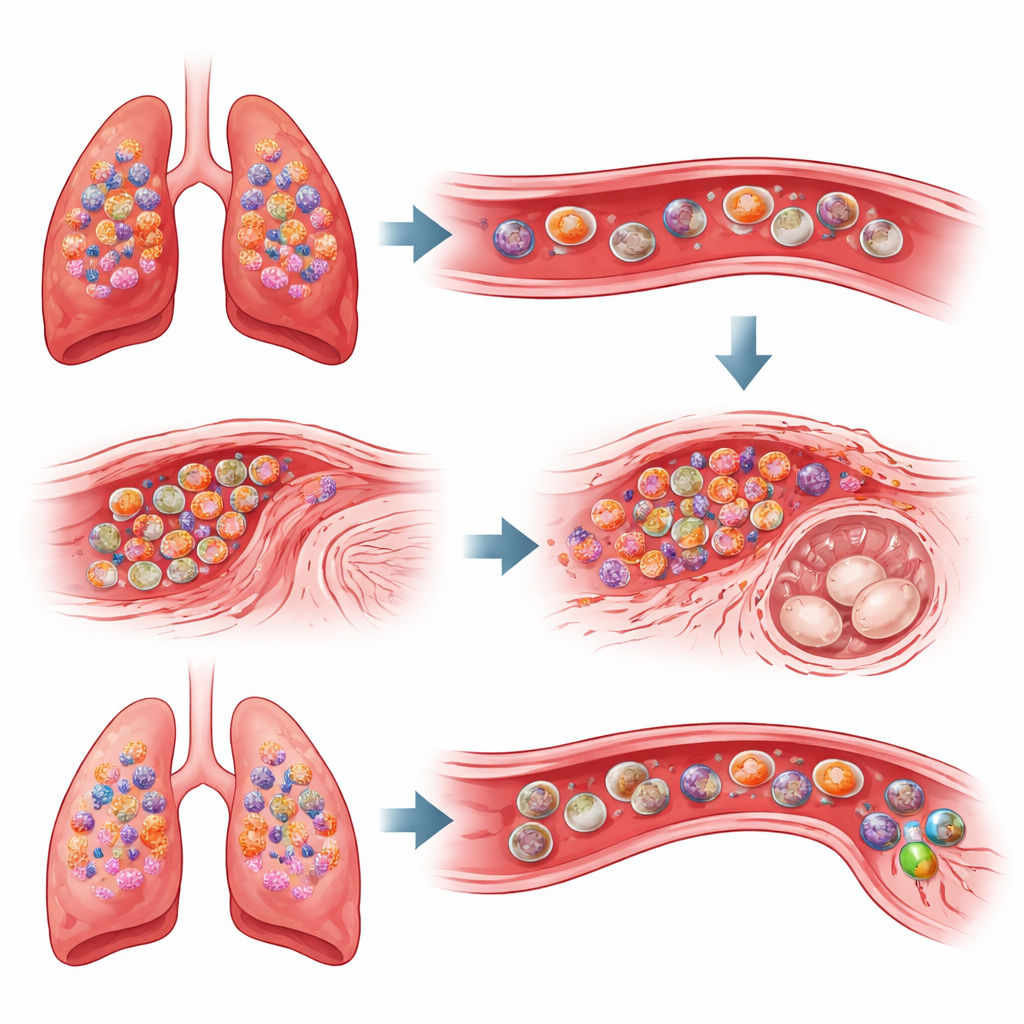
What This Means for Pregnant Patients
Overall, the work reveals a previously unrecognized “pulmonary–uterine axis,” where severe inflammation in the lungs sends immune cells and signals that quietly sabotage placental development and fetal growth. By dialing down this overreaction—either by blocking DPP4 with sitagliptin or by removing IL1R2—the uterine environment can be reset, allowing normal blood vessel formation and healthy embryos even while the respiratory infection runs its course. Although the findings are in mice and will require careful validation in human studies, they raise the possibility that drugs already in clinical use could be repurposed to shield pregnancies from the collateral damage of serious respiratory infections.
Citation: Shi, G., Xi, S., Lv, M. et al. Dipeptidylpeptidase 4 inhibition attenuates gestational pathologies via immune homeostasis restoration in the pulmonary-uterine axis. Nat Commun 17, 2851 (2026). https://doi.org/10.1038/s41467-026-69620-9
Keywords: pregnancy and flu, maternal immune system, placental development, DPP4 inhibitors, respiratory viral infection