Clear Sky Science · en
FATE-MAP predicts teratogenicity and human gastrulation failure modes by integrating deep learning and mechanistic modeling
Why early embryo errors matter
Many pregnancies never make it past the earliest weeks, often for reasons that remain invisible to both parents and doctors. One critical hurdle is a stage called gastrulation, when a simple ball of cells reorganizes into the basic layers that will form every organ in the body. If this step goes wrong, the result can be pregnancy loss or birth defects. Yet scientists have had few human-specific tools to see how medications and chemicals might derail this fragile process. This study introduces FATE-MAP, a new platform that combines stem cell models, advanced imaging, and artificial intelligence to forecast which compounds may harm early human development and to reveal how those failures arise.
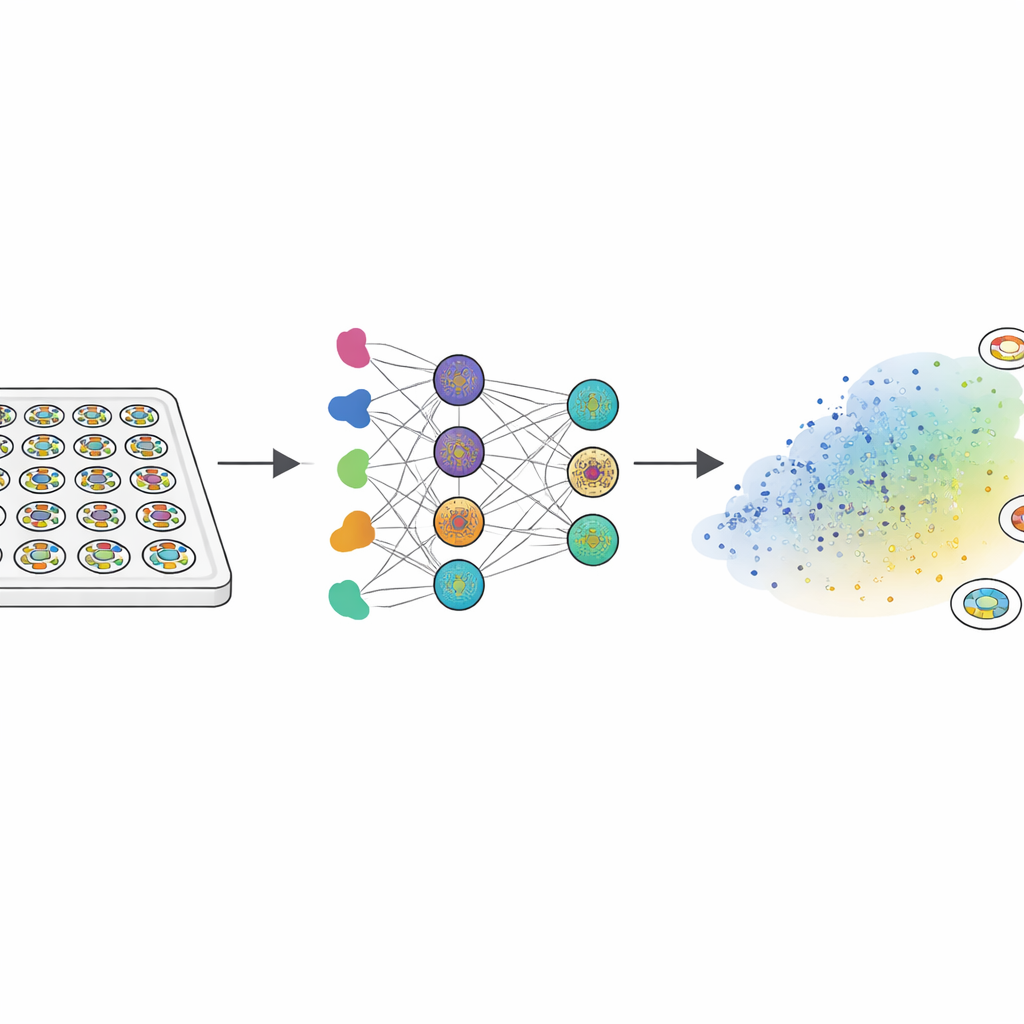
Building a lab stand‑in for the early embryo
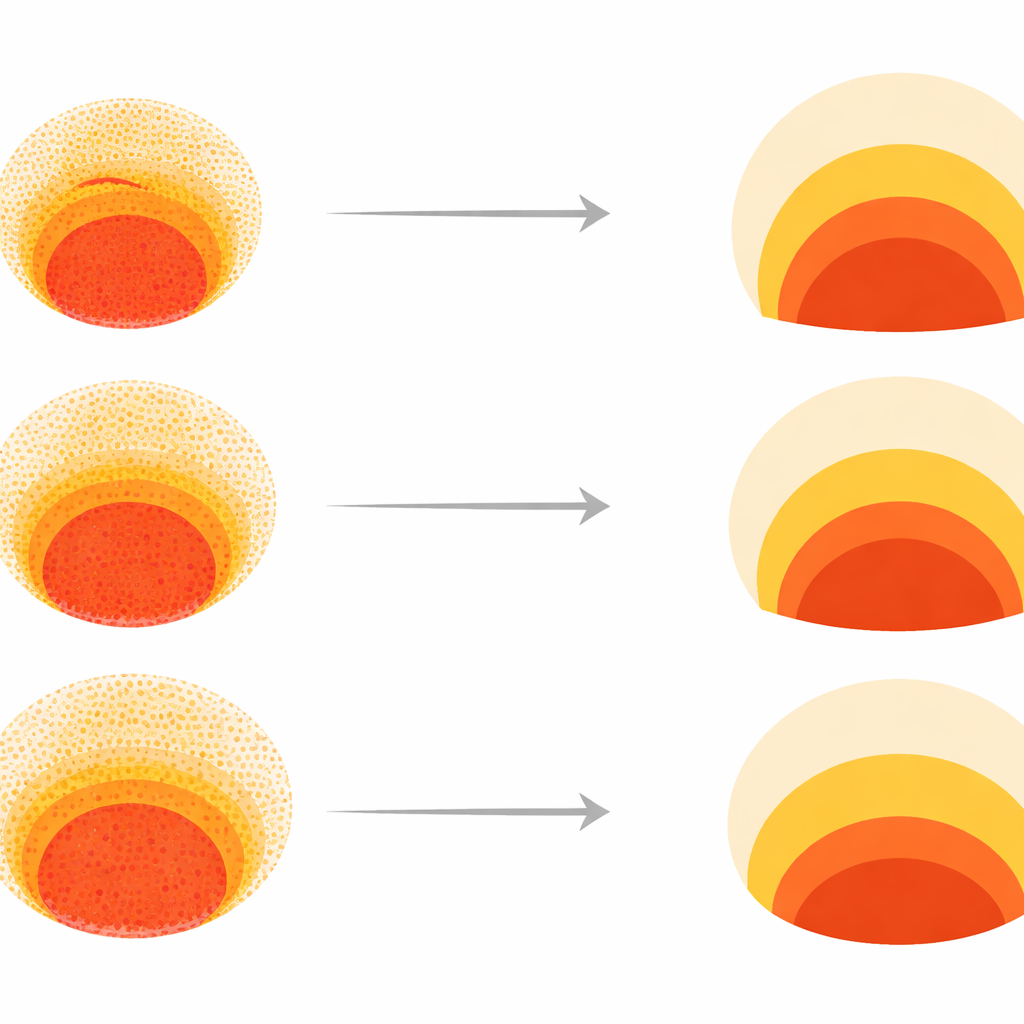
Because real human embryos cannot be studied in depth, the researchers turned to "gastruloids"—tiny, flat discs grown from human embryonic stem cells. When exposed to a signaling protein called BMP4, these discs self-organize into three concentric zones that mirror early embryo tissues: an inner region that stays stem‑like, a middle band that becomes mesoderm (precursors of muscle, bone, and blood), and an outer ring related to early extra‑embryonic tissue. The team treated about 2000 of these colonies with a library of 210 drugs that target key growth and signaling pathways. Using fluorescent markers and custom image analysis, they turned each colony’s pattern into a 150‑number fingerprint that captured how the three tissue types were arranged from edge to center.
Mapping normal patterns and failure modes
To make sense of this huge dataset, the scientists used a technique that clusters similar patterns close together in a two‑dimensional "morphospace." Most colonies fell into a broad region where all three tissue types appeared in the correct radial order, differing mainly in how wide each ring was. But several distinct "failure modes" emerged: some patterns lacked the central stem‑like region, others lost the mesoderm band, and some broke radial symmetry altogether. Because such defects would likely prevent proper body plan formation, the team treated these clusters as developmental failure modes rather than harmless variation. This map became the reference against which both experimental data and computer models could be compared.
Teaching computers to predict risky compounds
The next step was to see whether a computer could predict where a new compound would land in this morphospace using only its chemical formula. The authors used a transformer model—a deep learning approach originally developed for language—to convert each molecule’s SMILES string into a high‑dimensional numerical description. A neural network then learned to map these descriptions to the 150‑number colony fingerprints, and a second network translated those fingerprints into positions on the morphospace map. For dozens of drugs with known pregnancy outcomes, FATE-MAP’s purely in silico predictions matched or outperformed leading stem‑cell and computational teratogenicity tests at classifying compounds as risky or safe. Strikingly, several well-known teratogens all converged on the same failure cluster characterized by loss of mesoderm.
Revealing how failures happen, not just that they do
Black‑box AI alone cannot explain why a colony fails. To add mechanism, the team built a mathematical model of how three major signaling molecules—BMP, Wnt, and Nodal—spread across the colony and drive cells toward different fates over time. By adjusting parameters such as starting cell density and how resistant the central stem‑like cells are to differentiation (captured by a quantity they call SOX2 stability), they could simulate patterns and project them into the same morphospace. This revealed that most normal variation in patterns could be explained by two nearly independent axes: how crowded the cells are and how easily they let go of their stem‑like identity. Lower cell density allowed Wnt and Nodal signals to spread further inward, broadening the mesoderm band, while higher SOX2 stability kept the central region in a more primitive state despite differentiation cues.
From warnings to future safer medicines
Finally, the researchers used FATE-MAP to flag two clinical‑stage drugs with no prior human pregnancy data as potential teratogens. Both were predicted, and then experimentally confirmed, to produce the same mesoderm‑loss failure mode seen with classic retinoid teratogens, and they also caused characteristic defects in zebrafish embryos. More broadly, the work shows how combining realistic stem‑cell models, machine learning, and mechanistic equations can turn early human development into a mappable landscape. In the long run, platforms like FATE-MAP could help drug developers screen huge numbers of compounds for subtle developmental risks before they ever reach patients, while at the same time illuminating the fundamental rules that guide an embryo from a simple sheet of cells toward a fully formed body.
Citation: Rufo, J., Qiu, C., Han, D. et al. FATE-MAP predicts teratogenicity and human gastrulation failure modes by integrating deep learning and mechanistic modeling. Nat Commun 17, 3327 (2026). https://doi.org/10.1038/s41467-026-69596-6
Keywords: teratogenicity, gastrulation, stem cell models, deep learning, developmental toxicity