Clear Sky Science · en
Structural and mechanistic insights into azetidine-associated αKG-NHFe enzyme OkaE with multifunctional catalysis
Why a tiny enzyme matters for future medicines and pesticides
Many powerful drugs and insecticides rely on complex ring-shaped molecules that are hard for chemists to build. This study uncovers how a single fungal enzyme, called OkaE, can sculpt one such ring—an unusually strained four‑membered azetidine—and then further remodel the molecule in several different ways. Understanding how OkaE works could inspire greener routes to new medicines and highly selective insect control agents that spare humans and other vertebrates.
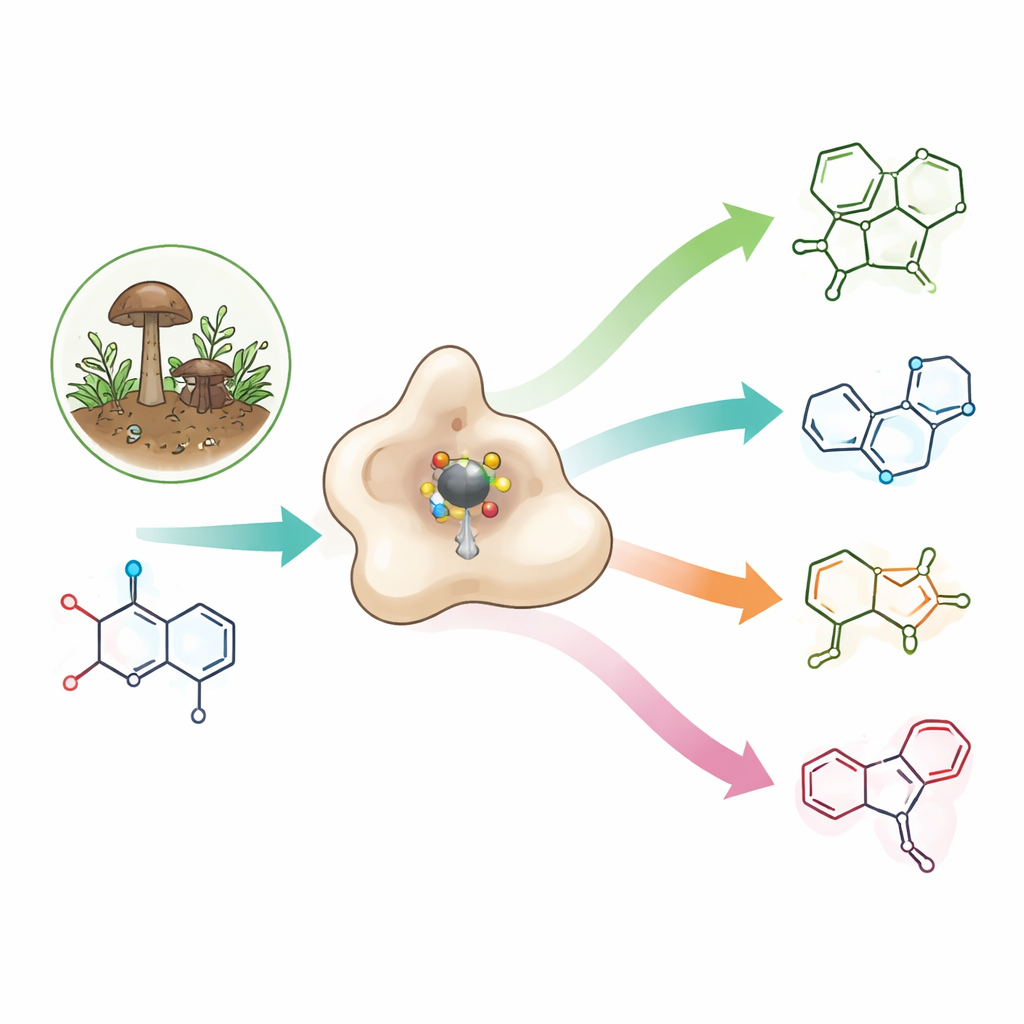
A rare building block with big pharmaceutical promise
Azetidines are tiny four‑membered nitrogen rings that pack a lot of strain into a small space. That strain often translates into useful properties: azetidines can improve how drug molecules fit into their targets, move through the body, and dissolve in water. They already appear in experimental antibiotics, anti‑cancer agents, and potential Alzheimer’s drugs, as well as in okaramines—natural insecticides made by certain fungi that selectively disable nerve channels in invertebrates while leaving human channels untouched. Yet, because azetidine rings are so strained and unstable, chemists usually need several energy‑intensive steps and harsh conditions to make them.
Nature’s unusual solution: the OkaE enzyme
Fungi tackle this challenge with specialized enzymes. Most known azetidine‑forming enzymes join a carbon atom to a nitrogen atom using a common cellular cofactor called SAM. OkaE breaks that rule. It belongs to a large family of iron‑dependent enzymes that normally insert oxygen atoms into molecules. OkaE uses iron and a helper molecule, alpha‑ketoglutarate, to form a highly reactive iron–oxygen species able to pluck hydrogen atoms from its substrate, okaramine A. Unlike its cousins, OkaE forges the azetidine ring by creating a new carbon–carbon bond inside this already complex natural product, marking it as an outlier even within a very versatile enzyme family.
One enzyme, many chemical tricks
When the researchers studied purified OkaE in the test tube, they expected just azetidine formation and a simple oxidation. Instead, they found a small chemical factory. Starting from okaramine A, OkaE produced at least four new molecules, called neuokaramines I–IV, in addition to known intermediates. These products showed that OkaE can carry out a cascade of transformations: forming the azetidine ring, adding hydroxyl groups, converting an alcohol into a carboxylic acid, opening bonds in the original framework, and even installing an additional tiny three‑membered oxygen ring (an epoxide). By varying the amount of its co‑substrate, alpha‑ketoglutarate, and feeding OkaE different intermediates, the team mapped how these reactions branch from a single starting molecule within one or a few enzyme cycles.
How shape and motion steer the reactions
To see how OkaE selects among these possibilities, the authors solved high‑resolution crystal structures of the enzyme bound to metal, helper molecule, and okaramine A, and combined them with quantum‑level computer simulations. OkaE has the classic "jelly‑roll" fold of its enzyme family, but its active site is decorated with sulfur‑containing methionine residues and a tryptophan that cradle one end of okaramine through subtle “Met–π” interactions between sulfur atoms and aromatic rings. This network holds the substrate so that two specific positions—one on a carbon next to the azetidine ring, and one on a nearby hydroxyl group—face the reactive iron–oxygen center. Simulations show that, after oxygen activation, the iron–oxygen fragment swivels into an orientation where it can abstract a hydrogen from either site, creating a fork in the reaction pathway. Altering single residues in the methionine–tryptophan network nudges the substrate closer to one site or the other, simplifying the product mixture or even collapsing OkaE’s behavior to a single main reaction.
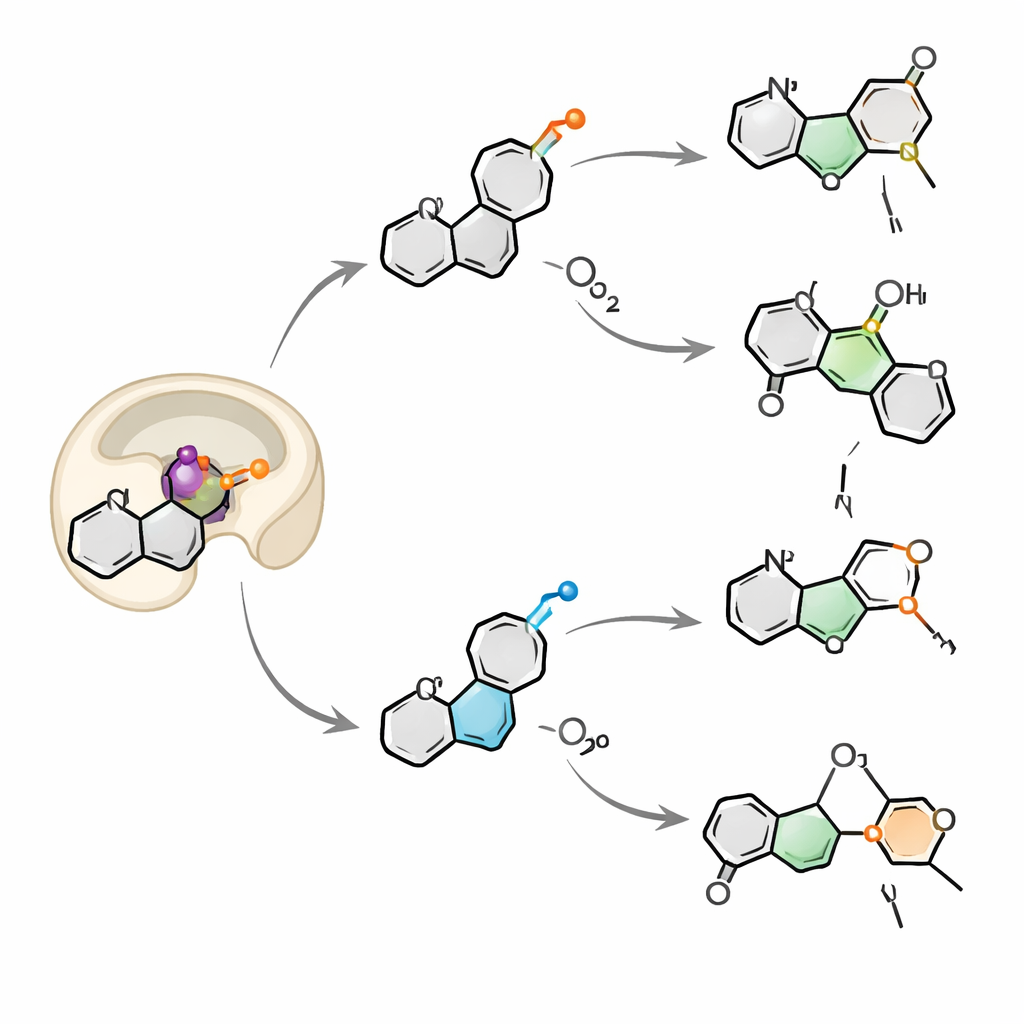
Breathing in two oxygens at once
By tracing oxygen atoms labeled with the heavier isotope 18O, the team discovered that OkaE draws oxygen from both molecular oxygen in the air and water in solution. For most products, the oxygen bound to iron can swap with water before ending up in the molecule, explaining the mixed labeling patterns. One product, neuokaramine IV, stood out: its pattern could only be explained if a second oxygen molecule is captured and built into the same substrate within a single catalytic cycle, forming and then breaking a delicate peroxide bridge. This “two breath” mechanism is extremely rare for this enzyme family and underscores how flexibly OkaE can choreograph radical and positively charged intermediates within the same active site.
What this means for future design
In plain terms, OkaE is a molecular multitool. It can not only assemble a difficult ring that chemists struggle to make, but also refashion that ring and its surroundings into several distinct scaffolds—all while running on air, iron, and a simple cellular acid. By revealing how a specific interaction network in the protein positions its substrate and how a swiveling iron–oxygen unit opens different reaction channels, this work provides a blueprint for engineering “programmable” biocatalysts. Such tailored enzymes could streamline the late‑stage modification of natural products, helping chemists build better drugs and insecticides with less waste and greater precision.
Citation: Wang, X., Yu, J., Liu, T. et al. Structural and mechanistic insights into azetidine-associated αKG-NHFe enzyme OkaE with multifunctional catalysis. Nat Commun 17, 2861 (2026). https://doi.org/10.1038/s41467-026-69519-5
Keywords: azetidine biosynthesis, nonheme iron enzyme, okaramine insecticides, biocatalyst engineering, oxidative cascade reactions