Clear Sky Science · en
Ligand-dependent Wnt signaling promotes gastric cancer metastasis through hyaluronan expression in microenvironment
Why this research matters
Gastric cancer is one of the world’s deadliest cancers largely because it often spreads to distant organs such as the liver. Many drugs focus on killing cancer cells themselves, but this study shows that the surrounding "soil" in which cancer cells grow—the supporting tissue and gel-like material around them—can be just as important. By uncovering how stomach tumors reshape their environment to thrive in the liver, this work points toward new ways to block metastasis rather than just shrinking primary tumors.
Signals that drive tumor growth
The researchers focused on a family of molecular signals called Wnt, which help regulate stem cells in healthy tissues but are frequently hijacked by cancers. In colon cancer, Wnt activity is usually driven by mutations inside the cancer cells themselves. In gastric cancer, however, such mutations are less common, yet Wnt activity is still high. That suggested many stomach tumors may depend on Wnt ligands—signal molecules secreted by cells—rather than permanent genetic switches, raising the question of how these outside signals influence tumor growth and spread.
Engineered mice that mimic human disease
To explore this, the team created sophisticated mouse models carrying combinations of three common gastric cancer mutations (in the genes Kras, Tgfbr2, and Trp53, grouped as KTP). Some mice also produced extra Wnt1 ligand specifically in the stomach (WKTP). Mice with only the cancer-driver mutations developed thickened stomach linings and abnormal cell types, but adding Wnt1 pushed these changes further, producing clearly dysplastic—pre-cancerous to cancerous—tumors. When the scientists grew mini-tumors (organoids) from these mice and transplanted them into recipient animals, both KTP and WKTP cells formed sizable tumors at the original site, showing that the three-driver mutation set was enough for local tumor formation.
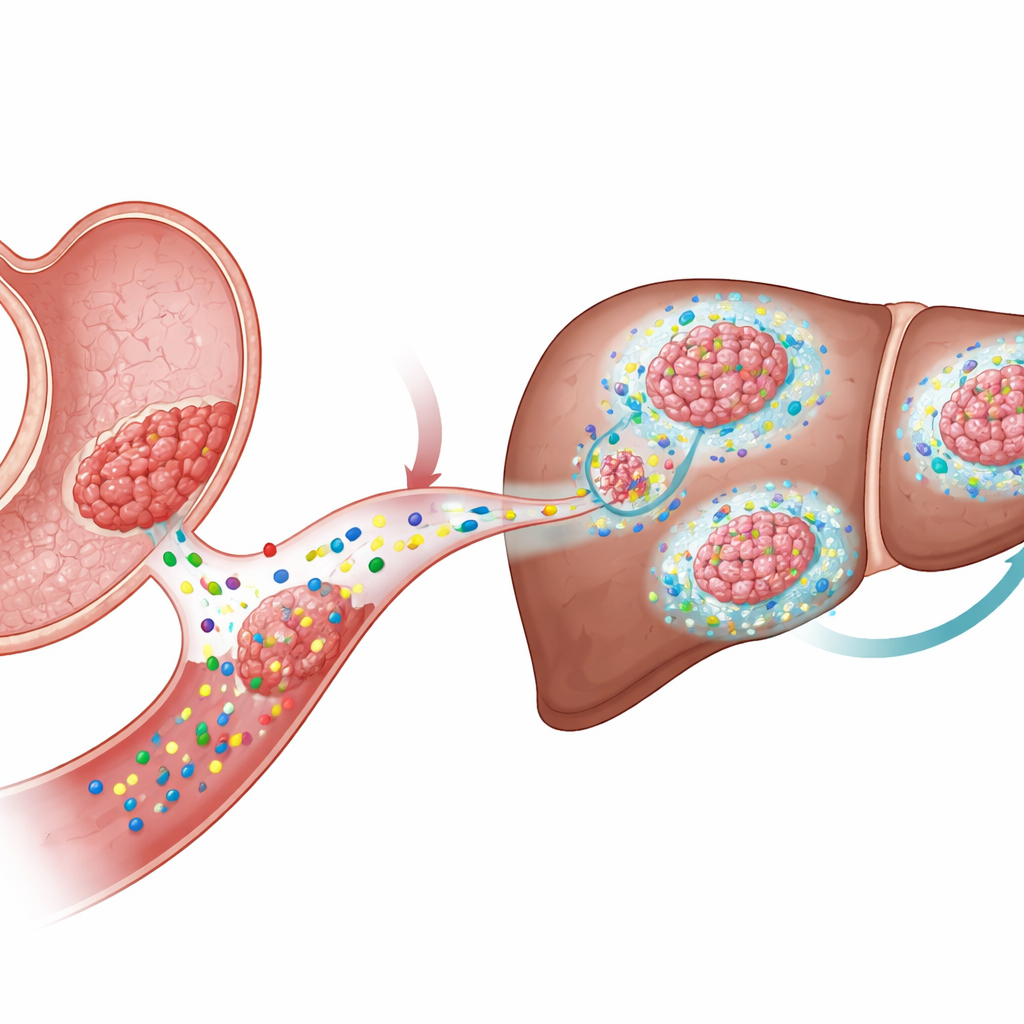
When and where spread to the liver occurs
The key difference emerged when the researchers looked at metastasis. They injected KTP or WKTP organoids into the spleen, a route that sends cells directly to the liver. Only the Wnt1-producing WKTP cells formed large, fibrotic metastatic tumors in the liver, even though both types of cells initially arrived there in similar numbers. Drugs that block release of Wnt ligands sharply reduced liver metastases and caused the remaining tumors to appear more differentiated and less proliferative. Interestingly, forcing constant Wnt activity inside KTP cells by disabling a gatekeeper gene (Apc) did not reliably produce metastases. This indicated that Wnt’s role in spreading disease depends on its action in the surrounding non‑cancerous cells, not just within the tumor cells themselves.
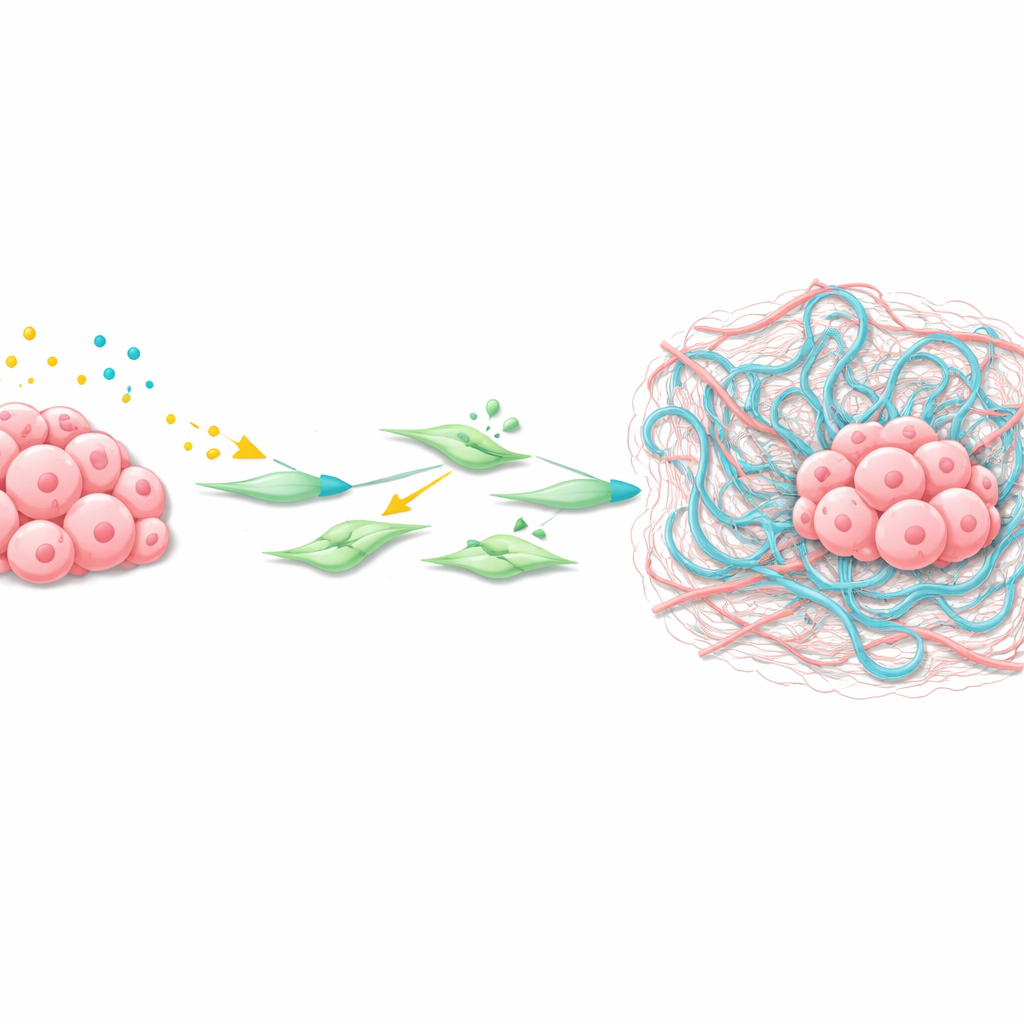
How the tumor reshapes its neighborhood
Using spatial transcriptomics—a technique that maps gene activity across thin tissue slices—the team zoomed in on the liver metastases’ microenvironment. They found a cluster of cancer-associated fibroblasts (CAFs) surrounding the tumors with strongly active Wnt and TGFβ signaling. These CAFs produced high levels of Has2, an enzyme that synthesizes hyaluronan, a long sugar polymer that accumulates as a viscous mesh in tissues. Experiments with isolated liver stellate cells and CAFs showed that Wnt ligands and TGFβ act together to turn on Wnt target genes, activate the fibroblasts, and greatly boost Has2 levels, causing strong contraction and remodeling of the surrounding matrix.
A sticky web that helps cancer settle
The researchers then demonstrated that hyaluronan itself is a key accomplice. In both mouse and human gastric cancer liver metastases, hyaluronan was abundant around tumor nests from very early stages. When WKTP cancer cells were engineered to make hyaluronidases—enzymes that break down hyaluronan—they still formed tumors at the injection site but produced far fewer metastases in the liver. This suggests that the sticky hyaluronan-rich matrix created by activated fibroblasts forms a protective, nurturing niche that helps disseminated gastric cancer cells survive, proliferate, and organize into full metastatic tumors.
What this means for future treatments
Taken together, the study reveals a chain of events: gastric tumors secrete Wnt signals; these cooperate with TGFβ to activate fibroblasts; the fibroblasts make large amounts of hyaluronan, building a fibrotic, gel-like microenvironment in the liver that favors metastatic outgrowth. For patients, this points to new strategies that do not target cancer cells alone. Therapies that block Wnt ligand production or reception, interfere with TGFβ-driven fibroblast activation, or prevent hyaluronan buildup could make distant organs less hospitable to wandering gastric cancer cells, potentially turning a rapidly lethal disease into a more controllable one.
Citation: Furutani, Y., Oshima, H., Hong, C.P. et al. Ligand-dependent Wnt signaling promotes gastric cancer metastasis through hyaluronan expression in microenvironment. Nat Commun 17, 2777 (2026). https://doi.org/10.1038/s41467-026-69470-5
Keywords: gastric cancer, metastasis, Wnt signaling, tumor microenvironment, hyaluronan