Clear Sky Science · en
Reverse hydrogen spillover accelerates electrocatalytic nitrate reduction to ammonia on Ru/WO3-x in acidic media
Cleaner Fertilizer from Dirty Water
Ammonia is the backbone of modern fertilizers and a promising way to store hydrogen fuel, but making it today relies on the century‑old Haber–Bosch process, which consumes huge amounts of energy and emits large quantities of carbon dioxide. At the same time, many industries dump nitrate‑rich acidic wastewater that can damage ecosystems. This study explores a way to turn that pollution into a resource: using electricity to convert nitrate in acidic water directly into ammonia, while avoiding wasteful hydrogen gas production and corrosion of the catalyst.
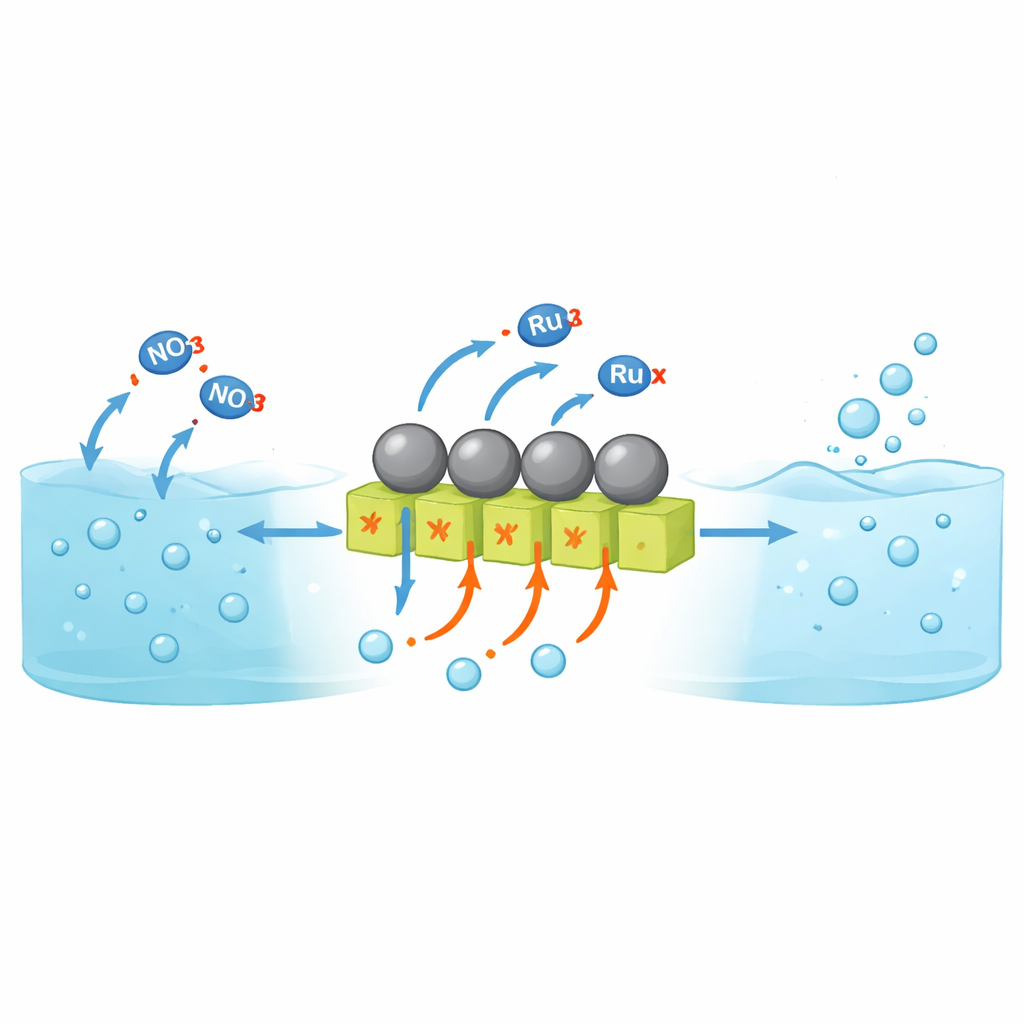
Why Turning Nitrate into Ammonia Is Hard
Electrochemical reduction of nitrate allows ammonia to be made at room temperature from water and waste streams instead of from fossil fuels at high temperature and pressure. However, most research so far has focused on neutral or alkaline solutions. In strongly acidic solutions—much closer to real industrial nitrate effluents—catalyst materials tend to corrode and another reaction takes over: protons combine to form hydrogen gas rather than helping to build ammonia. Conventional acid‑resistant metals like ruthenium are excellent at binding hydrogen, which is good for ammonia formation but also makes them highly active for hydrogen gas evolution. As a result, very little of the electrical current ends up making ammonia, especially at the high current densities needed for practical devices.
A Catalyst That Shares the Work
The researchers designed a composite catalyst that separates where hydrogen and nitrate are handled, while keeping them close enough to cooperate. They grew nanorods of tungsten oxide (WO3‑x) on a titanium support and then decorated their surfaces with tiny particles of ruthenium. Tungsten oxide is unusually good at soaking up protons from acidic solution and holding them inside its crystal structure, yet it is poor at releasing them as hydrogen gas. Detailed microscopy and X‑ray measurements show that strong electronic interactions arise at the interface between ruthenium and tungsten oxide, creating many contact sites where the electronic structure of ruthenium is subtly altered and oxygen vacancies form in the oxide. These changes help define where protons are stored and how they move.
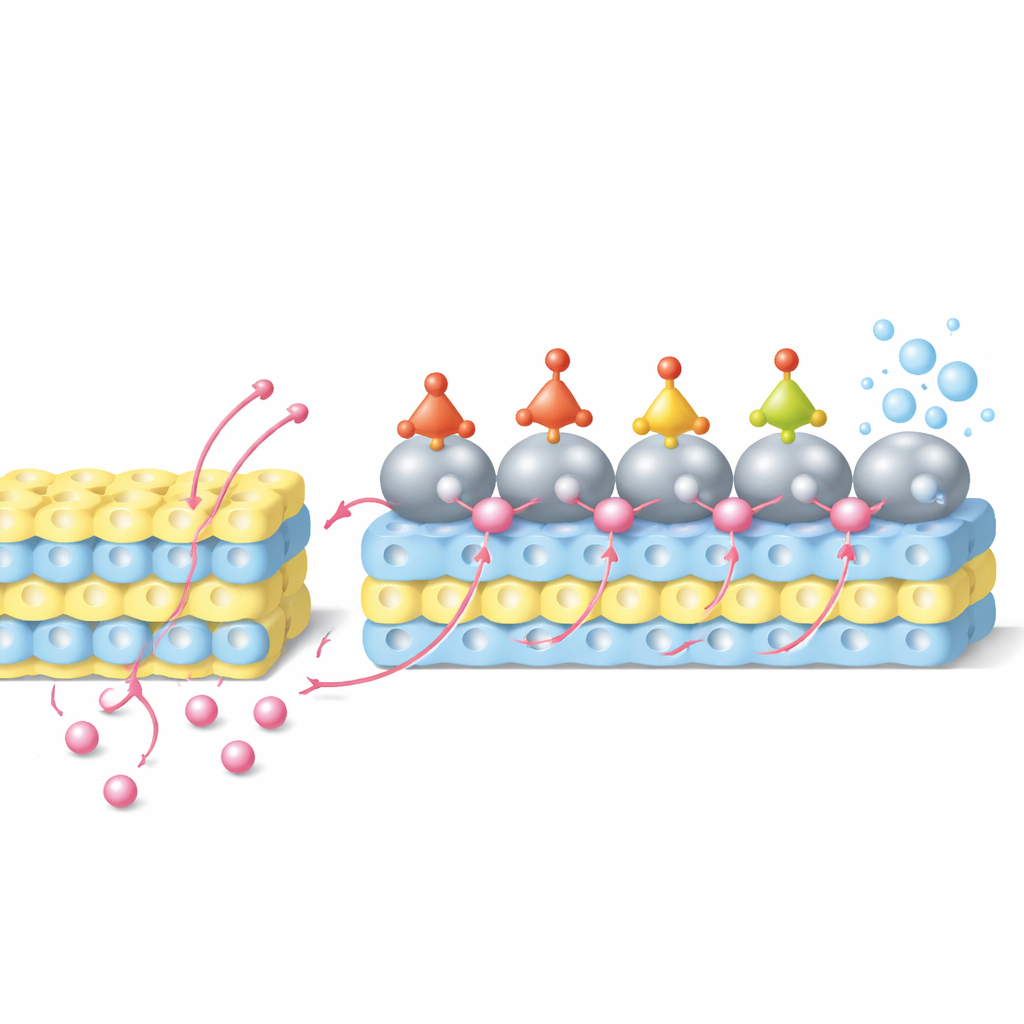
Reverse Hydrogen Spillover in Action
In this system, protons first enter the tungsten oxide lattice and are stored near the catalyst surface. Under an applied voltage, they then migrate from the oxide support back toward the ruthenium particles—a process termed reverse hydrogen spillover. At the same time, nitrate ions from solution land on ruthenium and are stepwise hydrogenated through a series of nitrogen‑oxygen intermediates until ammonia is formed. Computer simulations reveal that ruthenium atoms at the interface, which have donated some electron density to tungsten, provide especially favorable sites for these hydrogen‑adding steps, lowering the energy barriers for key transformations. Experiments using electrochemical methods, in‑situ spectroscopy, and isotope‑labeled hydrogen confirm that a significant fraction of the protons stored in the oxide indeed travel to ruthenium and participate in nitrate reduction instead of forming hydrogen gas.
Record Performance and a Working Device
Because the support acts as a fast proton reservoir and delivery network, the catalyst maintains a balanced and abundant hydrogen supply even at very high current densities. In acidic nitrate solution, the Ru/WO3‑x electrode achieves a current density of 500 milliamperes per square centimeter at a near‑zero applied voltage, while converting about 94 percent of the electrical charge into ammonia—values that surpass previously reported acidic nitrate‑to‑ammonia catalysts. The structure remains stable over long operation, and almost no unwanted nitrogen by‑products are detected. To demonstrate practical use, the team built a “batterolyzer” that pairs nitrate reduction on this catalyst with oxidation of sulfide on another electrode. The cell simultaneously generates electrical power and converts nitrate and sulfide pollutants into useful ammonium and sulfur‑based products.
Turning Waste and Electricity into Value
For a non‑specialist, the key message is that the authors have found a clever way to let one material store and shuttle hydrogen while another focuses on transforming nitrate into ammonia. By engineering how protons move—rather than simply making a more active metal—they greatly suppress wasteful hydrogen gas formation and boost ammonia output in harsh acidic conditions that resemble real industrial waste streams. This concept of sharing tasks between a support and a metal catalyst via controlled hydrogen spillover could be applied to many other electrochemical reactions, opening paths to cleaner fertilizer production and to devices that both clean wastewater and supply useful chemicals and energy.
Citation: Zhu, W., Lin, YC., Cong, J. et al. Reverse hydrogen spillover accelerates electrocatalytic nitrate reduction to ammonia on Ru/WO3-x in acidic media. Nat Commun 17, 2830 (2026). https://doi.org/10.1038/s41467-026-69335-x
Keywords: ammonia synthesis, nitrate wastewater, electrocatalysis, hydrogen spillover, tungsten oxide catalyst