Clear Sky Science · en
SPINK2 silencing suppresses leukemic proliferation and restores myeloid commitment via MECOM downregulation in acute myeloid leukaemia
Why this matters for blood cancer
Acute myeloid leukemia (AML) is a fast-moving blood cancer in which immature white blood cells take over the bone marrow, crowding out normal blood production. Many patients, especially those whose leukemia cells carry numerous chromosomal abnormalities (so‑called complex karyotypes), respond poorly to current drugs. This study uncovers a molecule called SPINK2 as a key helper that allows leukemic cells to keep dividing while refusing to grow up into normal, mature blood cells. Because SPINK2 can be switched off experimentally, it may offer a new way to slow the disease and push cancer cells back toward a healthier fate.
A switch found in early blood stem cells
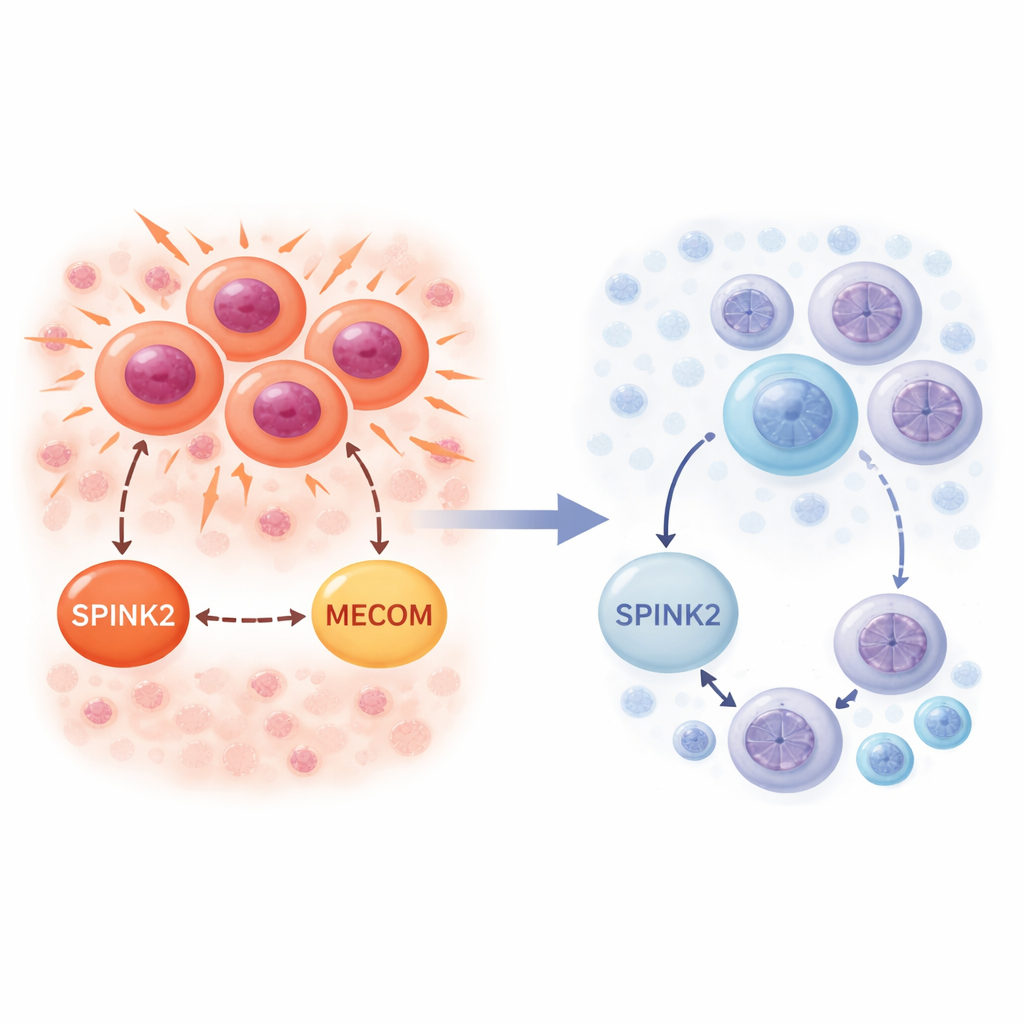
The researchers first asked where SPINK2 normally appears in healthy bone marrow. Using single‑cell RNA sequencing data, which measure gene activity cell by cell, they found that SPINK2 is mainly switched on in the most primitive blood stem and progenitor cells marked by CD34. These are quiescent, slow‑cycling cells that sit at the top of the blood cell family tree. As cells mature along either the myeloid or lymphoid pathways, SPINK2 activity drops sharply and is essentially absent in fully developed blood cells. This pattern suggests that SPINK2 is part of the program that defines very early stem‑like cells and is usually turned off once cells commit to a specific job.
When the same switch fuels leukemia growth
The team then looked at SPINK2 in leukemic bone marrow from patients with AML. Again using single‑cell data, they found SPINK2 active in leukemia cells that most closely resemble stem cells, as well as in rapidly cycling AML blasts. In contrast to the healthy setting, here SPINK2 went hand‑in‑hand with strong signals from cell‑cycle genes, especially those driving the DNA‑copying S‑phase. In bulk patient cohorts from major cancer genomics projects, higher SPINK2 levels were linked to worse survival and were particularly elevated in people with complex karyotype AML. Together, these observations indicate that leukemia cells have hijacked a stem‑cell gene, using SPINK2 to support relentless division while preserving a primitive identity.
Turning SPINK2 off slows cancer and nudges it to mature
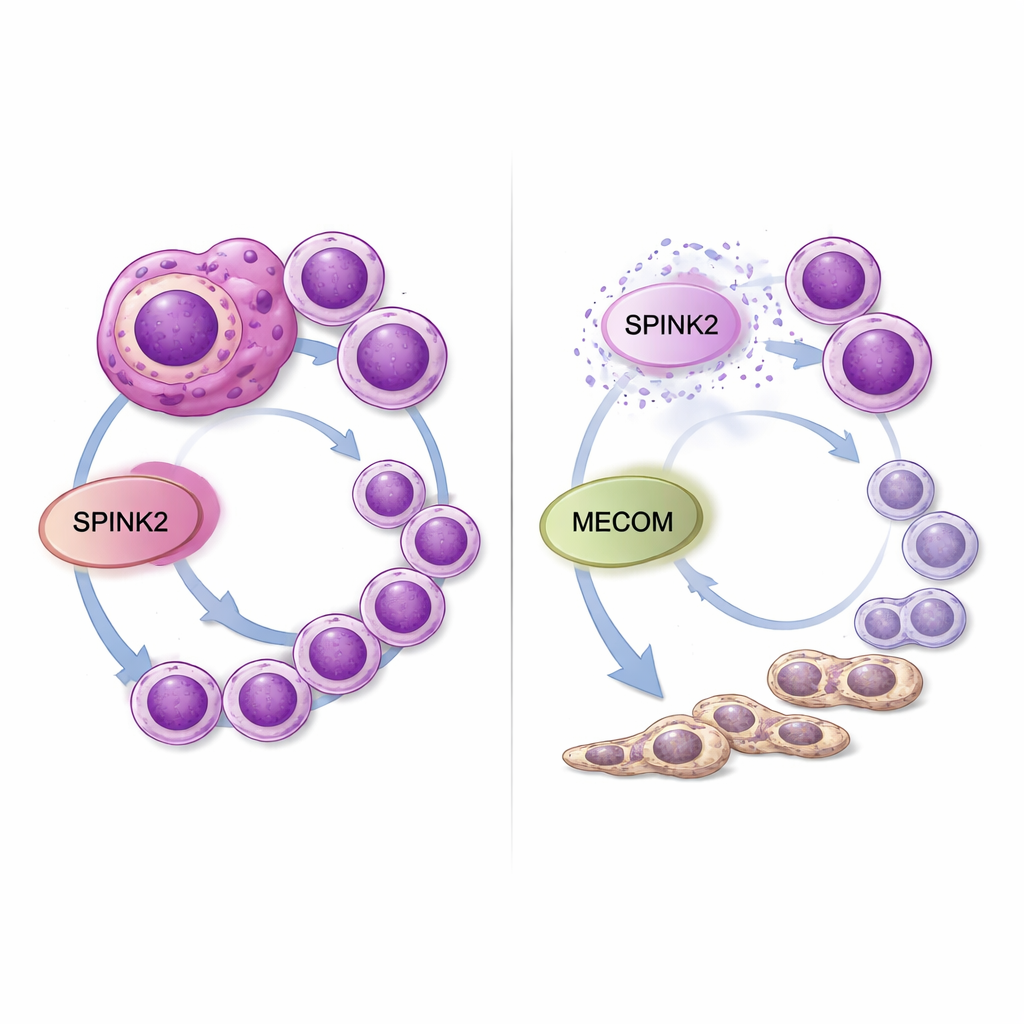
To test whether SPINK2 is merely a marker or a true driver, the scientists engineered an AML cell line called FUJIOKA—representing complex karyotype disease—to carry a doxycycline‑inducible short hairpin RNA that selectively silences SPINK2. When SPINK2 was switched off, leukemic cells divided far more slowly. Detailed cell‑cycle analysis showed that many cells got stuck in the G0/G1 phase and failed to progress through S and G2/M, without any notable increase in cell death. At the same time, cells began to display higher levels of surface markers typical of mature myeloid cells, indicating that the usual developmental block was easing. In other words, SPINK2 loss both removed the growth push and released the brake on differentiation.
A molecular link to another leukemia driver
RNA sequencing of FUJIOKA cells before and after SPINK2 silencing revealed broad changes in gene activity. Hundreds of genes that control the cell cycle, chromosome segregation, and mitotic checkpoints were turned down, while sets linked to protein synthesis and immune responses were turned up. One standout was MECOM, a well‑known leukemia‑promoting transcription factor that helps stem‑like cells self‑renew and prevents myeloid maturation. MECOM expression dropped sharply when SPINK2 was silenced. Analyses of a large pediatric AML dataset confirmed that SPINK2 and MECOM levels tend to rise and fall together specifically in patients with complex karyotypes. This points to a SPINK2–MECOM axis that may coordinate excessive self‑renewal and division in high‑risk AML.
What this means for future treatments
To a lay observer, these findings suggest that SPINK2 acts like a hidden dial in leukemia cells that is turned up too high: it keeps cells in a youthful, stem‑like state and encourages them to multiply uncontrollably. Dialing SPINK2 down in the lab both slows the cancer cells and encourages them to grow into more normal myeloid cells, while also reducing the activity of another dangerous gene, MECOM. Because SPINK2 is normally restricted to early blood stem cells and largely silent in most adult tissues, drugs that target this molecule or its partnership with MECOM might selectively weaken aggressive AML—especially complex karyotype cases—without severely harming normal blood production. Further work in animal models and drug‑development efforts will be needed, but this study marks SPINK2 as a promising new vulnerability in a form of leukemia that badly needs better options.
Citation: Ventura, A.B., Loconte, T., Ahmed, A. et al. SPINK2 silencing suppresses leukemic proliferation and restores myeloid commitment via MECOM downregulation in acute myeloid leukaemia. Cell Death Discov. 12, 135 (2026). https://doi.org/10.1038/s41420-026-02988-1
Keywords: acute myeloid leukemia, SPINK2, leukemia stem cells, MECOM, myeloid differentiation