Clear Sky Science · en
The Sirt2–Nur77 axis regulates muscle stem cell quiescence and senescence via epigenetic–metabolic synergy
Why muscles grow weaker with age
Aging often sneaks up on us as simple weakness: climbing stairs feels harder, jars are tougher to open, and legs tire more quickly. This study asks a fundamental question behind that everyday experience: why do our muscle stem cells, which normally repair and rebuild muscle, lose their edge with age? By uncovering a key control switch inside these cells, the authors point to new ways drugs might one day slow or even reverse age‑related muscle loss.
A hidden switch inside muscle stem cells
Skeletal muscle is constantly maintained by a small reserve army of muscle stem cells that normally rest quietly until injury or exercise calls them into action. In older animals, these cells wake up more slowly, regenerate muscle less effectively, and gradually disappear, contributing to sarcopenia, the age‑related loss of muscle mass and strength. The researchers focused on two proteins whose levels shift with age in many organs: Nur77, which declines, and Sirt2, which rises. They suspected that these opposing changes form a common aging “axis” and set out to test its role specifically in skeletal muscle.
How Sirt2 wears down Nur77
Inside muscle stem cells, Nur77 acts as a gene‑control factor that helps decide when a resting stem cell should activate and start building new fibers. The team discovered that Sirt2 physically binds Nur77 in the cell nucleus and chemically trims small acetyl groups from a precise spot on Nur77 (a single amino‑acid position called K310 in mice). When this acetyl mark is removed, Nur77 becomes unstable: it is tagged for destruction, broken down by the cell’s waste‑disposal machinery, and its levels fall. When the acetyl mark is present, Nur77 is stabilized and can bind the DNA of target genes more effectively.
From molecular tweak to failing muscle repair
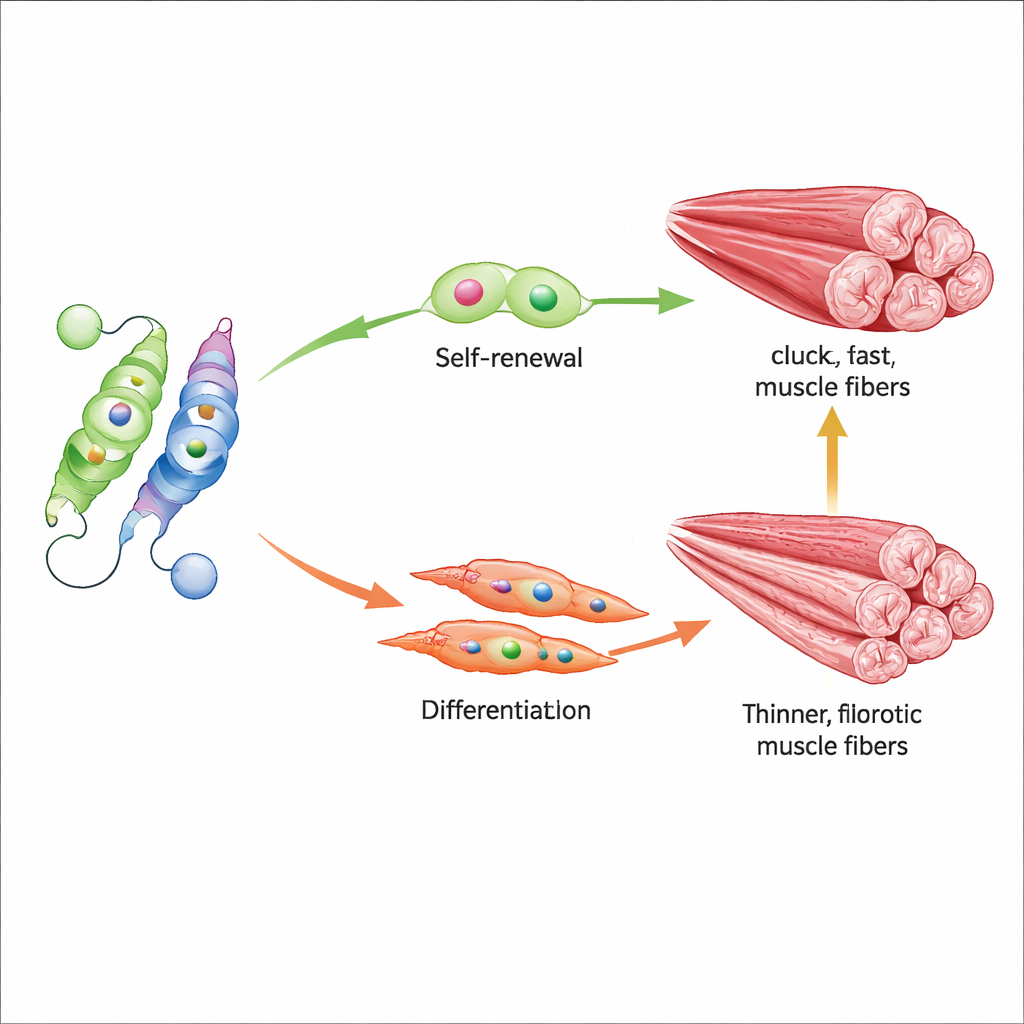
Nur77’s most important target in this context is Myf5, a gene that helps push muscle stem cells from deep rest into a ready‑to‑repair state. The researchers showed that acetylated Nur77 binds strongly to the Myf5 control region and switches it on, promoting timely activation of stem cells and the formation of new muscle fibers after injury. When Nur77 is missing entirely, or when the K310 site is locked in a de‑acetylated state, muscles develop smaller fibers, regenerate poorly after damage, and show more scarring. Stem cells also divide in a less favorable way, tending to exhaust their pool instead of balancing self‑renewal with differentiation. In aged mice, where Sirt2 is higher and Nur77 lower, this imbalance is especially pronounced.
Drugs that nudge the balance back
The study also tested two small molecules that push this molecular seesaw in opposite directions. AGK2 blocks Sirt2, indirectly preserving the acetyl mark on Nur77 and preventing its destruction, while CSNB directly activates Nur77. In old mice, treatment with either compound increased Nur77 activity, boosted Myf5 and early muscle‑building proteins, and measurably improved grip strength and running distance. On the tissue level, treated muscles better maintained fast, powerful fibers and showed less fibrosis, suggesting that tuning this single acetylation site can influence both stem cell behavior and overall muscle quality.
What this means for healthy aging
Taken together, the work outlines a simple but powerful chain of events: with age, rising Sirt2 strips a protective chemical tag from Nur77, Nur77 becomes unstable and declines, key repair genes like Myf5 are less active, stem cells renew and repair less efficiently, and muscles gradually atrophy and scar. By restoring the acetylated form of Nur77—either by inhibiting Sirt2 or directly stimulating Nur77—the authors could partially reset this chain and rejuvenate muscle function in old mice. While much remains to be done before such strategies could be safely applied to humans, the study spotlights a specific molecular switch that may one day be targeted to help people stay stronger, more mobile, and more independent in later life.
Citation: Wang, Y., Yang, Y., Yu, W. et al. The Sirt2–Nur77 axis regulates muscle stem cell quiescence and senescence via epigenetic–metabolic synergy. Cell Death Dis 17, 429 (2026). https://doi.org/10.1038/s41419-026-08645-w
Keywords: muscle aging, muscle stem cells, Nur77, Sirt2, sarcopenia