Clear Sky Science · en
Generation of proliferative hESC-derived grape-clustered hepatocyte organoids with multipolar architecture as regenerative counterpart via synergy of YAP and IGF2 pathways
Building Tiny Livers in the Lab
The liver quietly handles many of the body’s toughest jobs—detoxifying drugs, processing nutrients, and managing fats and sugars. Yet human liver tissue is scarce, and mature liver cells quickly lose their function in a dish. This study describes a way to grow tiny, grape-like clusters of human liver cells from embryonic stem cells that not only stay alive and divide for weeks, but also reproduce key features of real liver tissue, including the ability to move bile and other substances in a highly organized way.
Why Liver Cell Clusters Matter
Scientists have long hoped to build three-dimensional liver “organoids” that behave like real liver tissue. Existing organoids made from bile duct cells tend to form hollow cysts with simple inside–outside polarity, which does not match the complex structure of true liver cells. In the body, each hepatocyte has several “apical” surfaces that join with neighbors to form a branching bile canal network while simultaneously exchanging substances with the blood on its opposite side. Grape-like hepatocyte organoids derived from primary adult liver cells can mimic this multipolar shape and function, but they depend on donor tissue that is limited and variable. The new work shows that similar grape-clustered organoids can be produced from human embryonic stem cells, offering a potentially more plentiful and standardized source.
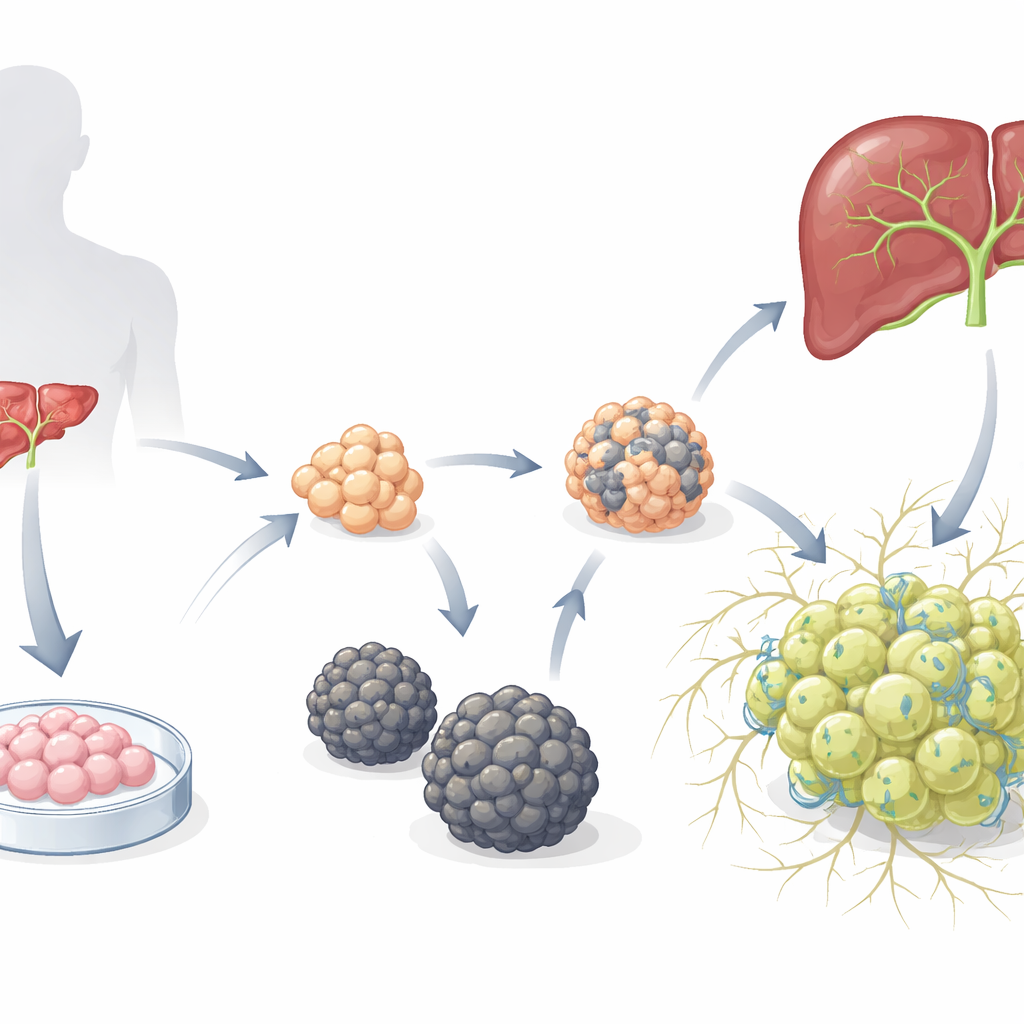
Two Very Different Mini-Liver Communities
The researchers began with previously established hepatoblast organoids—immature liver progenitors grown from human embryonic stem cells—and placed them into a medium designed to favor hepatocyte development. Over time, two distinct types of organoids emerged. One type formed smooth spheres (S-heporgs) that grew poorly, showed signs of aging and cell death, and failed to organize a proper bile canal network. The other type formed irregular, grape-like clusters (G-heporgs) that expanded up to several hundred micrometers across, contained many dividing and binucleated cells, expressed high levels of mature liver proteins such as albumin, and built intricate, branched canal structures between cells. Genetic and functional tests showed that G-heporgs were metabolically active and proliferative, whereas S-heporgs were stuck in a senescent, injury-like state.
Inflammatory Signals, Growth Factors, and a Regeneration Switch
An unexpected twist was that the failing S-heporgs seemed to help the more successful G-heporgs appear. As S-heporgs aged, they released inflammatory molecules and created a microenvironment resembling a damaged liver. In this setting, G-heporgs activated a growth program centered on the factor IGF2 and its receptor, which in turn engaged the PI3K–AKT signaling route known to drive cell division. Adding extra IGF2 to the culture boosted both the number and size of G-heporgs, while blocking its receptor or downstream signaling nearly eliminated them. At the same time, the G-heporgs showed strong activation of YAP, a protein that senses mechanical and injury cues and is crucial for liver regrowth after damage. When the team artificially turned up YAP activity with a small molecule agonist, the organoids could be expanded for more than two months and many passages without losing their grape-like architecture; turning YAP off sharply curtailed growth.
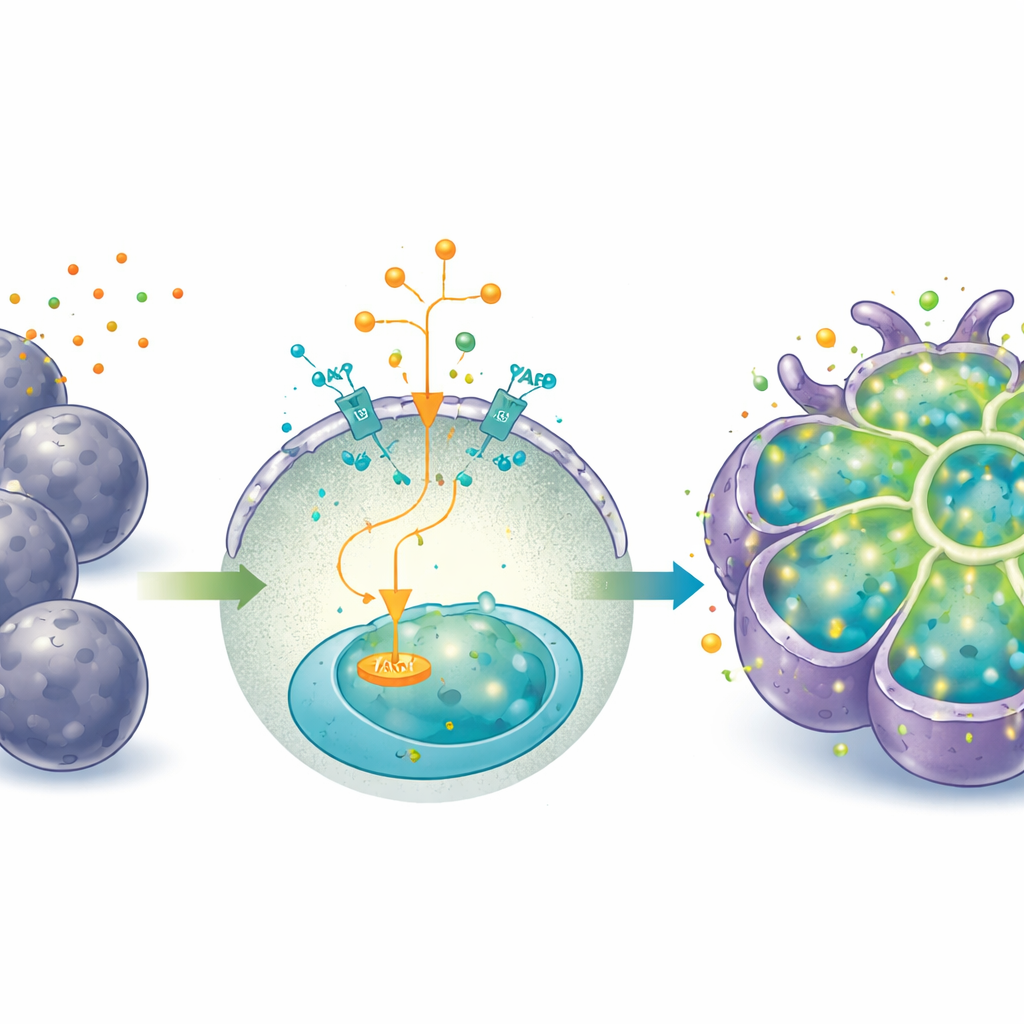
From Growth Mode to Work Mode
Prolonged YAP activation, while excellent for expansion, kept the cells in a more fetal, progenitor-like state and dampened some mature liver functions. To shift the organoids from “growth mode” into “work mode,” the researchers changed the medium—removing IGF2, YAP agonist, and a Wnt activator, and adding the hormone dexamethasone and the cytokine oncostatin M. Under these conditions, the organoids developed larger, polygonal cells with lower nucleus-to-cytoplasm ratios and more binucleated cells, all hallmarks of maturing hepatocytes. They produced more albumin and urea, handled a model dye (indocyanine green) in and out of the cells, and increased key drug-metabolizing enzymes, especially after exposure to known enzyme inducers.
Recreating Bile Flow and Metal Handling
A central achievement of these organoids is their ability to recreate polarized transport—the directed movement of substances to specific surfaces of the cell. The expandable G-heporgs formed bile canal-like channels marked by proteins that normally concentrate at apical membranes. Live imaging showed fluorescent test compounds being pumped into these tiny lumens, and drugs known to cause cholestasis in patients disrupted this canal network and caused the dyes to build up inside cells, closely mirroring toxic side effects in the clinic. The team also tracked ATP7B, a copper transporter mutated in Wilson disease, and found that in response to high copper, it moved from internal compartments to the bile-facing surfaces and lysosome-like structures, just as in native liver. This demonstrates that the organoids can model how hepatocytes direct metals and other molecules in a polarized fashion.
What This Means for Future Liver Research
By harnessing the combined action of IGF2 and YAP pathways, the authors created a renewable source of human hepatocyte organoids that assemble into grape-like clusters and build functional bile canal networks. While still somewhat less mature than adult liver tissue and lacking non-hepatocyte support cells, these structures capture key aspects of liver regeneration, polarity, and transport. For non-specialists, this means scientists are getting closer to lab-grown mini-livers that can be used to study inherited liver diseases, test drug safety—especially for bile-related toxicity—and eventually explore cell-based therapies, all without depending solely on scarce donor organs.
Citation: Wu, H., Liu, S., Chen, S. et al. Generation of proliferative hESC-derived grape-clustered hepatocyte organoids with multipolar architecture as regenerative counterpart via synergy of YAP and IGF2 pathways. Cell Death Dis 17, 381 (2026). https://doi.org/10.1038/s41419-026-08635-y
Keywords: liver organoids, stem cell derived hepatocytes, bile canaliculi, liver regeneration, drug induced liver injury