Clear Sky Science · en
TPTE, a testis-specific PTEN family member, drives spermatogenesis via PI(4,5)P2 synthesis
Why This Matters for Male Fertility
Many couples struggling to conceive never get a clear answer about why sperm are few in number or move poorly. This study uncovers a previously mysterious testis-specific enzyme, called TPTE, that helps guide both the genetic reshuffling of sperm precursors and the building of their whip-like tails. By showing how TPTE shapes key signaling molecules inside developing sperm, the research points to new molecular explanations for certain forms of male infertility and suggests future avenues for diagnosis and treatment.
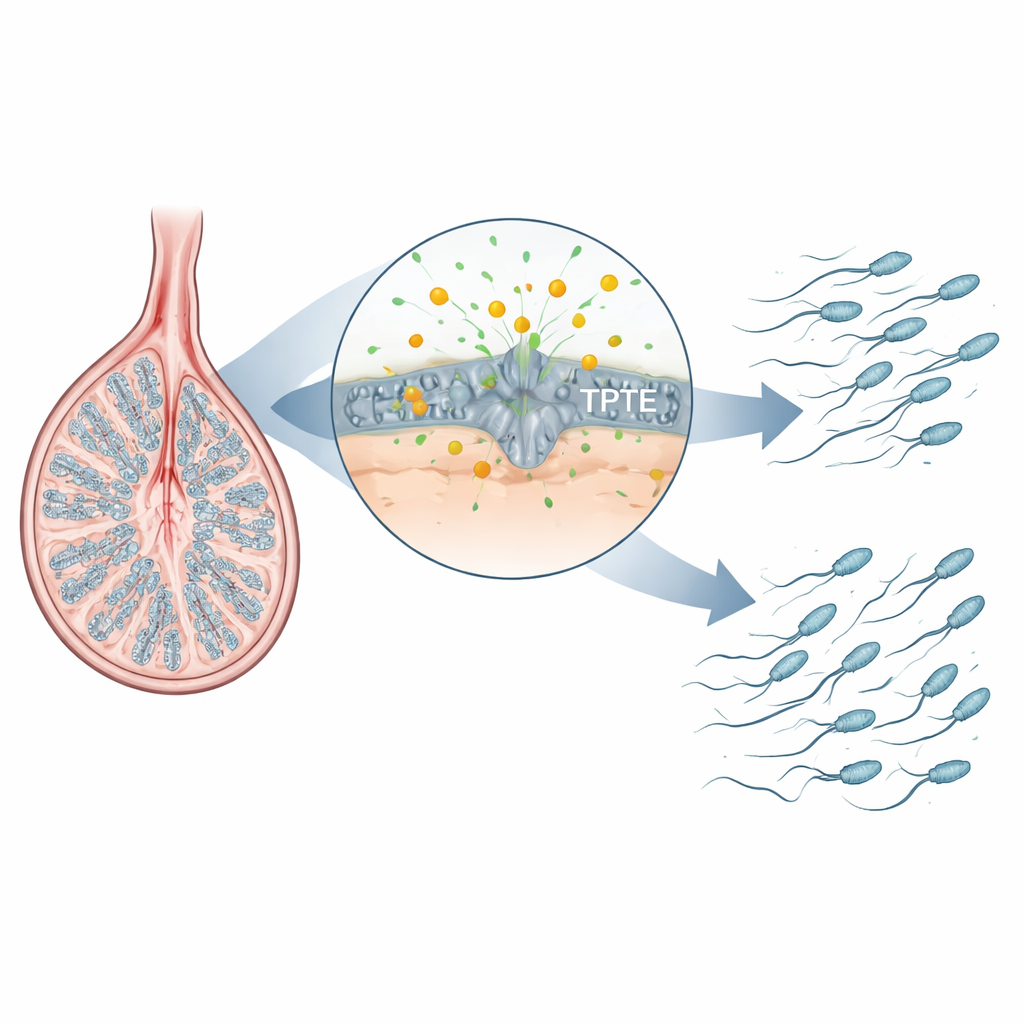
A Hidden Helper in Sperm Development
Sperm are made in a tightly choreographed sequence of steps inside the testis: stem cells divide, their descendants reshuffle DNA during meiosis, and the resulting round cells remodel into sleek, tailed sperm. The authors focused on a family of enzymes that process fatty signaling molecules in cell membranes. One well-known member, PTEN, is important in many tissues but is not essential for the later stages of sperm development. TPTE, however, is produced almost exclusively in germ cells of the testis, and its activity was poorly understood. By mapping where a key membrane lipid, PI(4,5)P2, appears in mouse testes, the researchers found it is especially enriched in spermatocytes and spermatids—the very stages where chromosomes are being repaired and tails are assembled—hinting that TPTE and this lipid might be critical for healthy sperm formation.
Linking TPTE Loss to Poor Sperm and Human Infertility
The team first asked whether TPTE is altered in men with severe infertility. By reanalyzing public RNA sequencing data from testicular biopsies, they found that TPTE levels are strikingly reduced in patients with non-obstructive azoospermia and other conditions marked by damaged or depleted germ cells. To probe cause and effect, they deleted the Tpte gene in mice using genome editing. Male mice lacking TPTE produced much smaller litters despite normal testis size. Their epididymides contained fewer sperm, and computer-assisted analysis showed reduced motility and progressive swimming. Under the microscope, many sperm had normal, oval heads but curled or folded tails, suggesting that TPTE is dispensable for building the head but crucial for maintaining sperm numbers and sculpting functional tails.
Guarding DNA Repair During Meiosis
To understand why sperm numbers fell, the researchers zoomed in on meiosis, where chromosomes exchange segments through precisely controlled DNA breaks and repairs. In TPTE-deficient testes, the overall layout of germ cell stages looked similar, but cell counts began to drop at the diplotene spermatocyte stage, and there was a surge of dying cells caught at metaphase I. Chromosome spreads revealed defects in the ladder-like structure that allows paired chromosomes to align, and a marker of DNA breaks, γ-H2AX, lingered abnormally on autosomes, indicating incomplete repair. Fewer foci of DNA-binding helpers RPA2 and DMC1 formed at break sites. Molecular analysis pointed to overactivation of a central growth pathway, AKT/mTOR, in the absence of TPTE, accompanied by lowered levels of RAD50, a core component that initiates double-strand break repair. Together, these findings suggest that TPTE normally generates PI(4,5)P2 to keep AKT/mTOR activity in check, preserving RAD50 levels and allowing meiotic DNA repair to proceed, thereby safeguarding the supply of viable spermatids.
Building Strong Tails Through Controlled Protein Production
TPTE also proved vital for the later remodeling of round spermatids into streamlined, motile sperm. Electron microscopy of TPTE-lacking sperm tails showed multiple axonemal cross-sections trapped in a single membrane and disrupted “9+2” microtubule arrangements, along with malformed supporting fibers. Staining of tail components such as acetylated tubulin and outer dense fiber protein ODF2 revealed patchy, weakened structures, and more elongated spermatids underwent cell death in the testis. Lipid staining showed that PI(4,5)P2 was markedly reduced and unevenly distributed along flagella when TPTE was missing. To see how this disturbance rippled through the cell’s protein-making machinery, the authors performed quantitative proteomics on isolated round spermatids. Hundreds of proteins, including several ribosomal components, were upregulated, and ribosome profiling indicated boosted polysome activity, a sign of heightened translation. Among these, the team highlighted PDLIM1, a known brake on microtubule organization: its protein levels rose sharply without a change in its messenger RNA, and its transcripts shifted into heavy polysome fractions, indicating overtranslation. This pattern again traced back to excessive AKT/mTOR signaling triggered by the altered lipid balance when TPTE is absent.
What This Means for Understanding Male Infertility
Taken together, the study portrays TPTE as a germ cell guardian that tunes membrane lipids to restrain AKT/mTOR signaling, enabling proper DNA repair and orderly protein synthesis. When TPTE is missing or reduced, as observed in several types of infertile human testes, DNA breaks in meiosis are not correctly resolved, some developing sperm die, and survivors often emerge with structurally flawed tails that swim poorly. For non-specialists, the key message is that male fertility depends not only on hormone levels and sperm counts, but also on subtle molecular regulators like TPTE that coordinate genetic integrity and tail architecture. In the future, screening TPTE and its pathway partners may help identify hidden causes of male infertility and inspire targeted therapies that restore balanced signaling in the testis.
Citation: Chen, X., Wang, T., Wu, H. et al. TPTE, a testis-specific PTEN family member, drives spermatogenesis via PI(4,5)P2 synthesis. Cell Death Dis 17, 378 (2026). https://doi.org/10.1038/s41419-026-08614-3
Keywords: male infertility, spermatogenesis, sperm motility, signal transduction, testis-specific genes