Clear Sky Science · en
TMBIM6 enhances dopaminergic neuron survival by modulating the IRE1a pathway in Parkinson’s disease
Why this research matters for people with Parkinson’s
Parkinson’s disease robs people of smooth movement because key brain cells that make dopamine slowly die. This study uncovers a built-in protective protein, called TMBIM6, that helps these vulnerable neurons cope with stress inside their protein-folding machinery. By showing how boosting TMBIM6 can keep neurons alive and improve movement in animal models, the work points to a fresh therapeutic angle beyond simply replacing dopamine.
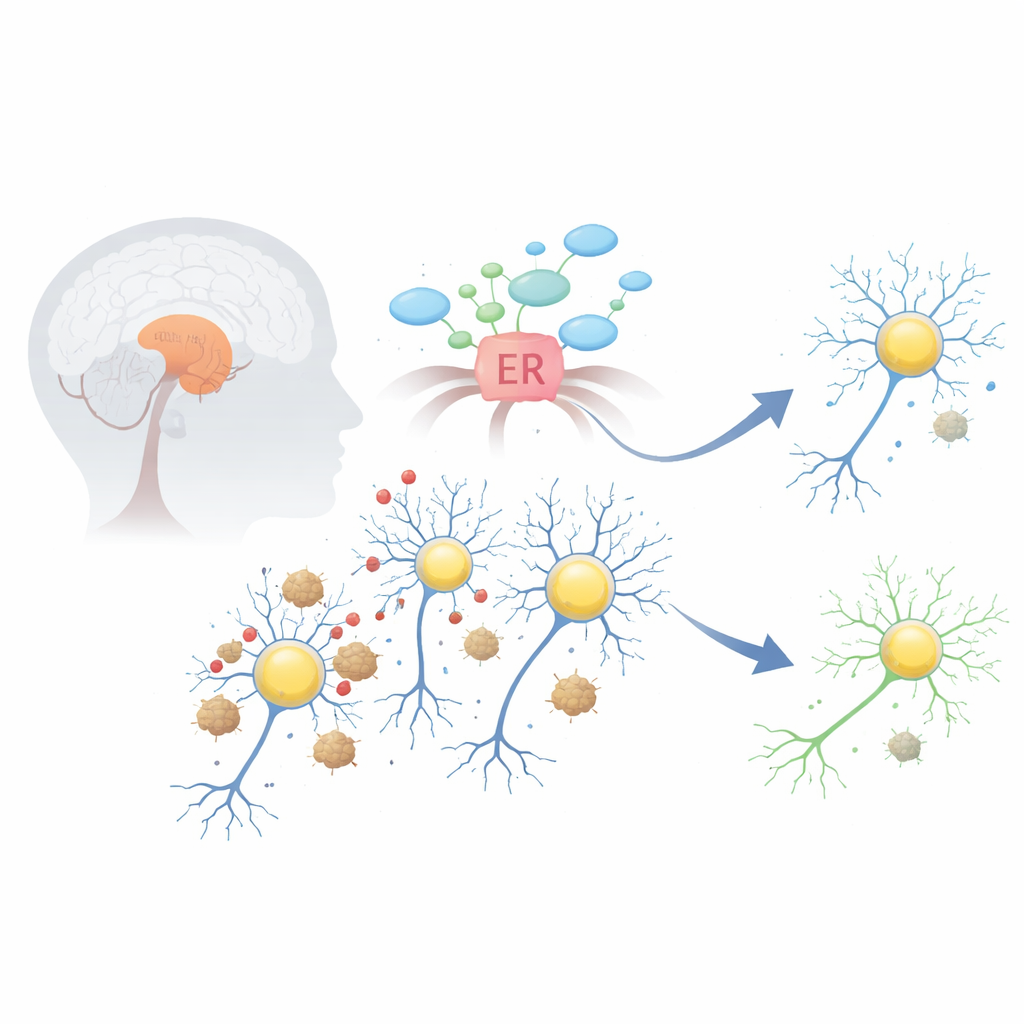
The trouble inside dopamine-producing brain cells
In Parkinson’s disease, a protein named alpha-synuclein can misfold and clump into sticky aggregates, forming structures known as Lewy bodies. These clumps disturb several vital systems within dopamine-producing neurons in a midbrain region called the substantia nigra. One of the earliest and most damaging effects is stress in the endoplasmic reticulum, the cell’s factory for folding and processing proteins. When this compartment is overwhelmed, it switches on an emergency program called the unfolded protein response, which can initially help but, if left on too long, begins to push cells toward self-destruction.
A natural shield: the TMBIM6 safety switch
The protein TMBIM6 sits in the membrane of the endoplasmic reticulum and normally acts as a brake on a key stress sensor called IRE1α. The researchers first asked whether TMBIM6 is present in the right place to matter in Parkinson’s. Using large gene-expression databases, they found that TMBIM6 is highly expressed in the human midbrain and other regions rich in dopamine neurons. In cultured cells and mouse neurons exposed to Parkinson’s-like toxins—such as 6-hydroxydopamine, rotenone, or alpha-synuclein fibrils—TMBIM6 levels rose early on, suggesting that cells turn this factor on as a fast defensive response to stress.
When protection fails in vulnerable neurons
Despite this apparent defense, dopamine neurons still die in Parkinson’s disease. To understand why, the team examined brain tissue and single-cell genetic data from people who had Parkinson’s. Overall, TMBIM6 protein was higher in diseased midbrain tissue than in controls, but a more detailed look revealed a crucial twist: within the very subtype of dopamine neurons known to be most vulnerable, TMBIM6 expression was actually lower than in more resistant neurons. At the same time, the usual tight coordination between TMBIM6 and other stress-response genes was disrupted. Together, these findings suggest that while some brain cells ramp up TMBIM6, the neurons that most need protection may lose this safety switch, leaving them exposed to chronic stress and death signals.
Proving cause and effect in cells and flies
To test whether TMBIM6 truly influences survival, the researchers dialed its levels up and down in several lab models. When they reduced TMBIM6 in dopamine-like mouse cells, these cells became far more sensitive to toxic alpha-synuclein fibrils and to chemical toxins that mimic Parkinson’s. The cells showed greater loss of mitochondrial function, higher activation of executioner enzymes like caspase-3, and more leakage of cell contents—hallmarks of programmed cell death. Similar results appeared in fruit flies engineered to produce less of the TMBIM6 equivalent: they lost dopamine neurons in the eye and brain and developed worse movement problems, especially when exposed to the pesticide rotenone.
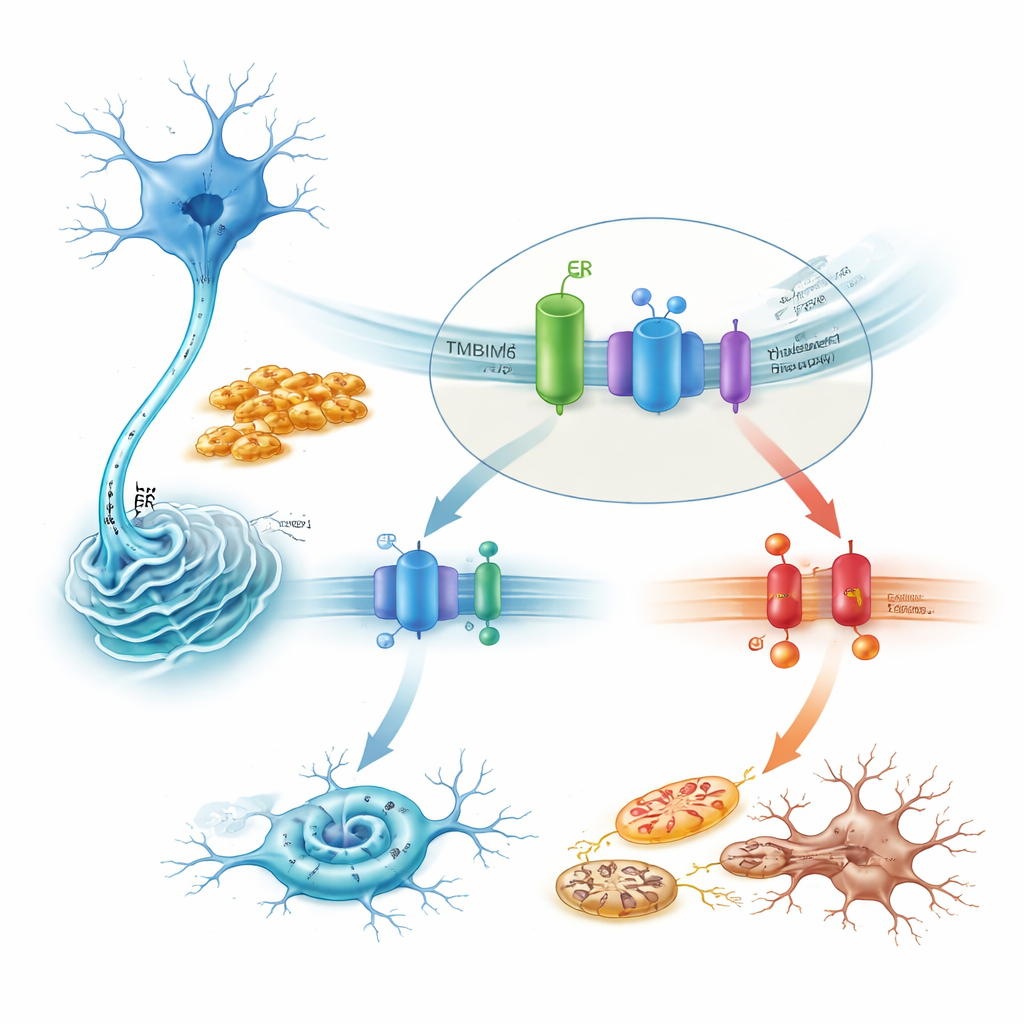
How TMBIM6 tames a lethal stress pathway
Zooming in on mechanism, the study shows that TMBIM6 physically associates with IRE1α under resting conditions. Toxic alpha-synuclein fibrils disrupt this partnership, freeing IRE1α to switch on strongly. In TMBIM6-deficient cells, this activation becomes exaggerated, as shown by increased splicing of the messenger RNA for XBP1 and decay of another IRE1α target. Blocking IRE1α with drugs or by silencing its gene largely rescued the heightened cell death seen when TMBIM6 was missing. Downstream, the harmful pathway ran through the stress kinase JNK, the pro-death protein BAX, and caspases, forming a chain that drives apoptosis. Conversely, overexpressing TMBIM6 dampened IRE1α activity, reduced these death signals, and protected mitochondria, without changing how much alpha-synuclein aggregated—indicating that TMBIM6 acts on the cell’s stress handling rather than on the clumps themselves.
Testing a gene-therapy-like approach in mice
To explore therapeutic potential, the authors used a harmless adeno-associated virus (AAV) to deliver human TMBIM6 directly into the substantia nigra of mice. Two weeks later, they injured the dopamine pathway with a standard Parkinson’s toxin, 6-hydroxydopamine. Control mice that received only a marker gene developed clear motor problems: reduced use of the affected forelimb and poorer balance on a beam test. Mice that had been pretreated with the TMBIM6-carrying virus, however, performed significantly better, making fewer missteps and completing the tasks more quickly. In cultured primary neurons, the same AAV construct eased cell death and caspase activation triggered by toxins or alpha-synuclein fibrils, strengthening the case that boosting TMBIM6 levels enhances neuron resilience.
What this could mean for future treatments
Together, these findings paint TMBIM6 as a guardian of dopamine neurons that works by keeping a dangerous stress sensor, IRE1α, under control and preventing a cascade of events leading to mitochondrial failure and apoptosis. In early disease, cells seem to raise TMBIM6 as a coping strategy, but in the most at-risk neurons the system ultimately falters, tipping the balance toward chronic stress and cell death. Restoring or amplifying TMBIM6—through drugs that enhance its function or gene therapies like the AAV approach tested here—could offer a way to slow or prevent neuron loss in Parkinson’s disease, complementing current treatments that mainly replace dopamine rather than protect the cells that make it.
Citation: Ahumada-Montalva, P., Muñoz-Carvajal, F., Bórquez-Macaya, S. et al. TMBIM6 enhances dopaminergic neuron survival by modulating the IRE1a pathway in Parkinson’s disease. Cell Death Dis 17, 385 (2026). https://doi.org/10.1038/s41419-025-08391-5
Keywords: Parkinson’s disease, dopaminergic neurons, endoplasmic reticulum stress, IRE1α pathway, neuroprotection