Clear Sky Science · en
Neuroinflammation and NeuroHIV: understanding the role of HIV-1 related factors in microglial activation
Why HIV Still Troubles the Brain
Decades after the discovery of HIV, many people living with the virus still struggle with memory problems, slower thinking, mood changes, and movement difficulties. These issues, grouped under the term “NeuroHIV,” persist even in the era of powerful antiretroviral drugs. This review article examines why the brain remains vulnerable, focusing on microglia—the brain’s resident immune cells—and how lingering HIV-related molecules can keep them in a chronic, damaging state of activation.
The Brain’s Watchdogs Turned Against It
Microglia normally act as the brain’s janitors and sentinels. They clear debris, sculpt connections between nerve cells, and respond quickly to infection or injury. Because microglia naturally express the receptors that HIV uses to enter cells and can live for years, they become long-term sanctuaries for the virus inside the central nervous system. Even when antiretroviral therapy suppresses active viral replication in the blood, infected microglia can continue to harbor HIV’s genetic material and produce viral proteins and RNA. These lingering viral products, along with small regulatory RNAs and tiny membrane packages called extracellular vesicles, can nudge microglia from helpful guardians into chronic troublemakers.
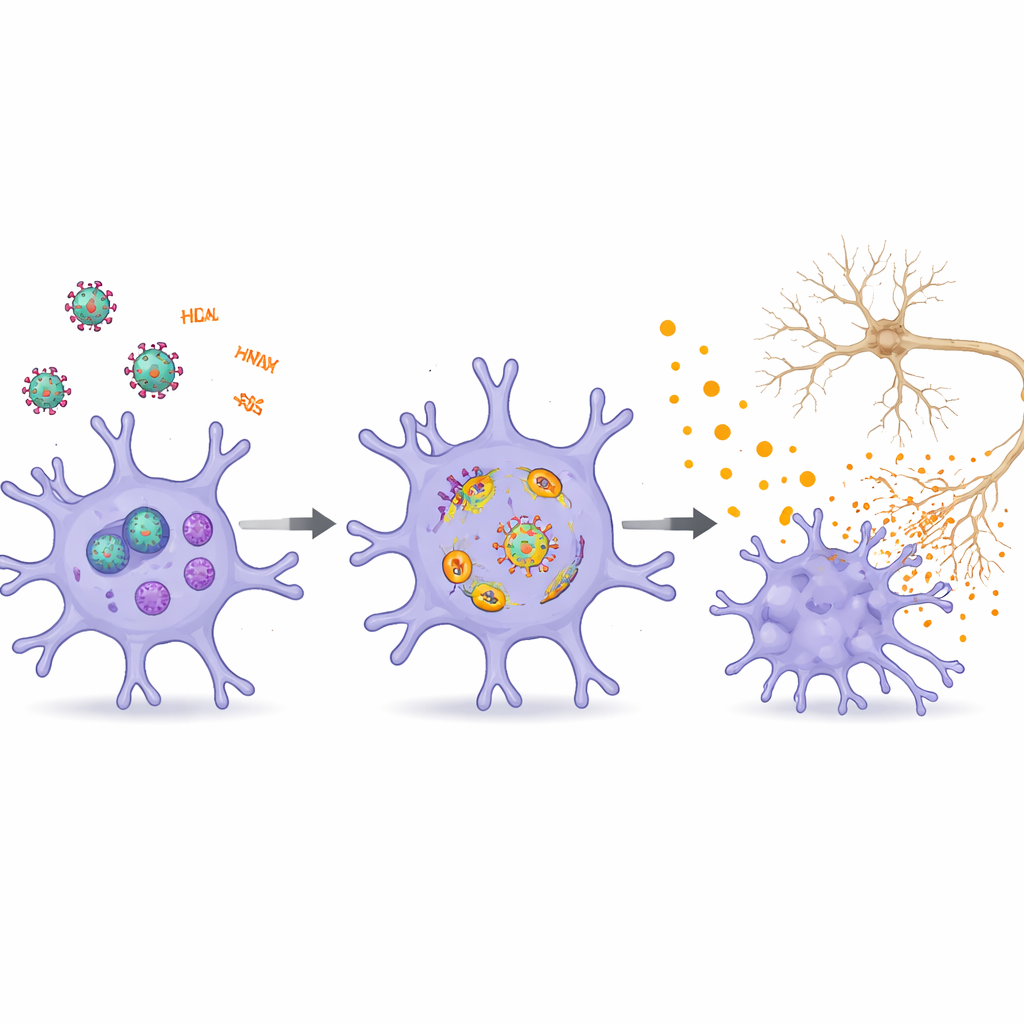
How Viral Fragments Stoke Ongoing Brain Inflammation
The authors describe how specific HIV components—especially the proteins Tat, gp120, Vpr, Nef, and various viral RNAs—repeatedly push microglia into an inflammatory mode. Tat and gp120, released from infected cells, bind to receptors on microglia and disturb calcium balance, mitochondrial health, and cellular recycling systems. This sets off internal danger sensors that trigger the release of chemical messengers capable of damaging neighboring neurons. Viral RNA fragments, even when they do not lead to full viral replication, are detected as alarm signals inside microglia. They activate innate immune pathways and inflammasome complexes that behave like molecular tripwires, culminating in bursts of inflammatory molecules and sometimes a fiery form of cell death that further fuels local damage.
Cell Stress, Iron, and Aging Inside Microglia
Beyond simple “on–off” immune activation, the review highlights deeper cellular changes that make microglia more toxic over time. Tat can drive microglia into a senescence-like state that resembles accelerated aging: these cells divide less, accumulate oxidative damage, and secrete higher levels of inflammatory factors. Tat also promotes a specialized form of cell death called ferroptosis, which depends on iron and runaway lipid damage and is increasingly linked to neurodegenerative diseases. At the same time, Tat disrupts mitophagy—the selective removal of worn-out mitochondria—and broader autophagy, the cell’s waste-disposal system. When these cleanup processes fail, damaged mitochondria and lipids build up, reactive molecules rise, and key inflammatory pathways such as NF-κB and the NLRP3 inflammasome are easier to activate, locking microglia into a self-sustaining harmful loop.
Crosstalk Among Brain Cells Spreads the Harm
The review places microglia within a larger network of brain cells influenced by HIV. Viral proteins and RNAs injure neurons directly by disturbing energy production, endolysosomal trafficking, and the delicate branches that support synapses, contributing to memory and learning problems. They weaken the blood–brain barrier, allowing more immune cells and toxic signals to enter the brain. Astrocytes and oligodendrocytes, which support neurons and maintain white matter, also respond to HIV factors by becoming inflamed, senescent, or prone to cell death. Extracellular vesicles shuttle viral proteins and regulatory RNAs between these cell types, amplifying distress signals and reinforcing cycles of neuroinflammation and network dysfunction.
New Paths Toward Calmer Brains
To a layperson, the key message is that NeuroHIV is not simply caused by active viral growth, but by a lingering “molecular noise” from HIV that keeps microglia and other brain cells on high alert for years. This chronic, smoldering inflammation gradually erodes brain health. By mapping the main molecular hubs—such as inflammasome components, NF-κB signaling, defective cellular recycling, iron-driven damage, and specific microRNAs—the authors outline promising targets for future therapies that go beyond standard antiretroviral drugs. Such treatments would aim to silence viral activity within brain reservoirs, restore healthy microglial behavior, and interrupt the vicious feedback loops of inflammation, offering hope for preserving cognition and quality of life in people living with HIV.
Citation: Zhao, J., Bu, F., Wu, H. et al. Neuroinflammation and NeuroHIV: understanding the role of HIV-1 related factors in microglial activation. Transl Psychiatry 16, 194 (2026). https://doi.org/10.1038/s41398-026-03941-7
Keywords: NeuroHIV, microglia, neuroinflammation, HIV brain complications, HIV reservoirs