Clear Sky Science · en
Platelets cause microvascular occlusion and delayed neurological deficits after subarachnoid hemorrhage in mice
When a Brain Bleed Strikes Twice
Surviving a burst brain aneurysm is only the first hurdle for many patients. Days after the initial bleed, roughly half develop new, often devastating, neurological problems such as confusion, weakness, or stroke-like symptoms. Doctors call these delayed neurological deficits, and until now, the exact trigger has remained murky. This study in mice, with supporting tests on human blood, pinpoints a surprising villain: tiny blood cells called platelets that normally help stop bleeding but, in this setting, appear to clog the brain’s finest vessels and choke off vital blood flow.
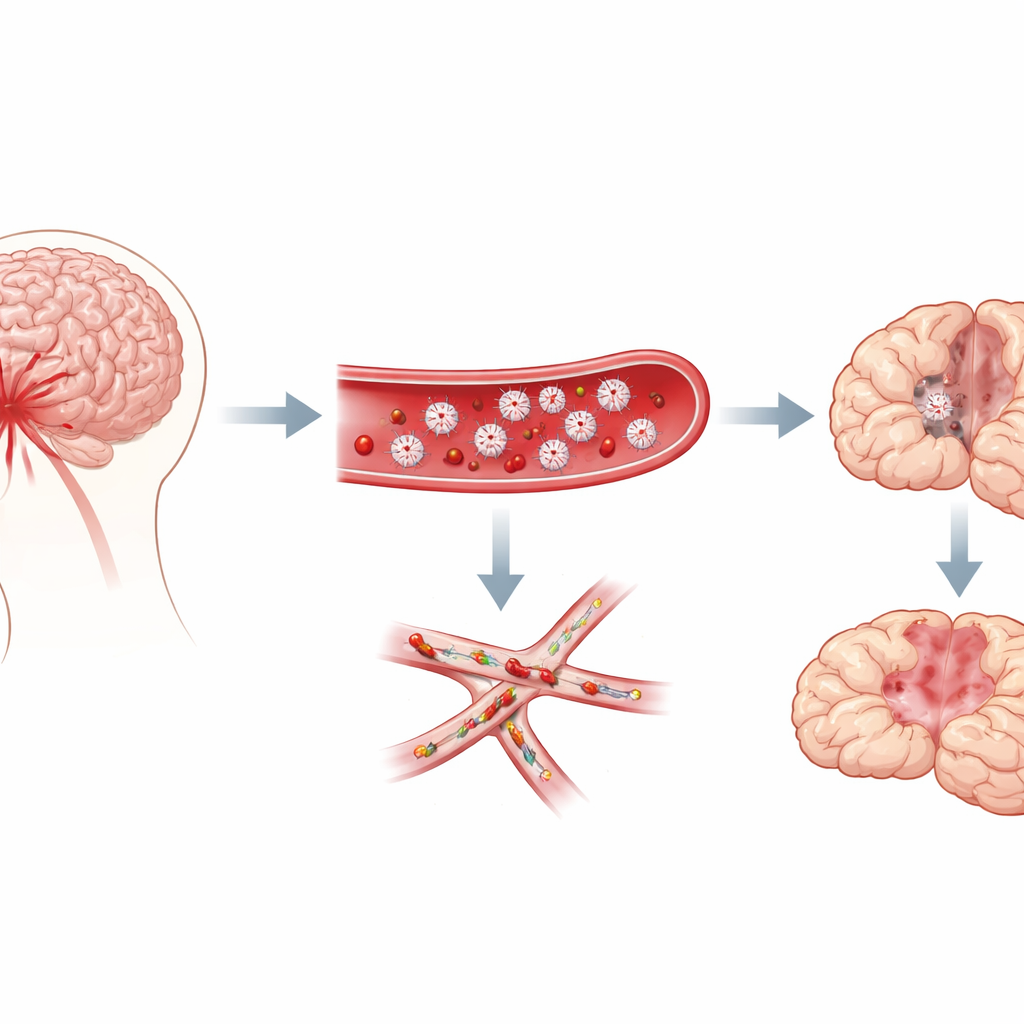
Tiny Clots in the Brain’s Smallest Vessels
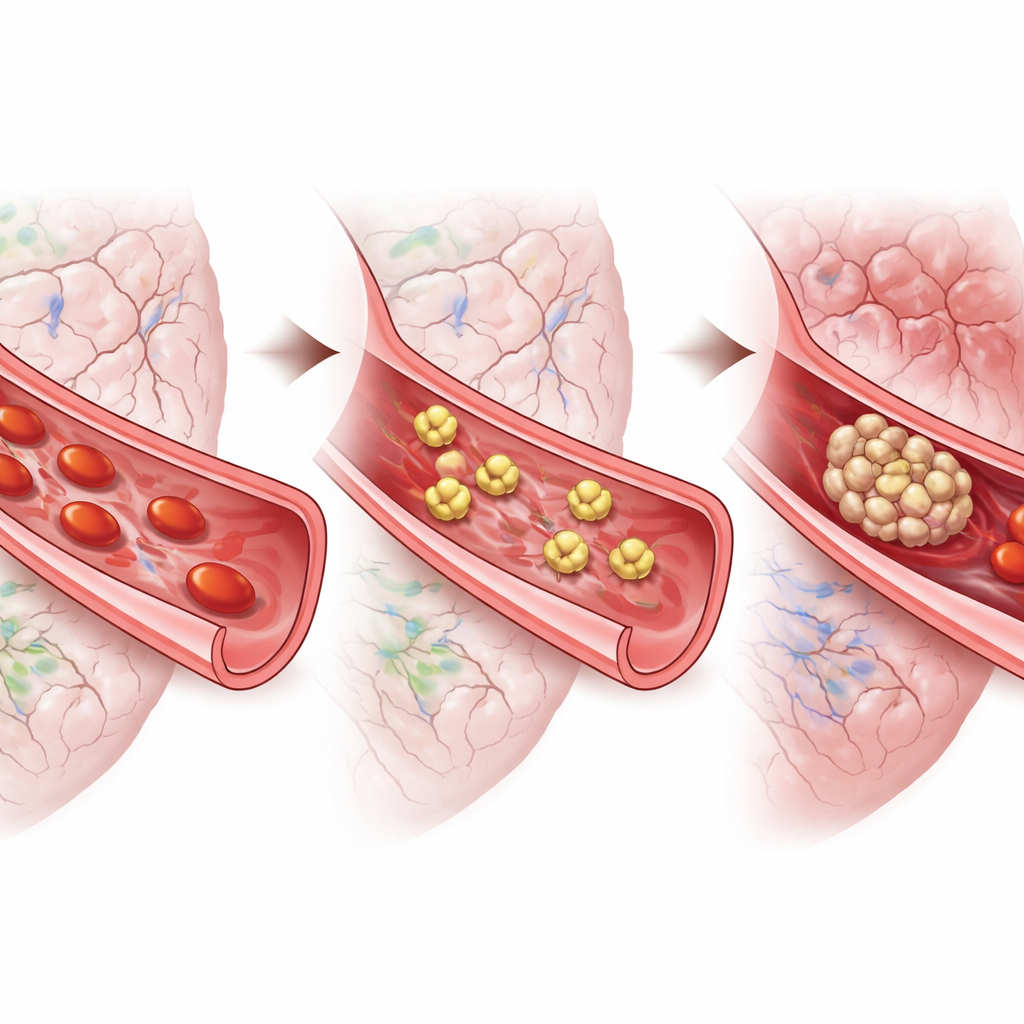
The researchers focused on subarachnoid hemorrhage, a kind of bleeding that floods the space around the brain after an aneurysm ruptures. Clinicians have long noticed that patients who go on to have delayed problems often show signs that their blood is unusually prone to clotting, and autopsies reveal countless “microthrombi”—pinpoint clots—peppering the brain. These clots are rich in platelets. Yet it was unclear whether platelets actually cause the delayed damage or merely show up as bystanders. Using a well-established mouse model of this hemorrhage, the team tracked both behavior and brain pathology over a week, asking whether platelet activity lined up with the onset of delayed deficits.
Switching Platelets On and Off
In the first set of experiments, the scientists measured chemical signals in the blood that are known to rouse platelets. Two, called PAF and TXB2, rose sharply on day one and again around day five—mirroring the high‑risk window when delayed problems typically appear. To test cause and effect, they then pushed platelets harder. When mice received extra PAF soon after the bleed, many died and their brains filled with more microthrombi. Giving PAF a few days later, during the “quiet” recovery phase, did not kill the animals but did cause worse neurological scores, more delayed deficits, and heavier microclot burdens. Mice genetically prone to delayed platelet activation showed a similar pattern: ordinary early recovery followed by a higher rate of late‑appearing deficits.
Removing or Calming Platelets
If cranking platelets up made things worse, could dialing them down help? To find out, the team used engineered mice whose platelets could be selectively removed with a toxin given before the hemorrhage. These animals did not bleed more or die more often, but they had fewer microthrombi, better early neurological function, and a markedly lower chance of delayed decline than normal mice. Because stripping platelets altogether would be unsafe in people, the researchers also tested several existing drugs that block different platelet receptors—the docking points through which various signals activate these cells or make them stick together. Many of these drugs improved early behavior and cut down microclots, but only one, tirofiban, which blocks a key “aggregation” receptor called GPIIb/IIIa, clearly reduced the long‑term rate of delayed deficits in female mice.
From Mice to People
To bridge the gap to human disease, the investigators collected platelets from patients who had suffered an aneurysmal subarachnoid hemorrhage. Under the microscope, these cells were highly activated and spread out, consistent with a hair‑trigger clotting state. Adding tirofiban to the samples made the platelets far less likely to spread, both in the first days after the bleed and later during the high‑risk window for delayed complications. Another drug that targeted a different platelet receptor had little effect. Together with the mouse results, these tests suggest that blocking platelet clumping, rather than merely muting some activation signals, may be the most promising strategy.
What This Means for Future Treatment
This work builds a strong case that platelets do more than plug the initial rupture after a brain bleed: days later, they help form clusters of tiny clots that block the brain’s smallest vessels and set the stage for delayed strokes. In mice, taking platelets away or blocking their main “Velcro” receptor sharply reduced these delayed injuries, and a drug already used in other settings, tirofiban, also tamed overactive platelets from human patients in the lab. While any antiplatelet treatment must be balanced against the risk of more bleeding, the study offers a clear proof‑of‑concept that carefully targeted platelet‑blocking drugs could one day help protect survivors of aneurysmal subarachnoid hemorrhage from a dangerous second wave of brain damage.
Citation: Dienel, A., Hong, SH., Torres, K. et al. Platelets cause microvascular occlusion and delayed neurological deficits after subarachnoid hemorrhage in mice. Exp Mol Med 58, 1242–1253 (2026). https://doi.org/10.1038/s12276-026-01696-1
Keywords: subarachnoid hemorrhage, platelets, microthrombi, delayed cerebral ischemia, antiplatelet therapy