Clear Sky Science · en
NV716 acts as an envelope-active adjuvant that enhances antibiotic accumulation in Pseudomonas aeruginosa
Why this research matters for future antibiotics
Drug-resistant bacterial infections are rising faster than new antibiotics can be discovered, and some of the toughest culprits are Gram‑negative bacteria like Pseudomonas aeruginosa. These microbes surround themselves with a formidable outer shell that keeps many medicines out. This study explores a helper compound, called NV716, that does not kill bacteria by itself but makes them far more vulnerable to certain existing antibiotics. Understanding how such a helper works could open a practical route to rescuing drugs we already have instead of waiting decades for entirely new ones.
A chemical partner that helps antibiotics get inside
The researchers focused on NV716, a positively charged, membrane-seeking molecule previously known to boost the activity of several antibiotics against Gram‑negative bacteria. They tested NV716 together with drugs from different families, such as tetracyclines and fluoroquinolones, against P. aeruginosa and Escherichia coli. The most dramatic effect was seen with the antibiotic doxycycline: when combined with NV716, the amount of doxycycline needed to halt bacterial growth dropped by more than one hundredfold in P. aeruginosa. By measuring antibiotic fluorescence inside bacterial cells, the team showed that this boost in killing power tracks closely with a surge in how much doxycycline actually accumulates within the microbes.
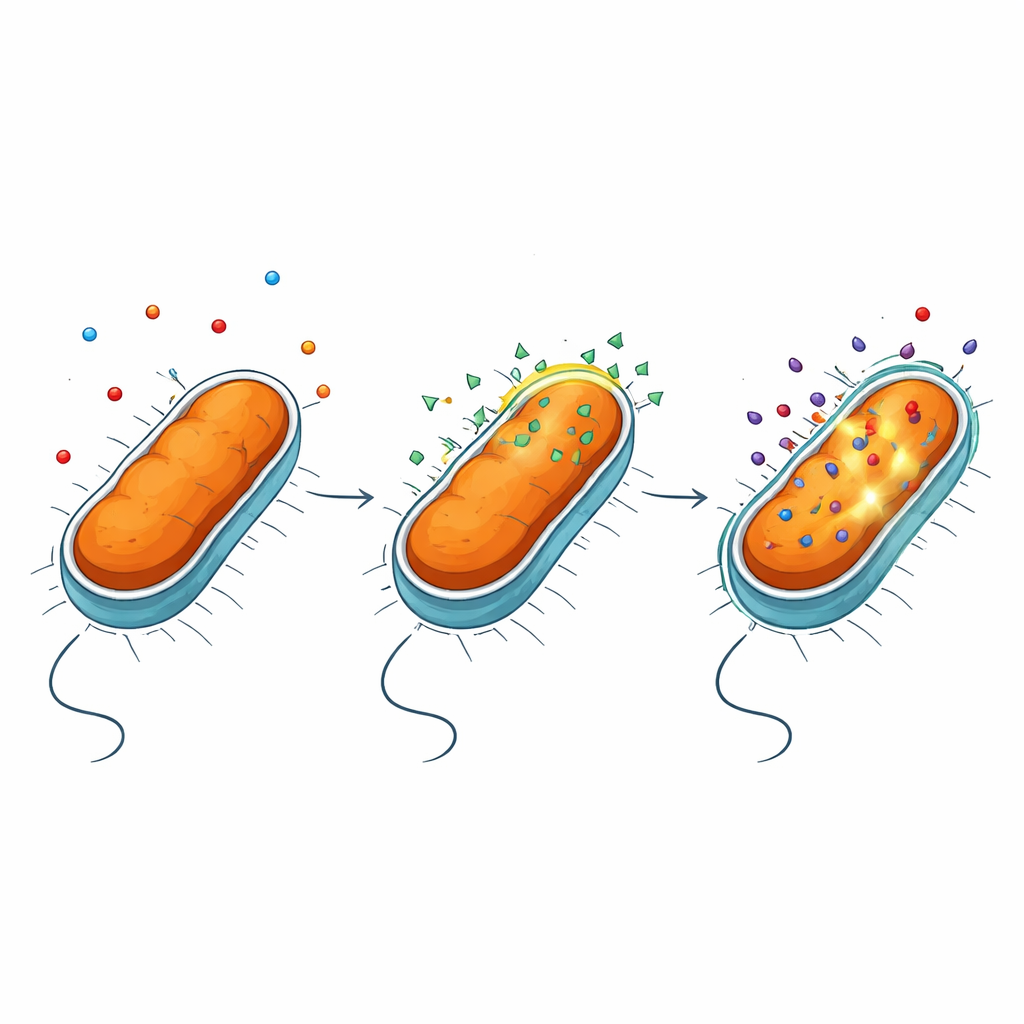
Opening the outer shield without rupturing the cell
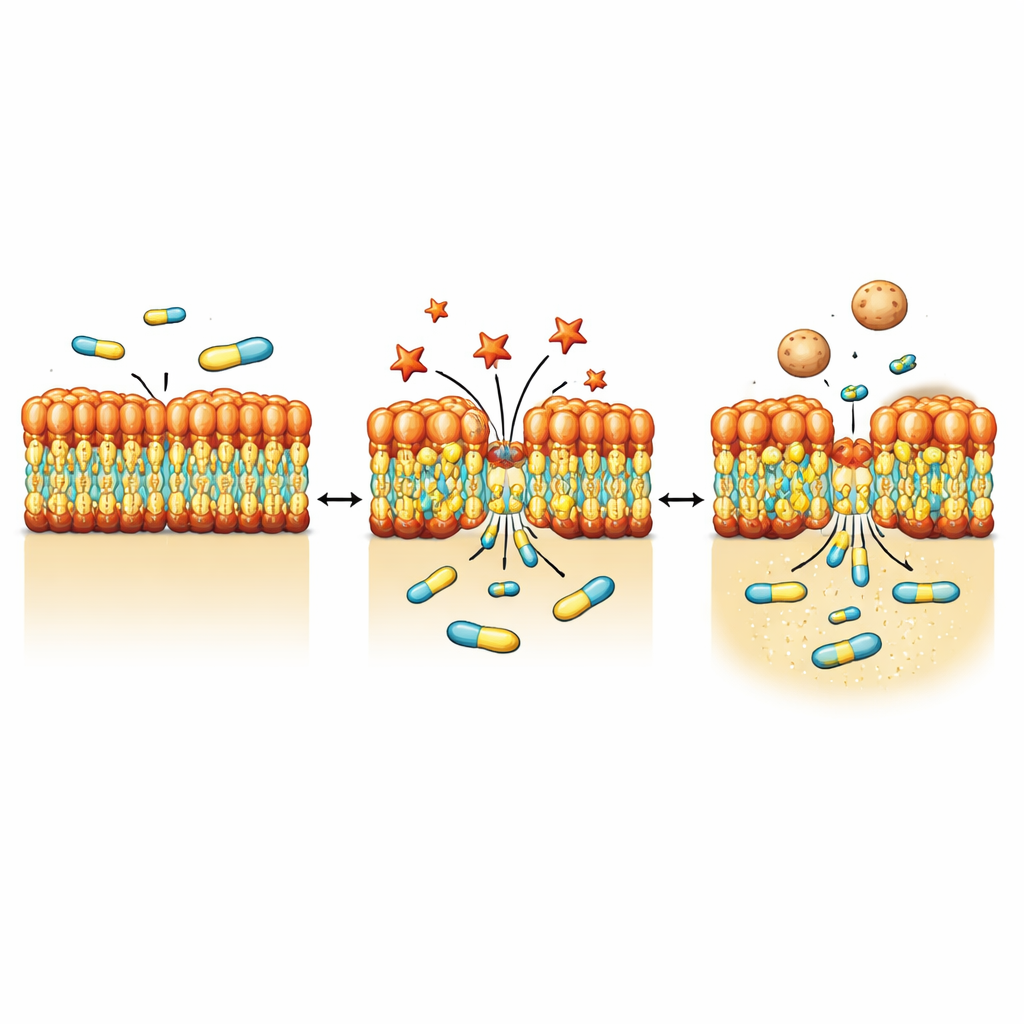
Gram‑negative bacteria are protected by an outer membrane rich in a complex molecule called lipopolysaccharide, or LPS. This layer acts like a highly selective filter, blocking many drugs. Using a series of fluorescence assays and mutant strains, the authors found that NV716 does not noticeably shut down the bacteria’s efflux pumps—the molecular machines that expel antibiotics. Instead, NV716 alters the behavior of the outer membrane itself. It binds to LPS at the cell surface and loosens the packing of these molecules, especially when the sugar “core” of LPS is shortened and the underlying lipid portion is more exposed. This controlled disturbance makes the membrane easier for doxycycline and some other antibiotics to cross, without tearing open the cell or damaging the inner membrane where vital processes occur.
Visualizing membrane stress and bubble-like shedding
To see how bacteria physically respond to NV716, the team turned to high‑resolution imaging methods including electron microscopy and soft X‑ray tomography. After exposure to NV716, P. aeruginosa showed small protrusions and patches bud off from the surface, forming numerous outer membrane vesicles—tiny spherical bubbles made from the same material as the outer shell. Population‑scale measurements confirmed that vesicle production rose by roughly an order of magnitude. Detailed imaging and protein analyses indicated that these vesicles carry outer membrane components such as porin proteins and LPS, but little detectable DNA, consistent with them budding from the surface rather than bursting from within. At the same time, a copper‑tagged version of NV716 revealed that the compound itself concentrates in the membrane region rather than freely diffusing through the cell interior.
From lab dishes to living hosts
Because strong effects in test tubes do not always translate to living organisms, the researchers assessed the NV716–doxycycline pairing in an infection model using larvae of the wax moth Galleria mellonella. Larvae treated with the combination showed a trend toward better survival compared with doxycycline alone, although the improvement was not statistically significant under the conditions tested. Computer‑based predictions of drug‑like properties suggest that NV716 has features consistent with interacting with membranes and may be tolerable in animals, but thorough safety and dosing studies remain to be done. The authors view NV716 mainly as a proof‑of‑concept: a molecule that demonstrates what is possible when the bacterial envelope is selectively modulated rather than destroyed.
What this means for tackling tough infections
In everyday terms, this work shows that it is possible to "pick the lock" of a hard‑to‑penetrate bacterium like P. aeruginosa without smashing the door down. NV716 nestles into the microbe’s outer shell, subtly rearranging its structure so that certain antibiotics, especially doxycycline, can slip inside more quickly and in greater amounts. The bacteria respond by shedding extra bits of their outer coat in the form of vesicles—signs of stress but not outright collapse. Although the first animal tests are modest, the study provides a quantitative blueprint for designing new helper molecules and hybrid drugs that target the outer membrane. Such strategies could extend the useful life of existing antibiotics and give clinicians new options against some of the most stubborn hospital infections.
Citation: Draveny, M., Chauvet, H., De Pauw, A. et al. NV716 acts as an envelope-active adjuvant that enhances antibiotic accumulation in Pseudomonas aeruginosa. npj Antimicrob Resist 4, 29 (2026). https://doi.org/10.1038/s44259-026-00203-w
Keywords: antimicrobial resistance, Pseudomonas aeruginosa, outer membrane, antibiotic adjuvant, doxycycline