Clear Sky Science · en
Safe focused ultrasound-mediated blood-brain barrier opening is driven primarily by transient reorganization of tight junctions
Why opening the brain’s gate matters
Many of the most promising treatments for Alzheimer’s disease, brain cancers, and other neurological conditions never reach their targets, because the brain is protected by a microscopic gatekeeper called the blood–brain barrier. This barrier shields delicate nerve tissue from harmful substances in the blood, but it also blocks most helpful drugs. The study summarized here explores a noninvasive way to open this barrier briefly with focused sound waves, revealing what happens to the brain’s blood vessels when the approach is used gently versus too aggressively.
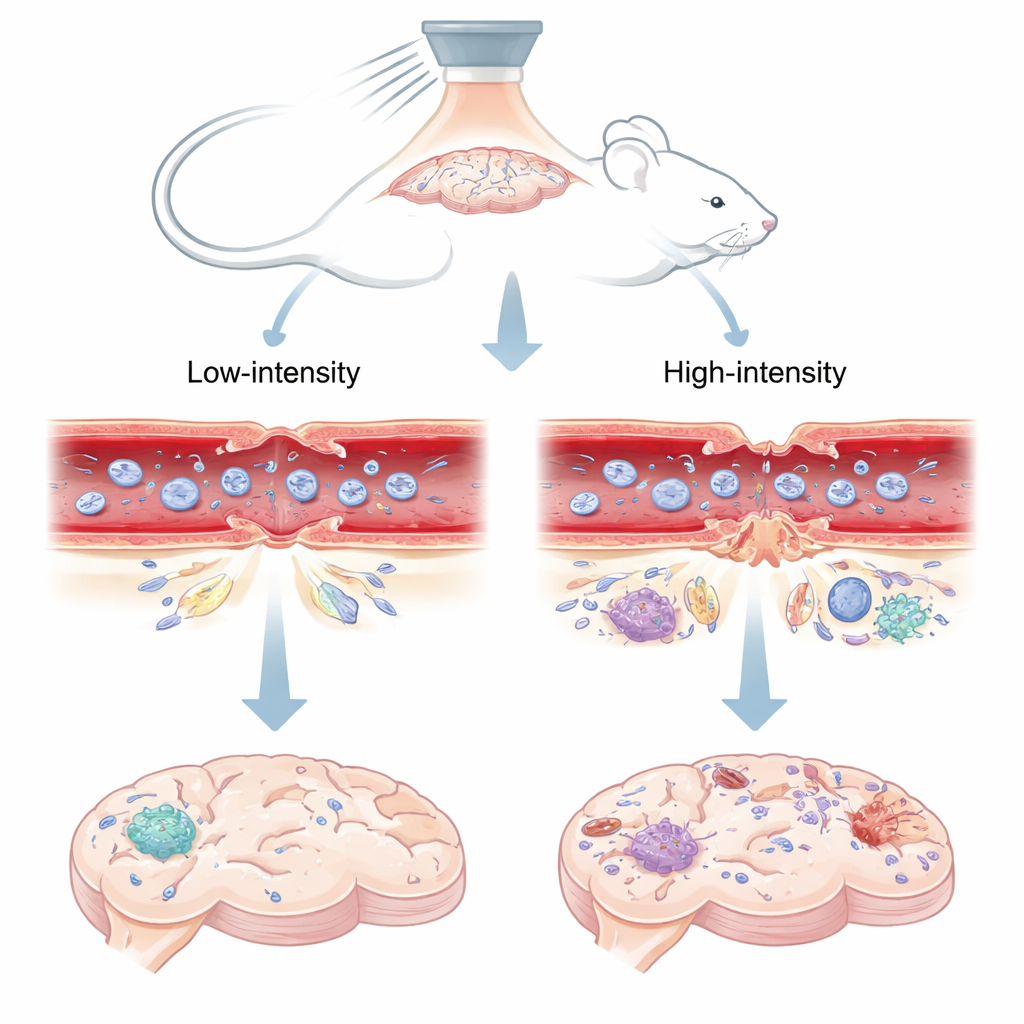
A sound-powered doorway into the brain
Researchers used focused ultrasound – concentrated sound waves from outside the skull – combined with tiny gas-filled bubbles injected into the bloodstream. When the sound waves hit these microbubbles inside brain blood vessels, the bubbles vibrate. At lower sound pressures, the bubbles wobble in a controlled way; at higher pressures, they can violently collapse. The team tested two levels in mice: a "low" setting known from earlier work to open the barrier safely and briefly, and a "high" setting associated with longer-lasting leakage and tissue damage. They then watched how a fluorescent dye and MRI contrast agent spread into the brain, which revealed when and where the barrier had opened and when it resealed.
Microscopic gates that shift or shatter
The blood–brain barrier is mainly formed by tight junctions – protein “zippers” that seal neighboring cells lining brain blood vessels. In special mice whose tight junctions glow under the microscope, the scientists could watch these zippers change shape after ultrasound. At the safe, low sound pressure, the junctions showed small, temporary gaps in both tiny capillaries and slightly larger arterioles. Within about three days, those gaps largely closed and the pattern of junctions looked much like the untouched side of the brain, matching the fact that dye and MRI contrast no longer leaked in. At the higher pressure, however, junctions were not just gapped but sometimes entirely missing, especially in larger vessels. These severe breaks persisted for at least 72 hours and were accompanied by continued leakage of tracer molecules.
Brain immune cells and blood proteins join the story
Opening the barrier also lets blood proteins enter brain tissue. One such protein, fibrinogen, plays roles in clotting and inflammation and is linked to damage in several brain diseases. Shortly after ultrasound at both pressures, fibrinogen appeared near leaky vessels alongside activated microglia, the brain’s resident immune cells. The microglia changed from a resting, branched appearance to a rounder, “on-alert” form. By 72 hours, fibrinogen was no longer detectable for either pressure, suggesting that even when junction damage persisted at the higher setting, some aspects of the leak had resolved. The researchers also tested mice lacking caveolae, tiny pockets in the vessel wall that can ferry molecules across cells. Barrier opening at the safe pressure was unchanged without these structures, pointing to the rearrangement of tight junctions – not an increase in this transport route – as the main mechanism for controlled opening.
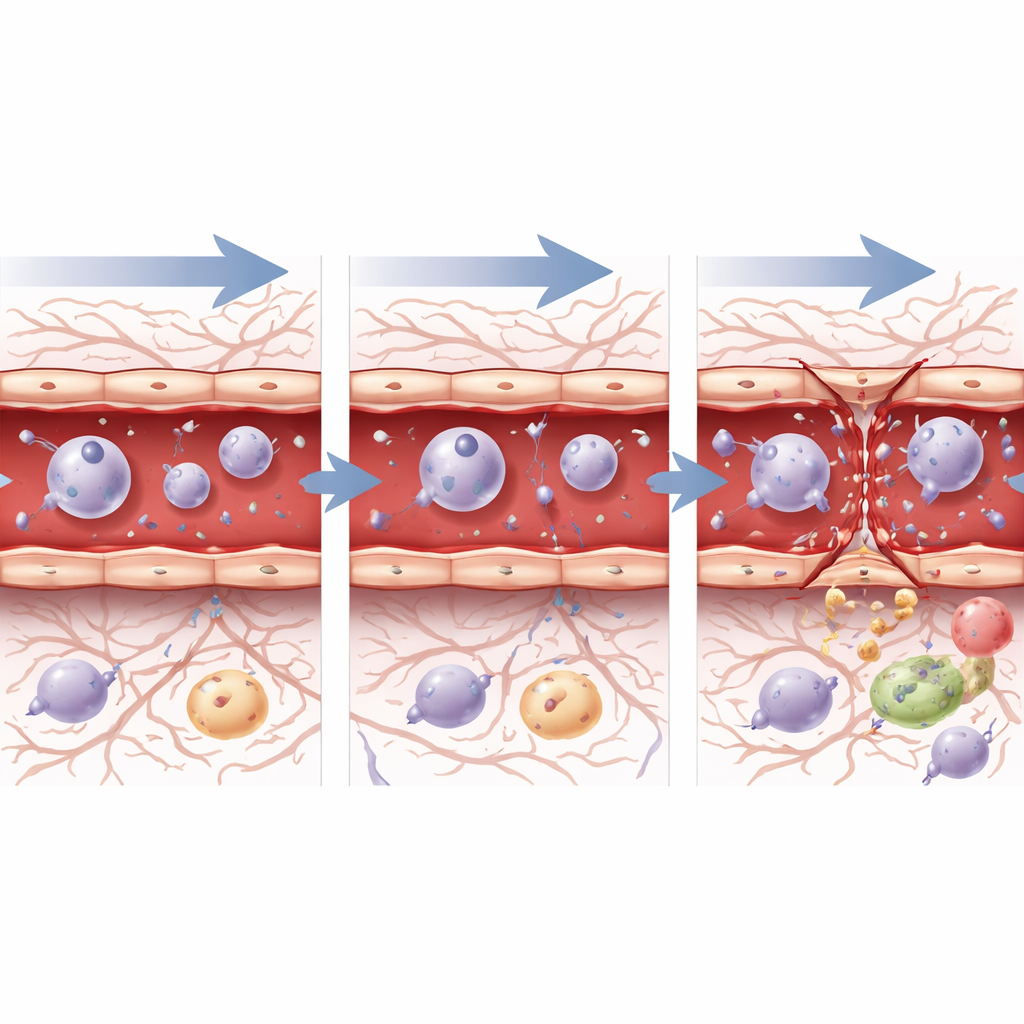
How blood vessel cells respond from within
To see how individual blood vessel cells react at the genetic level, the team isolated thousands of endothelial cells – the cells lining arteries, capillaries, and veins – and performed single-cell RNA sequencing. Within an hour of ultrasound, regardless of pressure, these cells ramped up genes linked to stress, cell death, inflammation, and repair, signaling that they sensed both injury and the need for recovery. By 72 hours, the picture diverged. After the safe, low pressure, cells showed gene activity consistent with wound healing, controlled cell growth, and gradual return toward a stable barrier state. After the higher pressure, however, cells still strongly expressed stress and “wound response” programs, and key junction-building genes remained suppressed, in line with the ongoing structural damage seen under the microscope. Pathways that guide blood vessel growth and barrier formation, such as TGF-beta, Notch, and Wnt signaling, shifted over time in ways that suggested active but incomplete repair, especially after the more aggressive setting.
What this means for future brain treatments
For people hoping for better brain therapies, this work clarifies a central safety question: how can we open the blood–brain barrier enough to let drugs in, without causing lasting harm? The study shows that at carefully chosen, lower ultrasound pressures, the barrier opens mainly because its protein “zippers” briefly loosen and then reorganize, with blood vessel cells engaging repair programs that close the gaps within days. Pushing the system harder shatters those zippers instead of merely shifting them, leaving prolonged leaks and prolonged cellular distress. In practical terms, the findings support using conservative ultrasound settings and careful monitoring in clinical applications, reinforcing the idea that safe brain drug delivery depends on transient reorganization – not destruction – of the brain’s microscopic gates.
Citation: Noel, R.L., Kugelman, T., Karakatsani, M.E. et al. Safe focused ultrasound-mediated blood-brain barrier opening is driven primarily by transient reorganization of tight junctions. Commun Eng 5, 58 (2026). https://doi.org/10.1038/s44172-026-00597-5
Keywords: focused ultrasound, blood-brain barrier, drug delivery, brain blood vessels, microbubbles