Clear Sky Science · en
Early transcriptional divergence underlies cell fate bias in bovine embryos
How early life’s first choices may shape a calf
Every mammal, including humans and cattle, starts life as a single cell that repeatedly divides. For decades, biologists assumed that the earliest cells in this tiny ball were all equal, each with the same potential to become any part of the future body or its supporting tissues. This study on cattle embryos challenges that simple picture, showing that some cells begin to drift toward particular futures far earlier than they look different under the microscope.
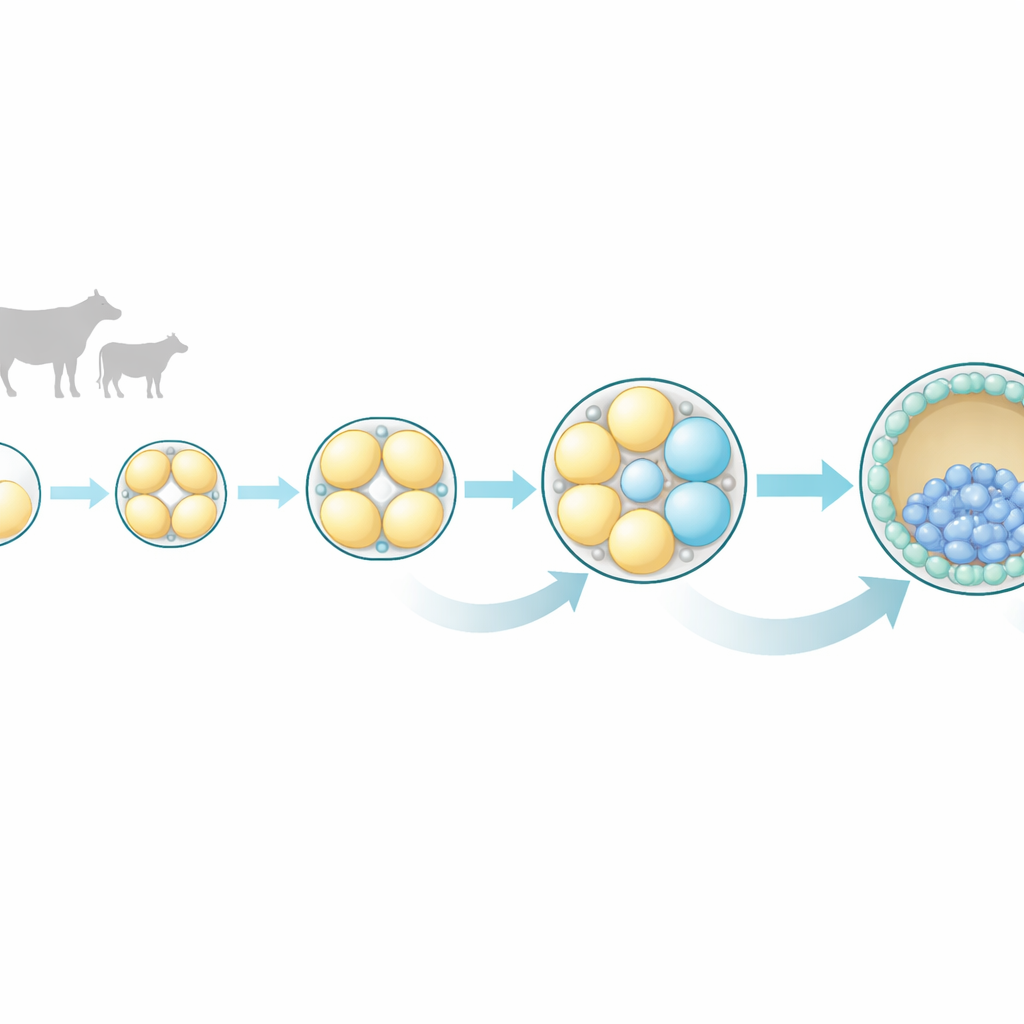
Early cell divisions are not as equal as they seem
The researchers focused on bovine embryos because their early development closely resembles that of humans in timing and pattern. They collected embryos at four very early stages: the single-cell zygote, and the 2-cell, 4-cell, and 8-cell stages. From each embryo, they carefully separated all individual cells and measured which genes were active in each one using single-cell RNA sequencing, a technique that reveals a cell’s molecular “to-do list.” This approach let them compare sister cells from the same embryo and ask how similar or different they really were as development progressed.
Growing differences appear by the 4-cell and 8-cell stages
At the 2-cell stage, the two sister cells looked nearly identical in their gene activity, supporting the traditional view of equal potential at this point. But by the 4-cell stage, subtle differences began to emerge, and by the 8-cell stage those differences became striking. Cells from the same 8-cell embryo no longer grouped together based on their gene expression profiles; instead, each cell followed its own molecular path. Many of the genes that varied the most were linked to communication pathways inside cells, especially a set known as MAPK, along with related Ras and Wnt signaling pathways. These pathways are already known in other species to guide whether a cell becomes part of the embryo proper or part of the outer layer that forms the placenta.
Hints of future roles in outer versus inner cell layers
The team paid special attention to a gene called CDX2, a classic marker of the outer layer (trophectoderm) that will eventually help form the placenta. At the 8-cell stage, some cells had higher CDX2 levels than their sisters. These CDX2-high cells also showed stronger activity of MAPK-related genes, and the link between CDX2 and these signaling genes grew tighter from the 4-cell to the 8-cell stage. This pattern suggests that, even before the embryo compacts into a smooth ball and positions cells on the outside or inside, some cells are already biased toward becoming part of the outer, placenta-forming layer.
Cell size tips the balance toward supporting tissues
Intriguingly, physical differences between cells matched these molecular biases. Within the same embryo, some blastomeres were larger than others at both the 4-cell and 8-cell stages. Larger cells tended to express more CDX2 and MAPK-related genes. They also showed higher amounts of a protein called YAP in their nuclei, a known driver of outer-layer identity in early embryos. When the scientists followed single large or small cells in culture, embryos derived from larger blastomeres compacted earlier, underwent one extra round of division before compacting, and formed fluid-filled cavities more reliably and stably. When reconstructed embryos were made entirely from the largest or smallest cells, those built from larger cells developed more robustly and produced more outer-layer cells while still maintaining inner, embryo-forming cells.
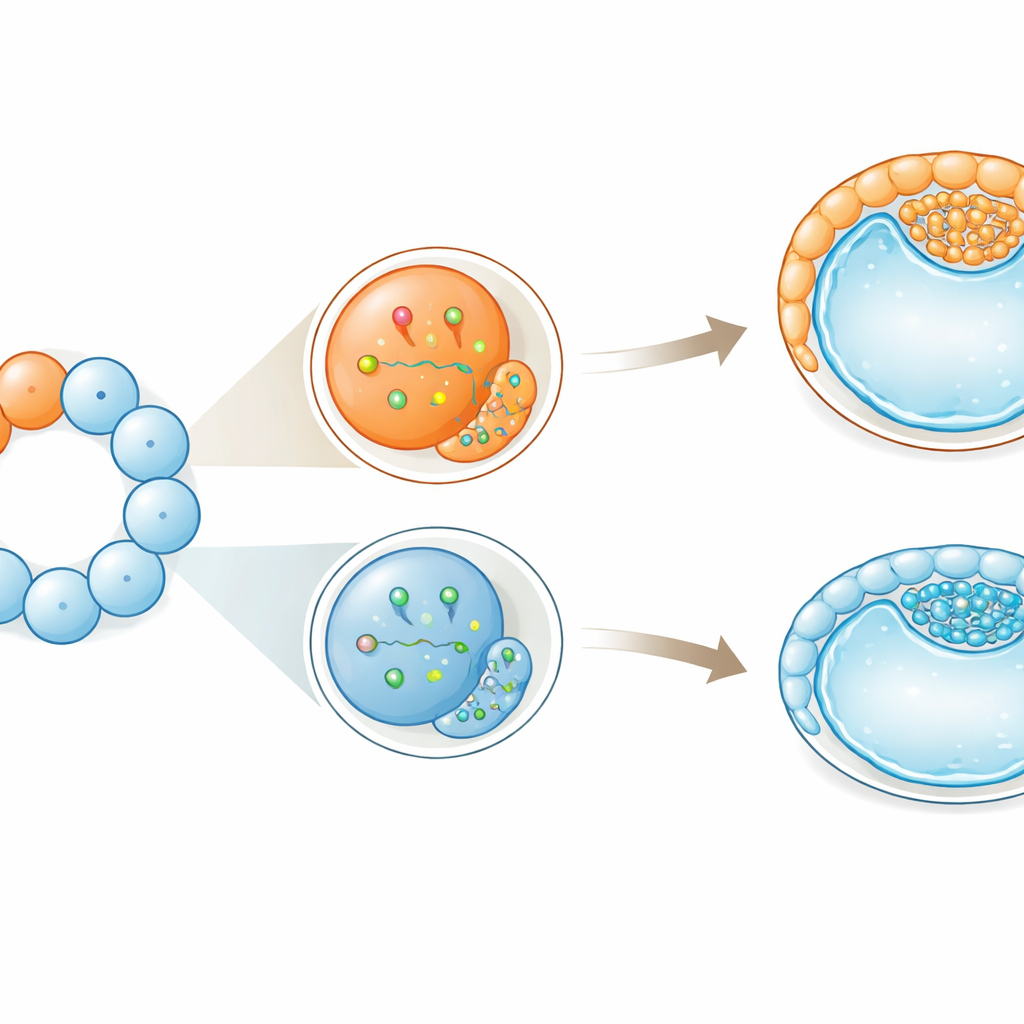
Early bias without loss of flexibility
Despite these early tilts toward particular roles, the cells did not lose their flexibility outright. Even the more “outer-leaning” large cells could still contribute to both the embryo and the supporting tissues. The authors propose that these early differences act like gentle pushes rather than strict orders: they bias the odds of a given fate without locking it in. In cattle, whose early development resembles that of humans more than that of mice, this suggests that symmetry-breaking—small, unequal distributions of molecules and cell behaviors—starts earlier than visible structures reveal. For assisted reproduction and embryo biopsy in both livestock and potentially humans, the work underscores that not all early cells are interchangeable, and that probing or removing them at these stages might subtly influence how the embryo’s first cell-fate decisions unfold.
Citation: Koyama, H., Mashiko, D., Ferré-Pujol, P. et al. Early transcriptional divergence underlies cell fate bias in bovine embryos. Commun Biol 9, 625 (2026). https://doi.org/10.1038/s42003-026-10198-9
Keywords: bovine embryo development, cell fate bias, single-cell RNA sequencing, trophectoderm lineage, early embryogenesis