Clear Sky Science · en
Glycan-based biological degraders targeting the cytokine immune axis
Turning Off Runaway Inflammation
Many hard-to-treat diseases, from rheumatoid arthritis to certain cancers, are fueled by small messenger proteins called cytokines that keep the immune system switched on. Today’s drugs mostly try to block these messengers, but in severe disease there can simply be too much signal to neutralize. This study explores a different idea: instead of just muffling the signal, can we physically remove these troublesome molecules from the bloodstream by sending them into the body’s cellular “recycling centers” for destruction?
A New Way to Remove Problem Molecules
The researchers focus on interleukin‑6 (IL‑6), a powerful inflammatory cytokine, and its soluble receptor, which together drive many chronic and acute immune disorders. Rather than designing yet another blocking antibody, the team builds “biological degraders,” or BioDegs. These are protein-based tools, such as antibodies or tiny antibody fragments, that first grab hold of IL‑6 or its receptor and then hitch a ride to the liver. There, they use a sugar-based handle to latch onto a natural receptor on liver cells, pulling the cytokine along into the cell’s waste-disposal system for breakdown.
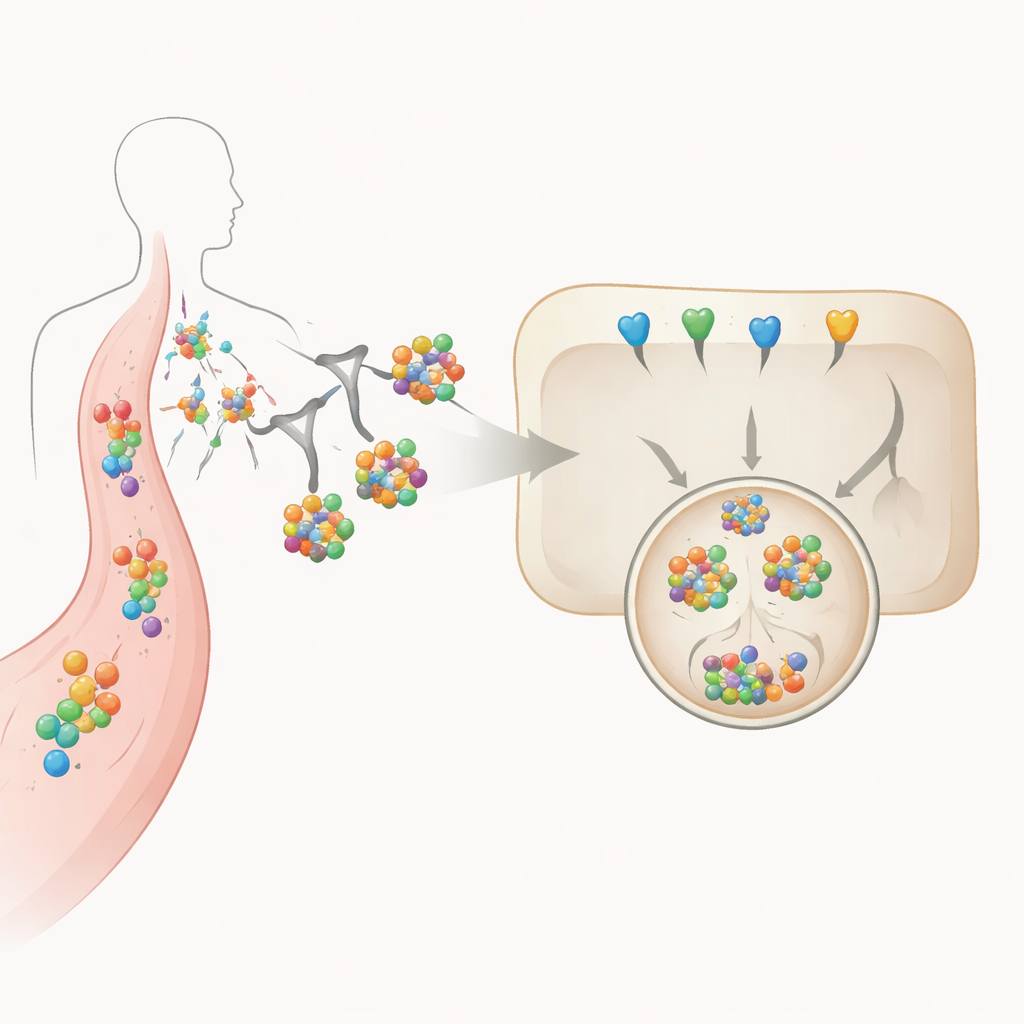
Using Sugars as Precision Delivery Tags
At the heart of this approach is a tri-branched sugar structure attached to the degrader proteins. This sugar is specifically recognized by the asialoglycoprotein receptor, a molecule found mainly on liver cells. When the sugar-tagged degrader binds IL‑6 or its receptor in the bloodstream, the whole complex is drawn to the liver cell surface, engulfed, and shuttled through internal compartments to the lysosome, an acidic vesicle packed with digestive enzymes. The authors systematically vary how many sugar groups are attached and which protein “scaffold” carries them, then measure how these design choices affect stability, binding strength, cellular uptake, and actual destruction of IL‑6 in liver-like cells grown in the lab.
Testing Different Biological Degrader Designs
The team compares several formats: full-size therapeutic antibodies that already target IL‑6 or its receptor, a compact camelid single-domain antibody fragment (known as a VHH) that binds IL‑6, and a soluble version of the IL‑6 receptor repurposed as a decoy. All are chemically decorated with the tri-branched sugar in graded amounts. In cultured liver cancer cells that naturally carry the relevant sugar receptor, the sugar-tagged antibodies and VHH successfully drag IL‑6 or its receptor into the cells and route them to lysosomes. The more sugar groups attached (within limits), the more efficiently the complexes are pulled inside, and the more IL‑6 disappears from the surrounding medium over about a day. Notably, the small VHH-based degrader, despite having fewer sugar attachment sites, proves especially effective at internalizing and clearing IL‑6, likely because its compact shape forms a more favorable complex for uptake.
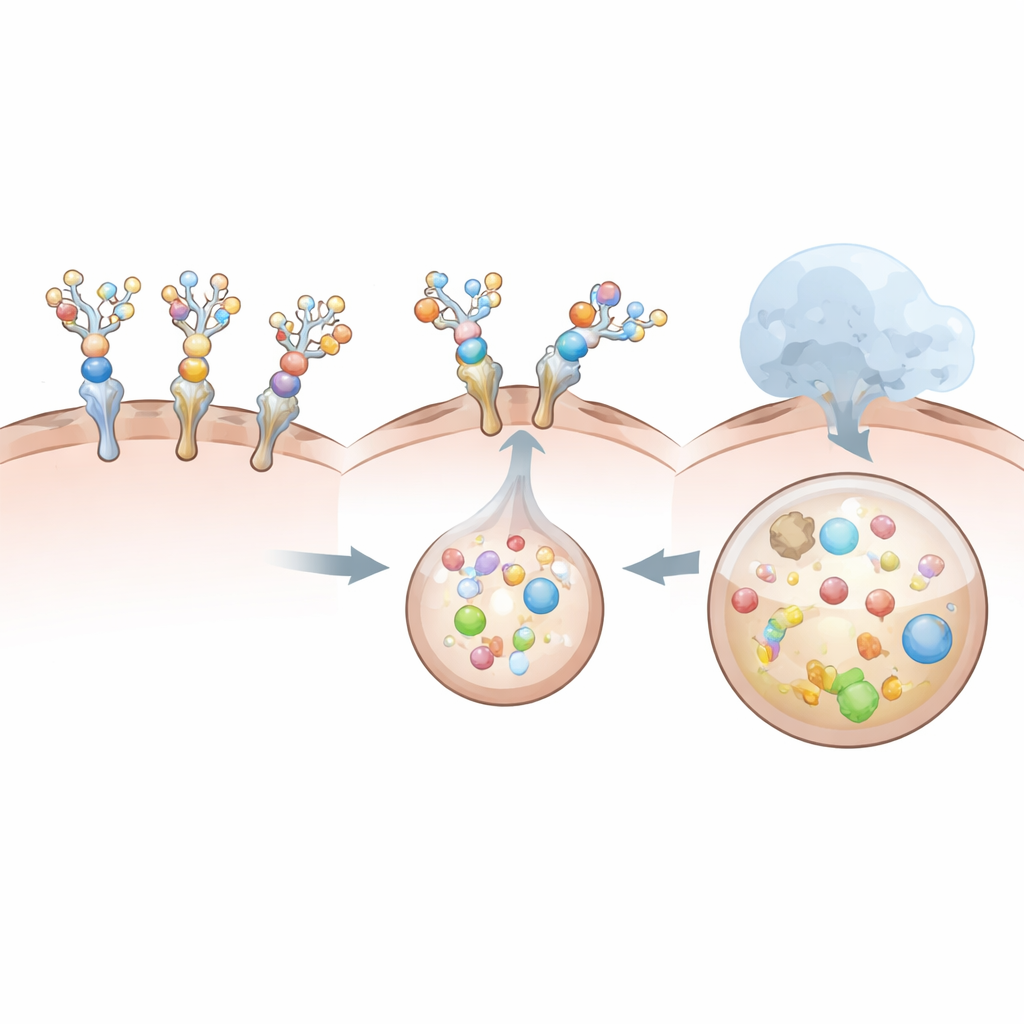
Why Some Designs Work Better Than Others
Not all degraders perform equally well. The soluble receptor-based design, although taken up readily by liver cells, turns out to be a poor escort for IL‑6 itself. Its natural interaction with IL‑6 is relatively weak and short-lived, and heavy sugar decoration further destabilizes this binding. As a result, the decoy receptor gets internalized alone while most IL‑6 remains outside, largely untouched. By contrast, the antibody and VHH degraders bind IL‑6 extremely tightly even after sugar attachment, and still engage the liver receptor strongly enough to ensure that cytokine and degrader are internalized together. These comparisons reveal that simply binding a target is not enough: the overall geometry, size, and cooperation between the degrader, its target, and the liver receptor all shape whether the complex is efficiently swallowed and destroyed.
Looking Ahead to Smarter Inflammation Control
To a non-specialist, the key message is that it may soon be possible not just to block inflammatory signals like IL‑6 but to selectively “vacuum” them out of circulation by redirecting them to the liver’s natural disposal machinery. This study lays out a practical roadmap for designing such glycan-based degraders, showing which molecular features make them most effective and which fall short. In the long run, refining these designs could lead to medicines that more completely clear disease-driving cytokines in conditions such as severe arthritis, cytokine storms, or certain cancers, while sparing healthy tissues by leveraging the liver as a safe, targeted cleanup hub.
Citation: Seifert, M., Kollenkirchen, T., Ernst, A. et al. Glycan-based biological degraders targeting the cytokine immune axis. Commun Biol 9, 530 (2026). https://doi.org/10.1038/s42003-026-10001-9
Keywords: targeted protein degradation, interleukin-6, lysosomal trafficking, extracellular degraders, immunotherapy