Clear Sky Science · en
Riboswitch-controlled lipid conversion enables functional membrane asymmetry in artificial cells
Why Building Fake Cells Matters
Every living cell constantly remodels its outer skin—the membrane—to sense its surroundings, send signals, and stay alive. Yet most artificial cells made in the lab have frozen, unchanging membranes, limiting how lifelike they can be. This paper describes a way to give artificial cells a kind of “programmable skin” that can change its makeup in response to a simple chemical cue. That ability could one day let tiny synthetic cells sense diseases, release drugs on demand, or help scientists probe how real cells work from the outside in.
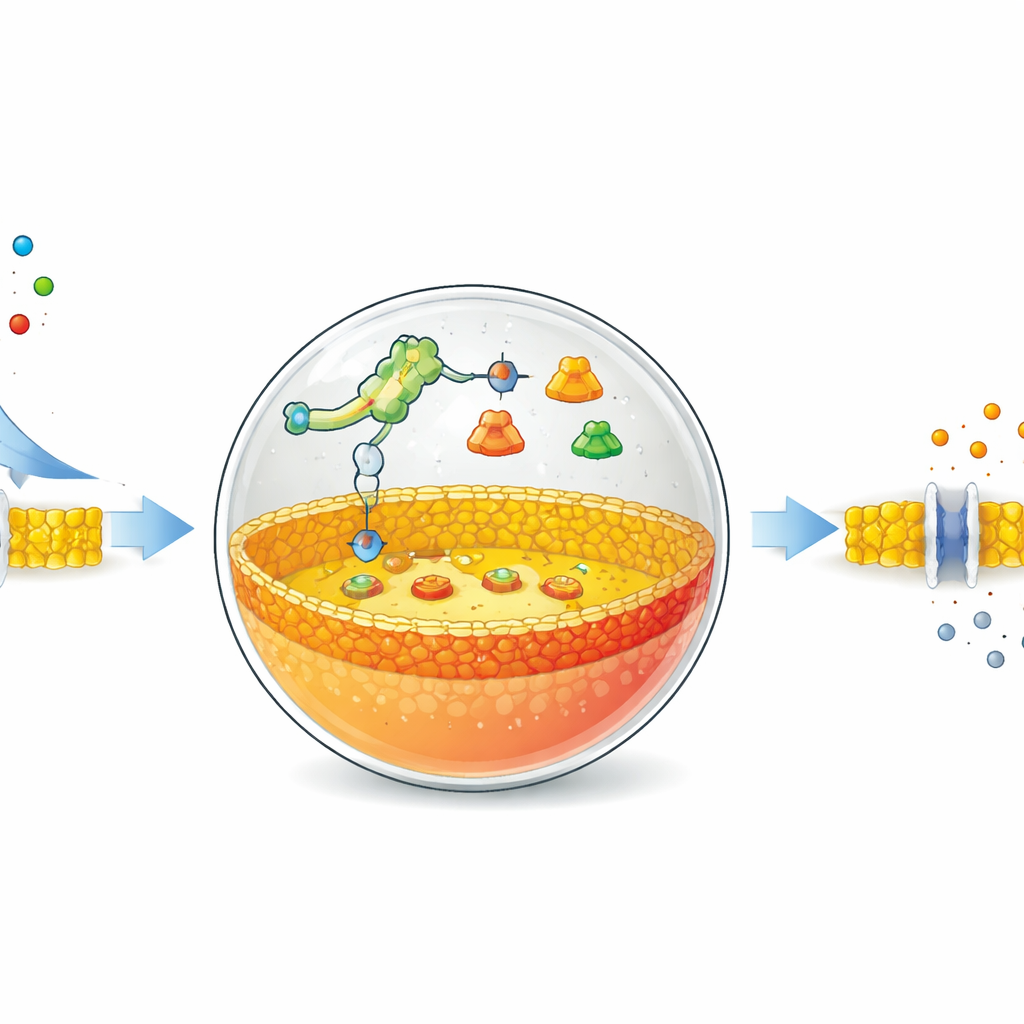
A Switch That Listens to a Simple Salt
The authors start with hollow bubbles made from fat molecules, similar to the ones that form natural cell membranes. Inside these bubbles they place a stripped-down protein‑making system and special DNA instructions. Central to their design is a riboswitch—an RNA segment that changes shape when it binds a particular small molecule and in doing so turns protein production on or off. Here, the riboswitch is tuned to respond to ordinary fluoride ions, such as from sodium fluoride salt. When fluoride slips through the membrane into the bubble, the riboswitch activates the DNA program and orders up production of a specific enzyme.
Rewriting the Membrane from the Inside
The enzyme chosen is phospholipase D, a protein that snips and edits fat molecules. In these artificial cells, the starting membrane is made mostly of a neutral fat called phosphatidylcholine. Newly made phospholipase D works only on the inner half of the membrane, converting some of these neutral fats into negatively charged ones called phosphatidic acid. As a result, the membrane becomes asymmetric: the inner side is more negatively charged than the outer side. Using fluorescent probes that stick to phosphatidic acid, the team tracked this change over time. They showed that by tuning the amount of DNA and fluoride, they could control how quickly and how strongly the inner membrane was remodeled, with significant changes appearing within about an hour.
Holding the Imbalance in Place
In soft, fluid membranes, fat molecules slowly drift from one side to the other, which would erase the carefully created imbalance. To counter this, the researchers added cholesterol, a stiffening component also abundant in human cell membranes. With cholesterol present, the newly formed negative lipids remained mostly on the inner side for at least 90 minutes, while much less of them leaked to the outer side. By comparing bubbles with and without cholesterol, and estimating how many modified lipids appeared on each side, they showed that cholesterol slows the flip‑flop of these molecules and helps preserve long‑lived asymmetry—an important feature of real cell membranes.
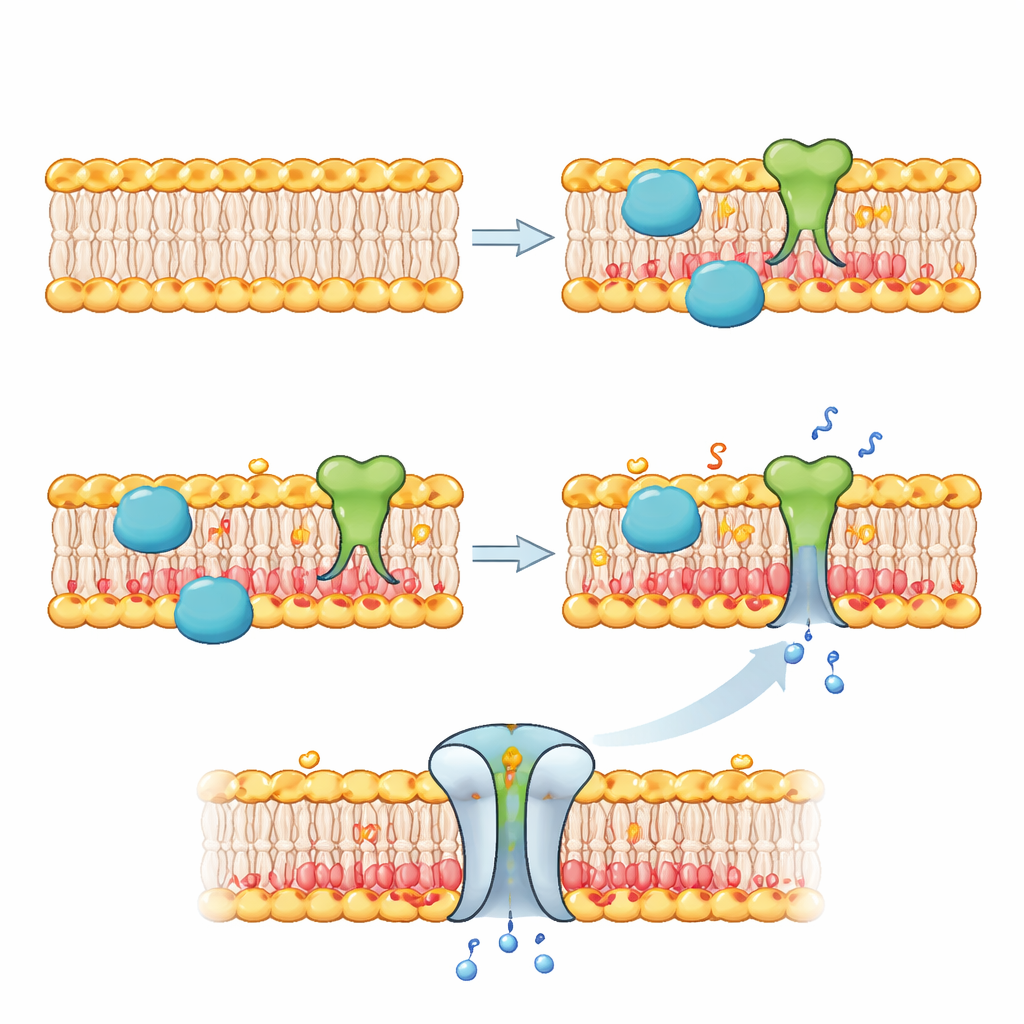
Docking Proteins and Opening Gates
Once they could rewrite the inner membrane, the authors used this new chemistry to control what happens at the boundary of the artificial cell. In one set of experiments, they used a variant activity of the same enzyme to attach “clickable” handles onto inner‑leaflet lipids, then anchored a fluorescent protein to those handles. Only when fluoride was added did the proteins gather along the membrane, demonstrating stimulus‑dependent protein recruitment. In another key test, they added a natural gate protein called MscL to the membrane. This channel tends to open when it senses negative lipids and tension. As phospholipase D built up negative lipids on the inner side after fluoride addition, MscL pores opened and allowed small dye molecules to flow into the bubbles, visibly turning them bright—clear evidence that membrane remodeling could switch a membrane protein from an inactive to an active state.
From Programmable Skins to Smart Synthetic Cells
To a non‑specialist, the core message is that the researchers have wired together a chemical sensor, a genetic switch, and a membrane‑editing enzyme inside tiny fat bubbles. A simple external cue—a bit of fluoride—causes the artificial cell to change the charge pattern of its membrane from the inside, which in turn controls which proteins cling to the surface and whether built‑in gates open or close. This approach turns otherwise passive bubbles into responsive, tuneable objects that more closely resemble living cells. Looking ahead, similar schemes could allow artificial cells to sense signals from tumors, remodel their membranes, and trigger cascades of reactions or drug release, offering a powerful new route to environmentally aware, programmable microscopic devices.
Citation: Kamiya, K., Lee, S. & Baba, K. Riboswitch-controlled lipid conversion enables functional membrane asymmetry in artificial cells. Commun Biol 9, 580 (2026). https://doi.org/10.1038/s42003-026-09890-7
Keywords: artificial cells, riboswitch, lipid membranes, membrane asymmetry, synthetic biology