Clear Sky Science · en
MOF-mediated H4K16ac is critical for blastocyst formation in sheep by shaping promoter accessibility and transcription
Why tiny sheep embryos matter to us
Before a pregnancy can safely take hold, a fertilized egg must transform into a tiny hollow ball of cells called a blastocyst. This brief window, just days after fertilization, is when the first cell fates are decided and when many pregnancies quietly fail. In humans and livestock, being able to grow healthy blastocysts in the lab is central to in vitro fertilization and animal breeding. This study looks inside sheep embryos to understand how a specific chemical tag on DNA-packaging proteins helps embryos successfully reach the blastocyst stage.
A closer look at the first days of life
In mammals, the journey from fertilized egg to blastocyst happens in under a week. During this period, the embryo must first switch on its own genome and then sort its cells into an inner cluster that will form the fetus and an outer shell that will become the placenta. These sweeping changes are guided not only by genes, but also by epigenetic marks—small chemical flags on DNA and its associated proteins that tell genes when to turn on or off. The authors focused on one such mark, called H4K16 acetylation, which is written by an enzyme known as MOF. Earlier work in mice, flies, and human cells had hinted that this mark is tied to active genes and early development, but its role in livestock embryos remained largely unexplored.
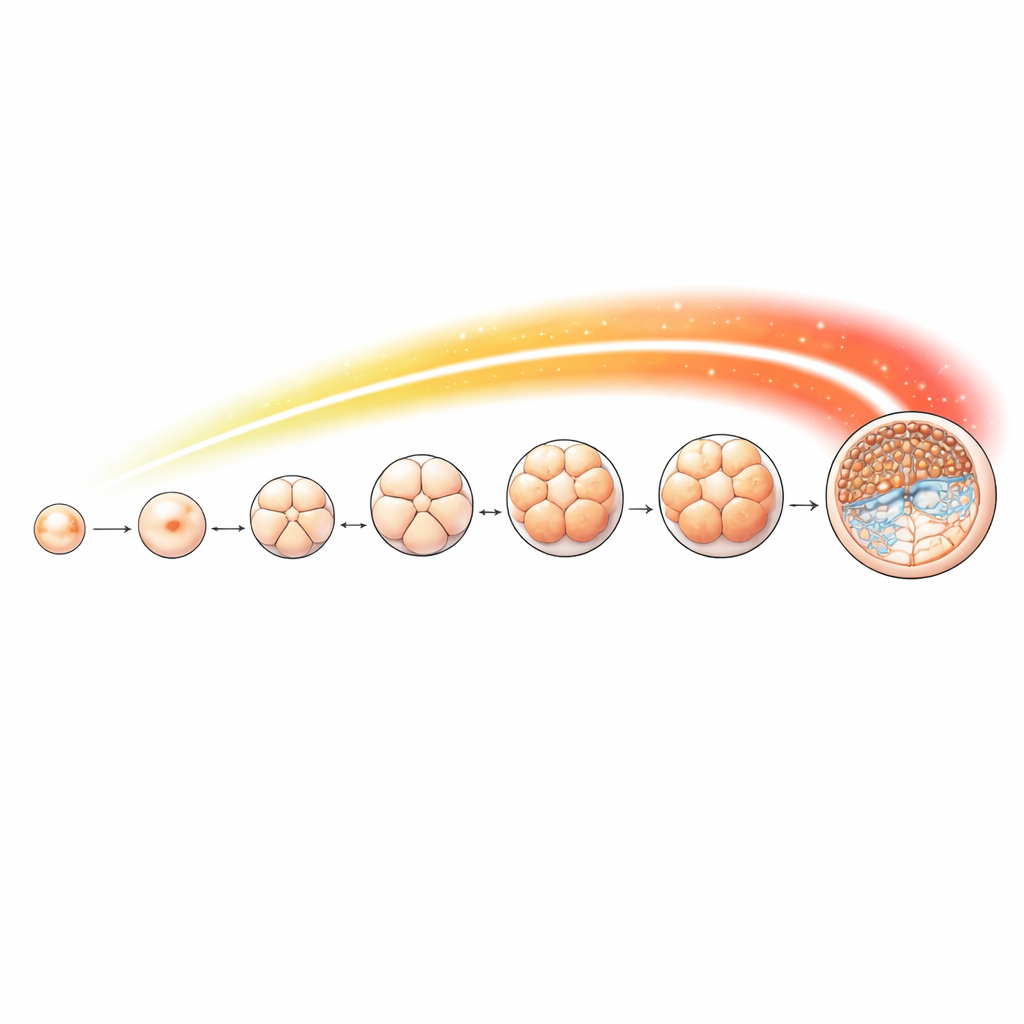
Mapping a chemical signal across early development
To track this mark in detail, the team collected sheep eggs and embryos at key stages, from unfertilized oocytes through 2-, 4-, 8- and 16-cell stages, morula, and finally blastocyst. They used a sensitive method to map where H4K16 acetylation sits along the genome at each step. The mark was present throughout development but changed in a stage-specific way. A particularly sharp rise occurred from the 8-cell stage onward, coinciding with the time when the embryo’s own genes take over control. Many of the sites where the mark accumulated lay near gene start regions that control important cellular processes such as RNA processing, chromosome segregation, and DNA replication—jobs that are especially demanding as cells divide rapidly and specialize.
What happens when the writer enzyme is blocked
To test whether this mark is truly necessary, the researchers used small molecules to inhibit MOF’s activity in early embryos grown in culture. Blocking MOF drastically reduced the fraction of embryos that reached the blastocyst stage and caused many to stall around the 8- to 16-cell phase. When the team examined blastocysts that did form under MOF inhibition, they found widespread disruption of gene activity: thousands of genes were turned up or down compared with untreated controls. Genes that normally carry strong H4K16 acetylation around their starting switches tended to fall silent when that mark was lost. Many of these genes belong to pathways crucial for managing RNA, building ribosomes, and transporting molecules in and out of the nucleus—core tasks for a growing embryo.
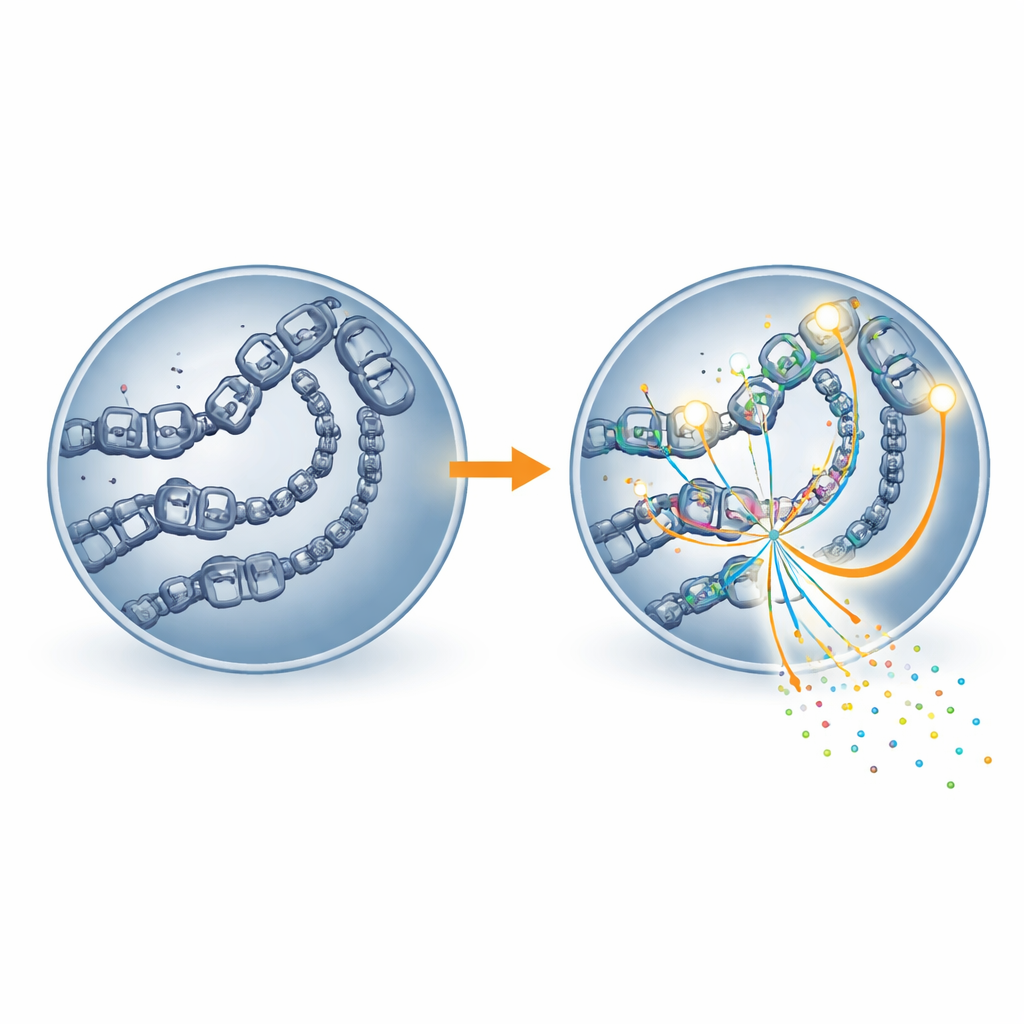
Opening the genome’s doors to transcription
The study went further by looking at how accessible the DNA was and where the cell’s main gene-reading machine, RNA polymerase II, was sitting. Regions enriched in H4K16 acetylation showed more open chromatin—the looser form of DNA packaging that allows molecular machines to reach genes. These same regions also hosted more RNA polymerase II. When MOF was blocked, sites that lost the mark became less accessible and showed reduced occupancy by RNA polymerase II, and the nearby genes were expressed at lower levels. In other words, H4K16 acetylation seems to act like a doorstop that keeps gene switches physically open so the transcription machinery can bind and work efficiently.
Implications for fertility and assisted reproduction
By combining genome-wide maps, gene activity measurements, and functional tests, the authors conclude that MOF-dependent H4K16 acetylation is a critical epigenetic checkpoint in sheep embryos. When this system functions properly, it helps shape open, accessible regions at key gene promoters, draws in RNA polymerase II, and supports the orderly gene programs needed to build a healthy blastocyst. When the system is disrupted, development falters. Because sheep embryos share many features with human embryos and are widely used as models, the findings suggest that this particular epigenetic mark—and the MOF enzyme that writes it—could become a useful indicator of embryo quality and a potential target for improving the success of assisted reproductive technologies.
Citation: Wang, L., Chen, B., Chen, X. et al. MOF-mediated H4K16ac is critical for blastocyst formation in sheep by shaping promoter accessibility and transcription. Commun Biol 9, 609 (2026). https://doi.org/10.1038/s42003-026-09868-5
Keywords: blastocyst development, epigenetic regulation, histone acetylation, RNA polymerase II, assisted reproduction