Clear Sky Science · en
An ontogeny-cytokine code determines macrophage response polarity and tumor outcomes
Why the Body’s ‘Big Eater’ Cells Matter in Cancer
Immune cells called macrophages are among the most abundant cells inside solid tumors. These cellular “big eaters” can either attack cancer or help it spread, and many new treatments aim to push them toward the cancer-fighting side. Yet results in patients have been disappointing, in part because we still lack a clear map of what makes a macrophage helpful or harmful. This study reveals a hidden “code” that links where a macrophage comes from, which signals it sees, and how it ultimately shapes tumor growth and metastasis.
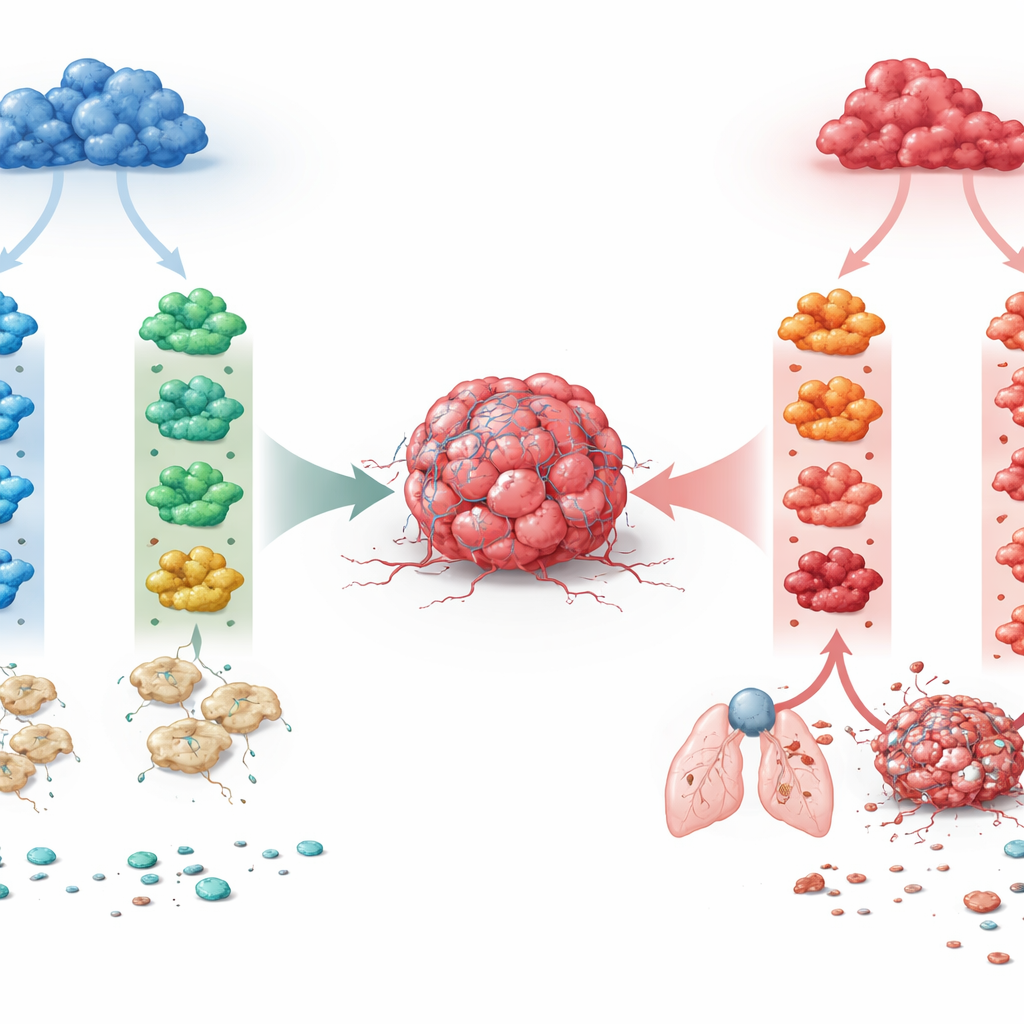
Two Macrophage Families, Many Possible Fates
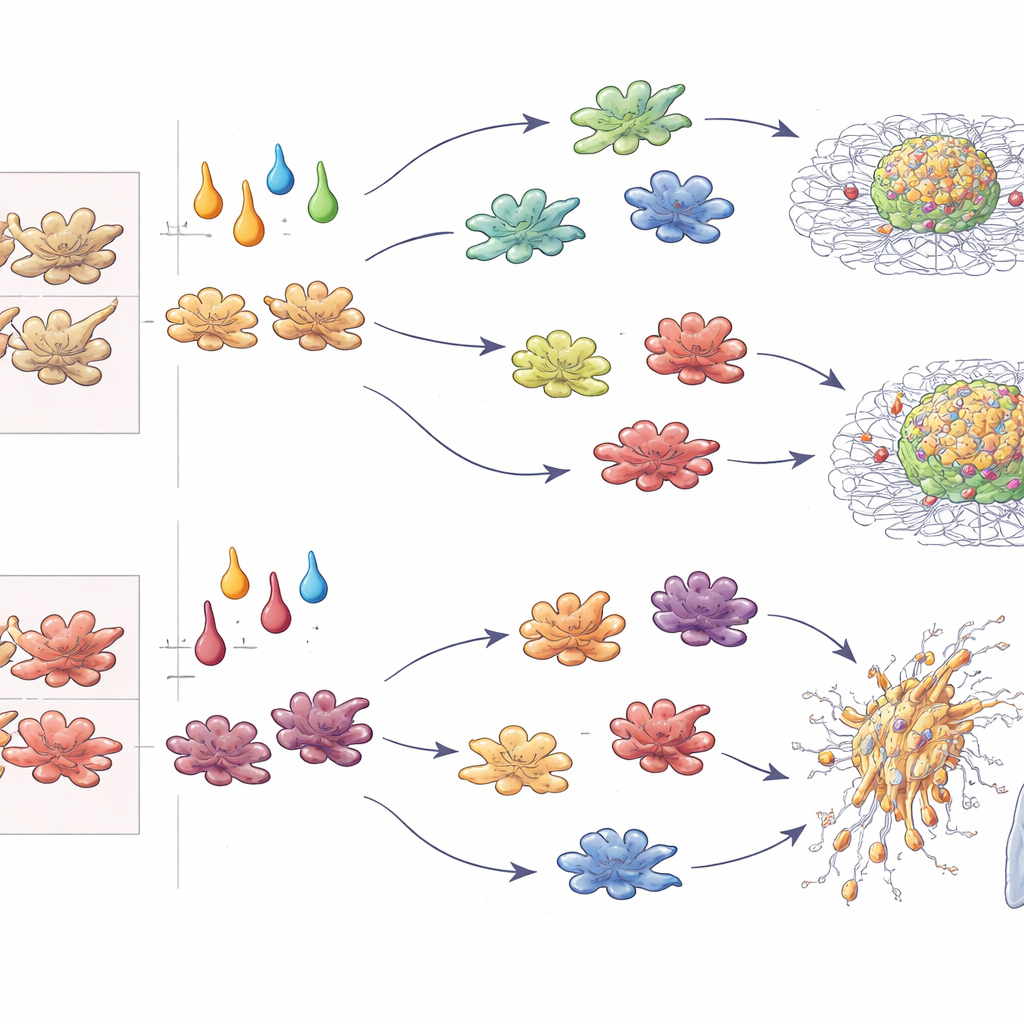
The researchers focused on two major macrophage lineages that exist in many tissues. One lineage depends on a growth factor called M-CSF and tends to maintain everyday tissue balance. The other arises under an inflammatory growth factor, GM-CSF, and is naturally more reactive. From mouse bone marrow, the team grew macrophages along either lineage, then exposed them to four immune signals frequently found in tumors: IFN-γ, IL-4, IL-10, and TGF-β. This produced eight well-defined macrophage states. By reading out global gene activity with bulk and single-cell RNA sequencing, they found that developmental origin was the dominant force: it separated the two lineages more strongly than any single cytokine. The same signal could therefore push the two macrophage families into very different genetic and functional programs.
How the Same Signal Can Flip from Friend to Foe
A key discovery was that IL-4, a cytokine long associated with wound repair and “alternative” activation, had opposite effects depending on macrophage origin. When IL-4 acted on M-CSF–derived macrophages, it triggered gene programs linked to tissue remodeling, induced the enzyme ARG1, gave cells an elongated shape, and reduced their ability to present antigen to T cells. Functionally, these macrophages supported tumor growth, invasion into surrounding matrix, and seeding of lung metastases. In contrast, IL-4 acting on GM-CSF–derived macrophages drove a more inflammatory, dendritic-cell–like state with strong antigen presentation and tumor-suppressive behavior. Thus, the same cytokine could either fuel cancer or help restrain it, depending on the macrophage’s developmental background. A similar, though less dramatic, lineage-dependent split was observed for IL-10, while IFN-γ remained consistently tumor-fighting and TGF-β consistently tumor-promoting across both lineages.
Watching Tumors and Macrophages Shape Each Other
To see how these programmed macrophages influence tumor cells over time, the team built three-dimensional spheroids that mixed fluorescently labeled cancer cells with differently polarized macrophages. Single-cell analysis over several days revealed an intriguing pattern. Macrophages, regardless of their starting state, gradually converged on a common “tumor-educated” profile dominated by oxidative stress responses. In contrast, tumor cells diversified. In the presence of control or IFN-γ–polarized macrophages, cancer cells kept dividing but showed limited invasive behavior. When co-cultured with IL-4– or TGF-β–treated M-CSF macrophages—or with IL-10/TGF-β–treated GM-CSF macrophages—tumor cells shifted toward an epithelial-to-mesenchymal, invasion-ready identity. Even though the macrophages’ gene signatures were later reshaped by the tumor, their initial programming had already steered cancer cells onto very different trajectories.
From Lab Spheroids to Spreading Tumors
Beyond gene readouts, the authors quantified how fast tumor spheroids grew and how deeply they invaded a surrounding gel. In 3D culture, M-CSF macrophages treated with IL-4 or TGF-β strongly boosted tumor expansion and radial invasion, while IFN-γ–polarized cells killed or constrained tumors. Strikingly, when the same cytokines acted on GM-CSF macrophages, IL-4 switched roles and now suppressed tumor growth, whereas IL-10 and TGF-β drove the most aggressive behavior. Injecting mixed spheroids into mice confirmed these patterns in vivo: TGF-β–conditioned macrophages promoted lung metastases regardless of origin; IFN-γ–conditioned ones consistently limited them; IL-4 either enhanced or blocked metastasis depending on whether macrophages came from the M-CSF or GM-CSF lineage. By combining growth, invasion, metastasis, and T-cell stimulation into standardized scores, the authors distilled these patterns into an “ontogeny–cytokine code” that predicts macrophage impact on tumors.
What This Means for Future Cancer Therapies
For non-specialists, the main message is that not all tumor-associated macrophages are created equal, and their behavior cannot be captured by a simple good-versus-bad or M1-versus-M2 label. Instead, this work shows that a macrophage’s developmental roots and its local cytokine environment interact like digits in a combination lock to decide whether it will nourish a tumor or help destroy it. Therapies that ignore this code—for example, by broadly depleting macrophages or delivering a cytokine without regard to lineage—may blunt their own effectiveness or even backfire. In the future, profiling whether a tumor is dominated by M-CSF or GM-CSF signaling, and then tailoring cytokine-blocking or cytokine-delivering drugs accordingly, could turn macrophages from uncertain allies into reliable partners in cancer treatment.
Citation: Schaer, D.J., Schulthess-Lutz, N., Peterhans, M.J. et al. An ontogeny-cytokine code determines macrophage response polarity and tumor outcomes. Commun Biol 9, 592 (2026). https://doi.org/10.1038/s42003-026-09853-y
Keywords: tumor-associated macrophages, cytokine signaling, cancer immunotherapy, tumor microenvironment, metastasis