Clear Sky Science · en
Intracellular complement Factor H promotes tumor progression through modulation of cell cycle and actin cytoskeleton
Why this hidden cancer helper matters
Cancer is often described as a battle between growing tumors and the immune system. This study reveals that one of the immune system’s own molecules, a protein called Factor H, can quietly switch sides. Instead of only working in the bloodstream to control inflammation, Factor H is found inside tumor cells and nearby support cells, where it helps cancers grow, survive, and potentially spread. Understanding this unexpected “double life” opens up new ways to predict how aggressive a tumor will be and how to design smarter therapies.
A familiar protein in an unexpected place
Factor H is best known as a guardian of the complement system, a part of innate immunity that helps clear microbes and damaged cells from the blood. High levels of Factor H around tumors had already been linked to worse outcomes in several cancers, including clear cell kidney cancer. Yet its standard, blood‑borne role could not fully explain why patients whose tumors overproduce Factor H tend to fare poorly. By combining patient data, single‑cell sequencing, and cell biology, the authors show that many cells inside kidney tumors—especially cancer-associated fibroblasts and malignant kidney cells—actively make their own Factor H and keep a portion of it inside the cell rather than secreting it.
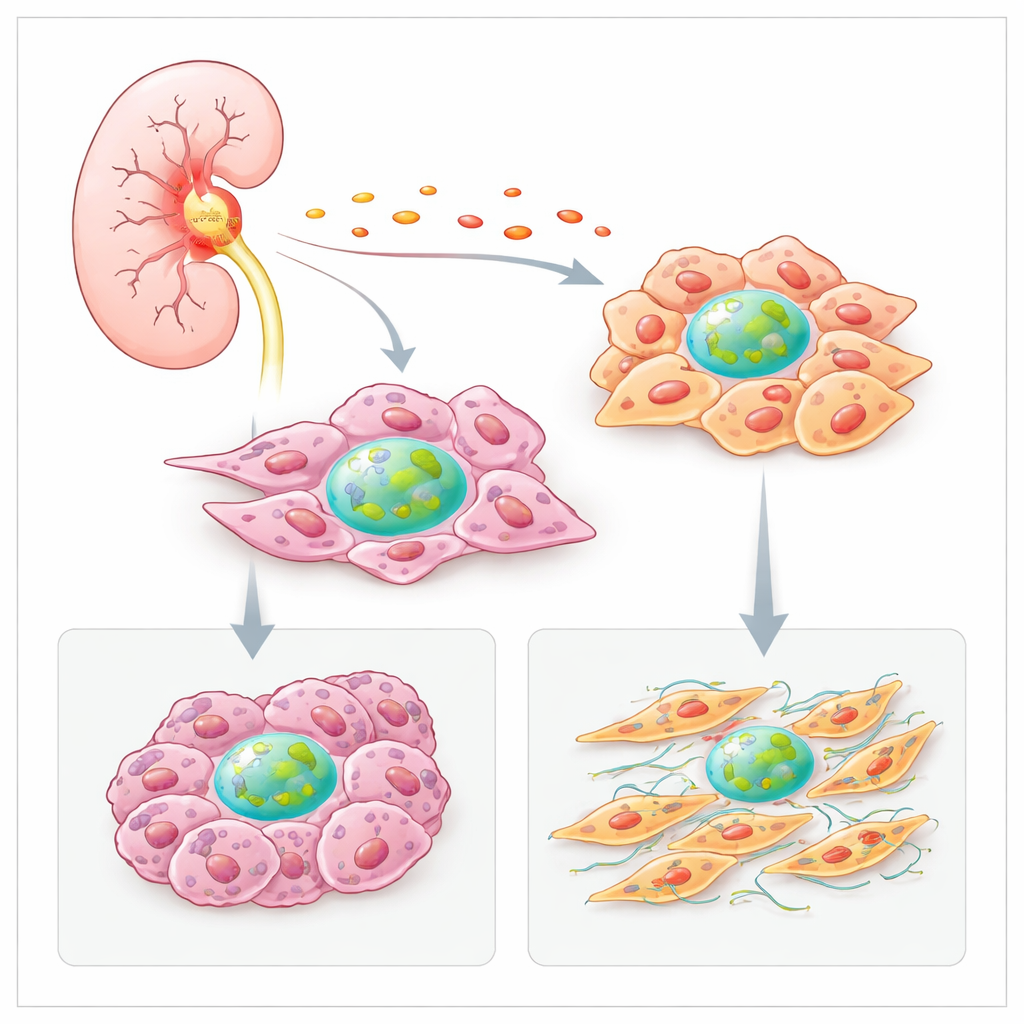
Inflammation turns Factor H production up
The team first asked why tumors produce more Factor H than healthy kidney tissue. They analyzed large cancer datasets and spatial maps of tumor slices and found that spots rich in fibroblasts and cancer cells also contained high levels of the Factor H gene. These hot areas frequently overlapped with signatures of inflammatory molecules, such as the cytokine IL‑6. In cultured kidney cancer cells, IL‑6 strongly boosted Factor H production, while other inflammatory signals increased it in fibroblasts. This suggests that the inflamed tumor environment itself pushes local cells to crank up their own Factor H, reinforcing a vicious circle of growth and survival.
Inside the nucleus, helping cells ignore stop signals
Looking inside cells, the researchers discovered a smaller, intracellular form of Factor H that appears in several compartments, including the nucleus. There, it physically interacts with key controllers of the cell cycle, particularly a transcription factor called E2F3 that helps drive cells from resting to dividing states. When the Factor H gene was silenced, fibroblasts and cancer cells slowed their replication, accumulated in the resting phase, and showed more active p53, a well‑known “guardian” protein that can halt growth or trigger cell death. Importantly, simply adding normal, secreted Factor H from the outside did not rescue this effect, underscoring that the internal pool is what keeps p53 in check and allows continuous proliferation.
Reshaping the cell’s skeleton for growth and movement
In kidney cancer cells, but not in fibroblasts, intracellular Factor H also bound components of the actin capping complex, which regulates how actin filaments grow and stabilize the cell’s internal scaffolding. When Factor H was silenced, cancer cells became rounder, lost their organized stress fibers, and struggled to form compact three‑dimensional spheroids—an experimental stand‑in for tumor growth and cohesion. Silencing both Factor H and the capping protein did not worsen the defects, suggesting they act in the same pathway. These changes occurred even when p53 was blocked, indicating that Factor H independently supports both cell division and the structural changes that can favor invasion and metastasis.
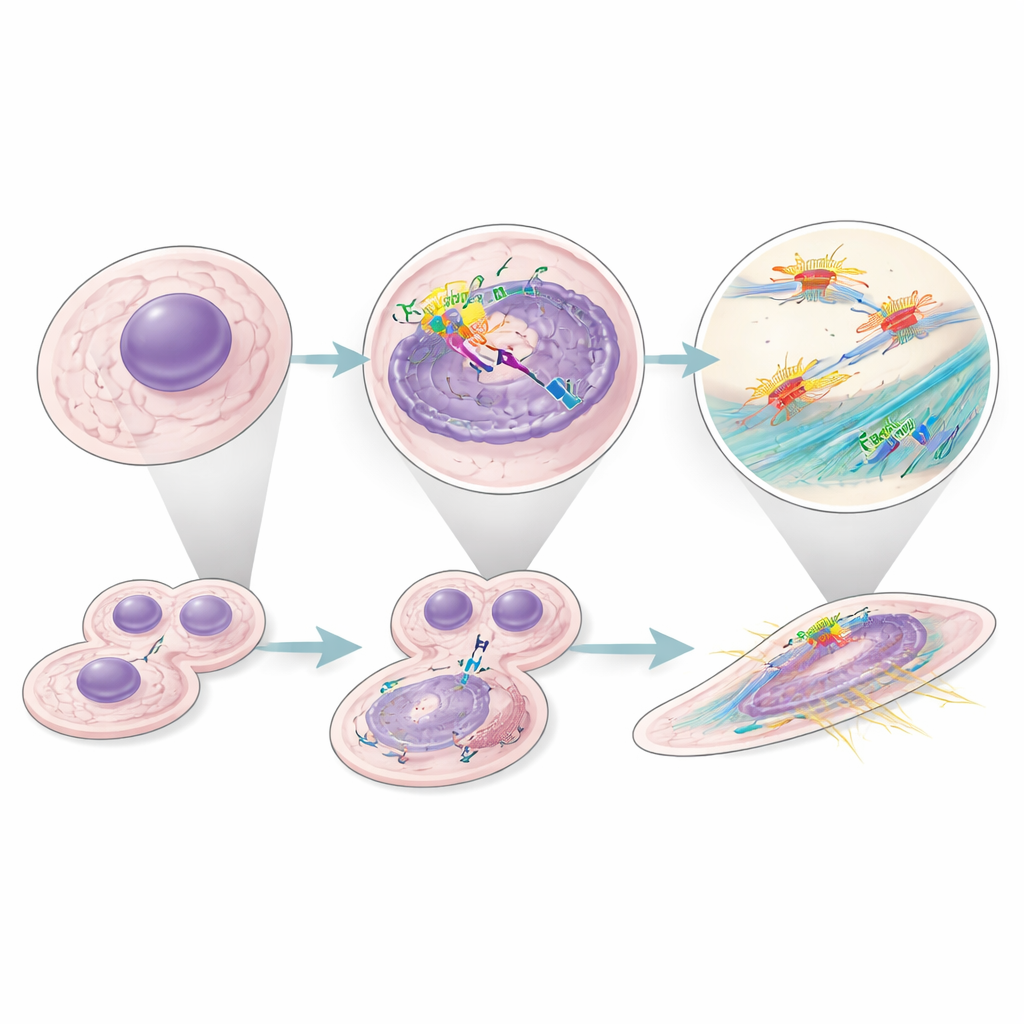
A wider impact across many cancers
To see whether this behavior was unique to kidney tumors, the authors mined many single‑cell and bulk RNA datasets from human cancers. In tumors where high Factor H expression predicted poor survival, fibroblast signatures were also linked to worse outcomes. Within kidney tumors, they identified a specific malignant cell state that strongly expressed Factor H, showed active cell‑cycle programs, and was associated with shorter patient survival. Together, these findings suggest that intracellular Factor H acts as a broad pro‑tumor agent, operating inside both cancer cells and their supportive fibroblasts in multiple cancer types.
What this means for patients and therapies
This work overturns the idea that Factor H is only a circulating regulator of immune reactions. Instead, it emerges as a multitasking, intracellular helper that encourages tumors to grow by easing cells through the cycle of division and fine‑tuning their internal scaffolding for movement. This helps explain why overexpression of Factor H is such a strong warning sign in cancer. It also means that drugs designed to block Factor H on the tumor surface or in the blood may not fully disarm its hidden, internal functions. Future therapies will likely need to target both its external and internal roles to effectively slow tumor progression.
Citation: Rezola Artero, M., Minery, A., Revel, M. et al. Intracellular complement Factor H promotes tumor progression through modulation of cell cycle and actin cytoskeleton. Commun Biol 9, 524 (2026). https://doi.org/10.1038/s42003-026-09807-4
Keywords: complement factor H, clear cell renal cell carcinoma, cancer-associated fibroblasts, tumor microenvironment, cell cycle regulation