Clear Sky Science · en
Delphi consensus recommendations for preventing and treating cardiac implantable electronic device infections beyond current guidelines
Why Protecting Heart Devices Matters
More and more people rely on implanted heart devices—such as pacemakers and defibrillators—to keep their heartbeat steady and prevent dangerous rhythm problems. While these gadgets save lives, they can also become the focus of serious infections that are hard to diagnose, difficult to treat, and sometimes fatal. This article describes how an international group of heart and infection specialists joined forces to agree on practical, day‑to‑day steps doctors can take to better prevent and manage these infections, especially in situations where formal guidelines are vague or silent.
How Experts Reached Common Ground
The authors used a structured process called the Delphi method to gather and refine expert opinion. Twenty seasoned clinicians from several European countries, all experienced in caring for patients with cardiac implantable electronic devices (CIEDs), took part. They began by identifying real‑world problems that existing guidelines do not fully resolve, such as how to handle infections in very frail patients or when it is safe to re‑implant a device after a serious bloodstream infection. The panel then rated a series of statements over multiple anonymous voting rounds. This approach was chosen to reduce the influence of seniority or loud voices and to highlight true areas of agreement and disagreement.
Simple Sterile Habits With Big Impact
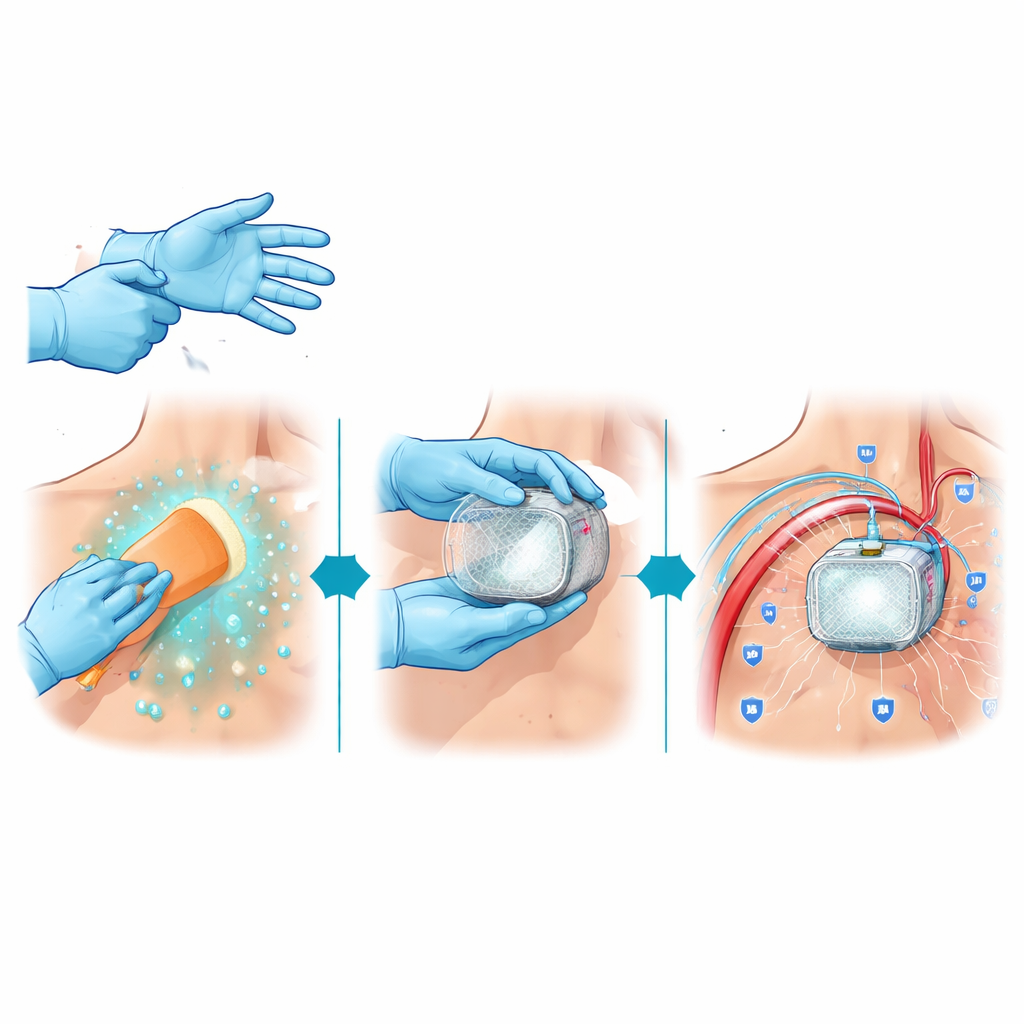
One of the clearest messages from the panel is that small changes in operating‑room habits can make a meaningful difference. The experts strongly agreed that surgeons implanting or replacing heart devices should wear two pairs of sterile gloves and change the outer pair after the patient is covered with drapes, before touching the new device or its leads. Studies show that double‑gloving greatly reduces hidden glove tears and contamination with blood, which in turn lowers the chance that germs will reach the device pocket. The panel also favored routinely securing patients with straps on the procedure table to limit sudden movements that could disrupt the sterile field or dislodge freshly placed leads.
Tailoring Protection to Each Patient
Another strong theme is that not every patient has the same infection risk. The experts endorsed the use of scoring tools that combine simple clinical factors—such as prior procedures or other illnesses—to estimate a person’s chance of developing a device infection. When risk is high, they recommend additional safeguards like antibiotic‑releasing envelopes that surround the device at the time of implantation. The group also supported the cautious use of taurolidine solutions, a type of antimicrobial fluid with activity against a broad range of microbes and their protective biofilms, as an extra layer of defense during procedures. Together, these measures reflect a more personalized, risk‑based strategy rather than a one‑size‑fits‑all approach.
Choices When Removing a Device Is Hard
Completely removing an infected device system is often the best way to cure infection, but in some frail patients or in technically difficult cases this may be unsafe or impossible. For these challenging situations, the panel favored decisions made by a multidisciplinary “heart team” that includes cardiologists, surgeons, and infection specialists, and that takes the patient’s preferences into account. Options may include careful surgical cleaning of the pocket, repositioning the device under the chest muscle, and using targeted local or regional antibiotics, sometimes combined with taurolidine washing. The experts also moderately supported earlier re‑implantation after bloodstream infection has cleared, and same‑session re‑implantation on the opposite side of the chest in patients who critically depend on pacing, provided blood cultures and heart ultrasound show no deeper infection.
Unanswered Questions and Practical Takeaways
Not every proposal won strong backing. The panel did not agree that heart device procedures should be limited to only very high‑volume centers, reflecting concerns about access to care and recognition that operator skill is not defined by a single number. Only moderate support emerged for routinely using nerve blocks to reduce pain or special iodophor‑coated drapes to further sterilize the skin, mainly because data specific to heart devices remain limited. Overall, the experts offer a pragmatic checklist of practices that go beyond current guidelines: reinforce meticulous sterile technique, stratify patients by infection risk, consider modern protective tools and solutions, and discuss complex cases in a team. For patients, the message is that doctors are refining how they implant and manage heart devices to make these life‑saving technologies safer and to continually push infection rates as low as possible.
Citation: Baldauf, B., Bode, K., Biffi, M. et al. Delphi consensus recommendations for preventing and treating cardiac implantable electronic device infections beyond current guidelines. Sci Rep 16, 13135 (2026). https://doi.org/10.1038/s41598-026-49515-x
Keywords: cardiac implantable electronic devices, device infection prevention, pacemaker and defibrillator care, Delphi expert consensus, taurolidine and antibiotic envelopes